Efficient Visible‑Light Photocatalysis via WS₂/Bi₂MoO₆ Heterostructures: A Simple Solvothermal Approach
We report a facile solvothermal synthesis of WS₂/Bi₂MoO₆ heterostructured photocatalysts that harness visible light (>420 nm) for efficient rhodamine B (RhB) degradation. Pre‑exfoliated WS₂ nanoslices serve as a 2‑D substrate, onto which Bi₂MoO₆ nanoflakes grow, forming a tight nanojunction that promotes rapid charge separation and transport. Comprehensive characterization by XRD, XPS, SEM, TEM/HRTEM, and UV‑vis DRS confirms the phase purity, intimate interfacial contact, and enhanced light absorption. Photocatalytic tests reveal that the 5 wt% WS₂/Bi₂MoO₆ composite achieves a 95% RhB removal in 100 min, outperforming pure Bi₂MoO₆ and mechanical blends. Hole (h⁺) trapping studies identify h⁺ as the primary oxidant. Repeated cycling demonstrates structural stability, underscoring the composite’s potential for environmental remediation. Photocatalysis offers a clean pathway for pollutant degradation, yet conventional wide‑band‑gap semiconductors such as TiO₂ and ZnO are limited to UV activation [1,2]. To exploit the abundant solar spectrum, narrow‑band semiconductors with efficient visible‑light absorption are essential [3–6]. However, rapid recombination of photo‑induced carriers often hampers their performance [7]. Constructing heterojunctions introduces internal electric fields that suppress recombination and enhance charge separation [8,9], making them attractive for visible‑light photocatalysis. Bi₂MoO₆, a layered oxohalide, has emerged as a promising visible‑light photocatalyst due to its sandwiched crystal structure [10,11]. Its intrinsic carrier recombination limits its activity, but coupling with two‑dimensional (2‑D) materials can strengthen interfacial charge transfer [12–15]. Transition metal dichalcogenides (TMDs), particularly few‑layer WS₂, exhibit unique electronic properties and high surface areas, positioning them as ideal sensitizers for heterojunction photocatalysts [16–20]. While MoS₂/Bi₂MoO₆ hybrids have shown superior RhB degradation [21], WS₂/Bi₂MoO₆ systems remain unexplored. In this study, we present a streamlined solvothermal route to WS₂/Bi₂MoO₆ heterostructures using pre‑exfoliated WS₂ nanoslices. We demonstrate that the resulting composites achieve superior visible‑light photocatalytic activity, elucidate the interfacial charge dynamics, and confirm long‑term stability. Commercial WS₂ powder (Aladdin Industrial Corporation) was dispersed in 20 mL of a 40 % (v/v) ethanol/water mixture. After 10 h of probe sonication, the suspension was centrifuged at 3000 rpm for 20 min to remove unexfoliated aggregates. The clear supernatant, containing few‑layer WS₂ nanoslices, was collected. UV‑vis absorption at 630 nm, analyzed via the Lambert–Beer law, yielded a concentration of 0.265 ± 0.02 mg mL⁻¹. In a typical solvothermal procedure, 2 mmol of Bi(NO₃)₃·5H₂O and Na₂MoO₄·2H₂O (Bi/Mo = 2:1) were dissolved in 10 mL of ethylene glycol under stirring. Separately, an aliquot of exfoliated WS₂ nanoslices was dispersed in 20 mL ethanol and sonicated for 45 min. This dispersion was then added to the metal precursor solution, stirred for 10 min to achieve homogeneity, and transferred to a 50 mL Teflon‑lined autoclave. After heating at 160 °C for 10 h, the autoclave was cooled naturally. The solid product was washed with ethanol and deionized water, then dried at 80 °C in a vacuum oven for 6 h. WS₂ loadings of 1, 3, 5, and 7 wt% were prepared; a Bi₂MoO₆ reference (no WS₂) was also synthesized under identical conditions. Powder XRD (Bruker D8, Cu‑Kα, λ = 1.5406 Å) identified crystalline phases. SEM (JEOL JSM‑6701F) and TEM (JEOL 2100) examined morphology; HRTEM (JEOL 2100) resolved lattice fringes. UV‑vis diffuse reflectance (Cary 500, integrating sphere) measured optical absorption (200–800 nm). XPS (Shimadzu, Al‑Kα) determined surface elemental states. 50 mg of catalyst was dispersed in 50 mL of 10 mg L⁻¹ RhB. After 30 min in the dark (to reach adsorption equilibrium), the suspension was irradiated with a 300 W Xe arc lamp filtered by a 420 nm cut‑off filter (power density = 150 mW cm⁻²). At 10 min intervals, 2 mL aliquots were withdrawn, centrifuged, and analyzed at 553 nm (Shimadzu UV‑2550). Photocatalytic degradation followed a pseudo‑first‑order kinetic model:
$$
\ln(C_0/C)=k t
$$
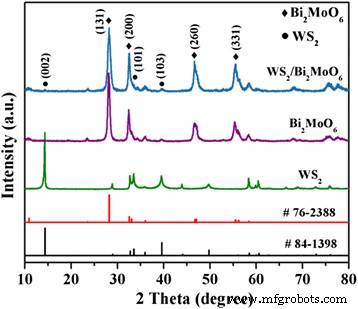
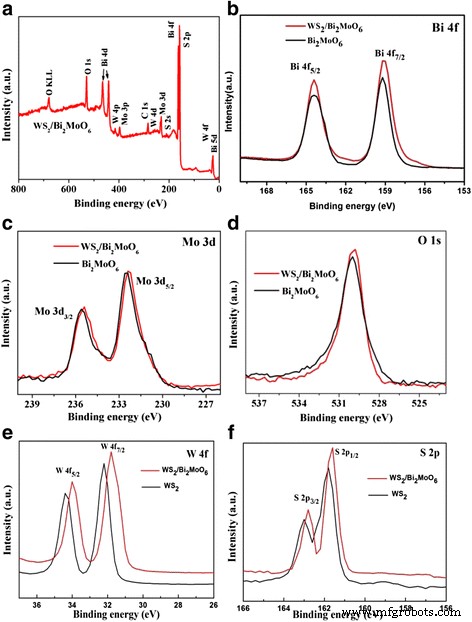
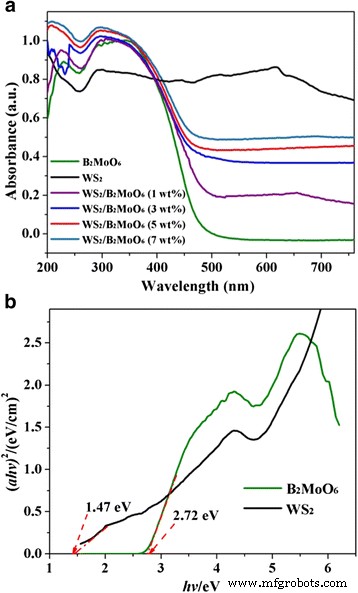
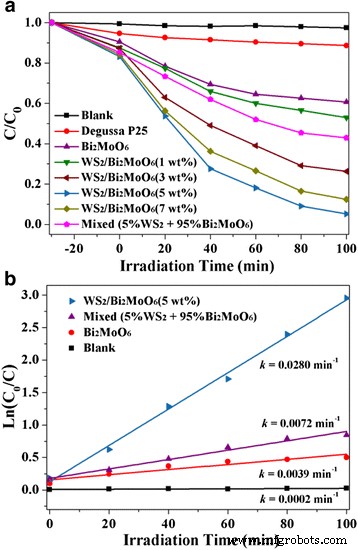
where k is the rate constant. Scavenger tests (IPA, BQ, EDTA, N₂) identified the active species. XRD patterns confirm the coexistence of hexagonal WS₂ (JCPDS 84‑1398) and orthorhombic Bi₂MoO₆ (JCPDS 76‑2388) without impurity peaks, indicating successful heterostructure formation. SEM images (Fig. 2) show that raw WS₂ forms multilayered flakes (~20 µm), while exfoliation yields 2‑D sheets (tens of nm to 1–2 µm). Pure Bi₂MoO₆ appears as microspheres composed of nanoplates; when deposited on WS₂ (Fig. 2d), the nanoplates uniformly coat the WS₂ sheets, establishing a hierarchical architecture. X-ray diffraction patterns of Bi₂MoO₆, few‑layer WS₂, and WS₂/Bi₂MoO₆ (5 wt%) composite TEM/HRTEM (Fig. 3) reveals WS₂ nanosheets (purple arrows) with a lattice spacing of 0.227 nm (WS₂ (103)) and Bi₂MoO₆ nanoplates (50–100 nm) exhibiting 0.274 nm fringes (Bi₂MoO₆ (200)). The coherent interface confirms efficient interfacial charge transfer. TEM (a) and HRTEM (b, c) images of WS₂/Bi₂MoO₆ (5 wt%) composite Survey spectra (Fig. 4a) detect W, S, O, Bi, Mo, and C, with no contaminants. Bi 4f, Mo 3d, and O 1s peaks confirm Bi³⁺ and Mo⁴⁺ oxidation states. Slight binding‑energy shifts (~0.2 eV) in WS₂/Bi₂MoO₆ versus pure components indicate strong electronic coupling at the interface. Survey XPS spectra of the WS₂/Bi₂MoO₆ composite (a) and high‑resolution spectra of Bi 4f (b), Mo 3d (c), O 1s (d), W 4f (e), and S 2p (f) UV‑vis DRS (Fig. 5a) shows that pure Bi₂MoO₆ absorbs up to ~450 nm, while WS₂/Bi₂MoO₆ composites exhibit a pronounced red‑shift and stronger absorption extending to 800 nm, especially at 3–7 wt% WS₂. Band‑gap energies, derived from Tauc plots (Fig. 5b), are 1.47 eV for WS₂ and 2.72 eV for Bi₂MoO₆. a UV‑vis diffuse reflectance spectra. b Tauc plots (αhv)² vs. hv. Under visible light, pure Bi₂MoO₆ degrades ~39 % of RhB in 100 min, whereas WS₂/Bi₂MoO₆ composites achieve 48 % (1 wt%), 74 % (3 wt%), 95 % (5 wt%), and 88 % (7 wt%). The 5 wt% sample outperforms a mechanically blended 5 %/95 % mixture, evidencing the role of intimate heterojunctions. Photocatalytic activity (a) and kinetic fit (b) for RhB degradation. Rate constants (k) derived from pseudo‑first‑order kinetics: 0.0280 min⁻¹ for WS₂/Bi₂MoO₆ (5 wt%)—a 3.8‑fold and 7.1‑fold increase over blended and pure Bi₂MoO₆, respectively. UV‑vis spectra of RhB during degradation over WS₂/Bi₂MoO₆ (5 wt%). Peak shifts from 552 nm to 537 nm indicate stepwise de‑ethylation and eventual mineralization. Four consecutive cycles of RhB degradation show negligible loss of activity for WS₂/Bi₂MoO₆ (5 wt%). XRD after cycling (Fig. 8b) confirms preserved crystal structure, indicating robust operational stability. a Cycling runs for RhB degradation. b XRD patterns before and after four cycles. Scavenger experiments (Fig. 9) reveal that h⁺ are the dominant oxidants; ·OH and ·O₂⁻ play minor roles. Band‑edge calculations (E_CB, E_VB) yield +0.43 eV (WS₂ CB) and –0.31 eV (Bi₂MoO₆ CB), facilitating electron transfer from Bi₂MoO₆ to WS₂ and prolonging hole lifetime on Bi₂MoO₆ VB (+2.41 eV). The CB of WS₂ (+0.43 eV) is less negative than the O₂/·O₂⁻ redox potential (–0.046 eV), so ·O₂⁻ formation is limited; however, Bi₂MoO₆ CB can reduce O₂ to ·O₂⁻. Overall, holes are the primary species driving RhB oxidation, with minor contribution from ·O₂⁻. Rate constants (k_app) for RhB degradation with different scavengers. Proposed photocatalytic mechanism of WS₂/Bi₂MoO₆ under visible light (>420 nm). We have demonstrated a simple solvothermal route to WS₂/Bi₂MoO₆ heterostructures that exhibit exceptional visible‑light photocatalytic activity and robust stability. The intimate WS₂/Bi₂MoO₆ interface promotes rapid electron transfer, suppresses recombination, and extends light absorption into the visible spectrum, enabling 95 % RhB removal in 100 min. The composite’s structural integrity after multiple cycles underscores its practical potential for environmental remediation.Abstract
Background
Methods
Preparation of Few‑Layer WS₂ Nanoslices
Synthesis of WS₂/Bi₂MoO₆ Composites
Characterization
Photocatalytic Activity Assessment
Results and Discussion
Microscopic and Structural Characterization

XPS Analysis
Optical Properties
Photocatalytic Performance
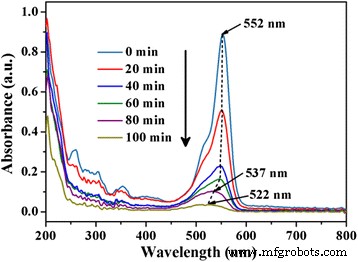
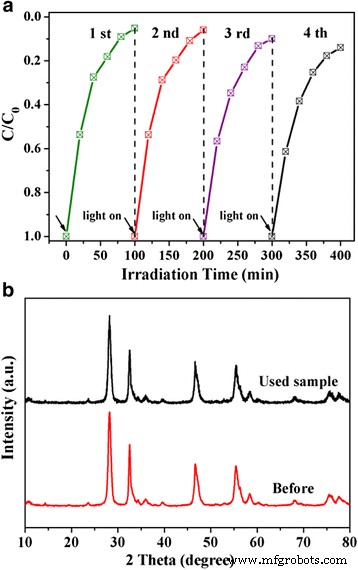
Stability Tests
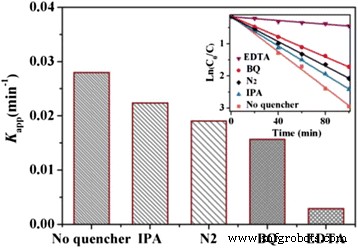
Mechanistic Insight
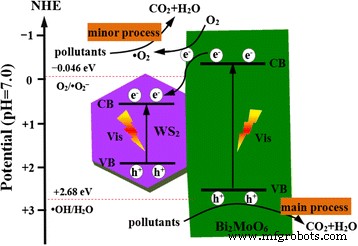
Conclusions
Nanomaterials
- Efficient Synthesis and Optical Characterization of Sub‑Micron Selenium Nanocrystals and Nanorods
- Sol‑Gel Fabricated SiO₂@C/MWNT Nanocomposites Deliver Superior Li‑Ion Battery Anodes
- One‑Pot Synthesis of Color‑Tunable, Conductive CuS‑Coated CuSCN Composites
- High‑Performance Flexible Transparent Electrodes from Silver Nanowires with Tailored Aspect Ratios
- One‑Pot Synthesis of Cu₂ZnSnSe₄ Nanoplates and Their Efficient Visible‑Light Photocatalytic Degradation of Rhodamine B
- Polycatechol‑Modified Fe<sub>3</sub>O<sub>4</sub> Magnetic Nanoparticles: A Highly Selective, Magnetically Recoverable Adsorbent for Cationic Dye Removal
- Low‑Cost Hydrothermal Synthesis of Ultralong Copper Nanowires for Flexible Transparent Conductive Electrodes
- Ligand‑Free Iridium Nanoparticles: A Simple Aqueous Synthesis and Demonstrated In‑Vitro Biocompatibility
- One‑Pot Room‑Temperature Synthesis of 10‑nm 2D Ruddlesden–Popper Perovskite Quantum Dots with Tunable Emission
- 5‑Minute Microwave‑Assisted Synthesis of Polydopamine‑Passivated Carbon Dots with 5 % Quantum Yield and 35 % Photothermal Efficiency for Cancer Therapy



