Enhancing Supercapacitor Energy Density with Anionic Surfactant‑Intercalated Thermally Reduced Graphene Oxide in Ionic Liquid Electrolytes
Abstract
Thermally reduced graphene oxide (TRG) composites were engineered with controllable interlayer spacing by intercalating the anionic surfactant sodium dodecyl sulfate (SDS). A simple post‑processing step involving rinsing the SDS‑intercalated TRG (TRGS) with the ionic liquid (IL) 1‑ethyl‑3‑methylimidazolium bis(trifluoromethylsulfonyl)imide (EMI‑TFSI) further expanded the interlayer distance through Coulombic interactions. Electrochemical testing in an EMI‑TFSI electrolyte showed that the specific energy density of the resulting TRGSE electrodes rose from 34.9 to 61.8 Wh kg⁻¹ at 1 A g⁻¹, confirming that a larger interlayer spacing increases the accessible surface area for IL ions.
Background
Supercapacitors, or electrical double‑layer capacitors (EDLCs), are prized for their high power density, long cycle life, wide temperature range, and minimal maintenance, making them attractive for electric vehicles and portable electronics [1, 2]. Energy storage in EDLCs relies on ion adsorption at the electrode/electrolyte interface, so maximizing the specific surface area of the electrode and optimizing the electrode/electrolyte interface are critical design goals [3–6].
Ionic liquids (ILs) provide wide electrochemical windows (>3 V) and excellent thermal stability, enabling higher cell voltages and energy densities compared to aqueous electrolytes [7–10]. However, the typically large ionic diameters in ILs can limit access to the micropores of high‑surface‑area carbons. Reduced graphene oxide (RGO) offers a high theoretical surface area (2630 m² g⁻¹) and excellent conductivity, but the absence of oxygen functionalities after reduction renders it hydrophobic and prone to restacking [11–14].
Surfactants such as SDS can intercalate between graphene sheets via electrostatic attraction between the anionic headgroup and residual oxygen species, while the hydrophobic tails anchor to the basal planes, thereby preventing restacking and enabling dispersion in aqueous media [17, 18]. Prior work has shown that surfactant‑intercalated RGO exhibits higher capacitance in aqueous media than in ILs, likely because the IL ion size (≈0.7 nm for EMI‑TFSI) exceeds the interlayer spacing (≈0.4 nm) in surfactant‑only systems [19].
In this study, we introduced a two‑step intercalation strategy: (1) SDS intercalation to yield TRGS; (2) rinsing TRGS with EMI‑TFSI to form TRGSE, where Coulombic forces between SDS and IL molecules further swell the interlayer spacing.
Results and Discussion
Morphology
Scanning electron microscopy (SEM) revealed that pristine TRG tends to agglomerate into graphite‑like particles, whereas TRGS and TRGSE display a more exfoliated, crumpled morphology with wrinkled edges, indicating successful SDS intercalation and subsequent IL‑induced spacing (Figure 2).

a TRG, b TRGS, and c TRGSE
Thermal Stability
Thermogravimetric analysis (TGA) showed a minor weight loss near 100 °C for all samples due to moisture. TRG exhibited a gradual 8 % loss up to 500 °C before rapid decomposition, while TRGS and TRGSE displayed a steeper 24–28 % loss around 200 °C, reflecting the decomposition of SDS and IL components (Figure 3).
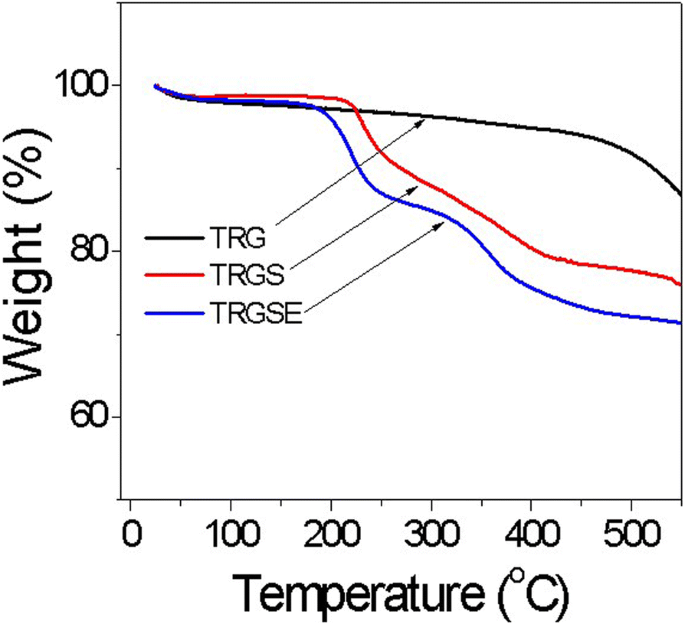
The thermal gravimetric analysis for TRG, TRGS, and TRGSE
Spectroscopic Characterization
Fourier‑transform infrared (FTIR) spectra confirmed the interaction between SDS and the IL. The SO₂ symmetric vibration shifted from 1084 to 1080 cm⁻¹ in TRGS, and further to 1195/1226 cm⁻¹ in TRGSE, indicating strong ionic exchange (Figure 4).
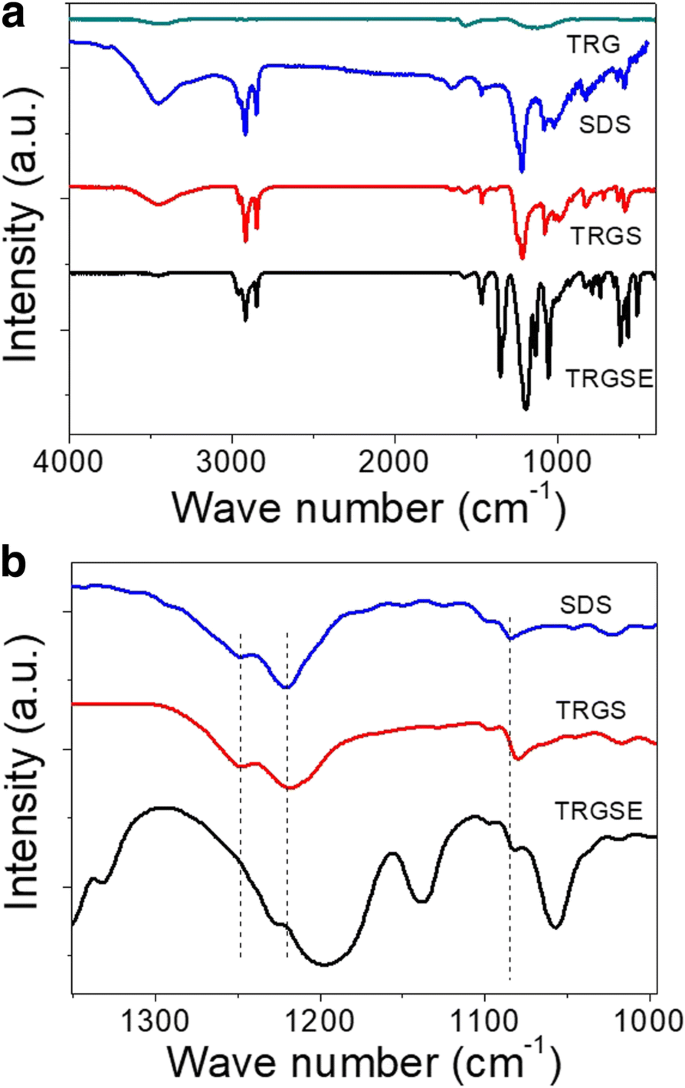
a FTIR spectra of TRG, SDS, TRGS, and TRGSE. (b) Zoom of a specific wavenumber region
Interlayer Spacing
Powder X‑ray diffraction (XRD) revealed a (001) peak at 24.6° for TRG (d = 0.36 nm), 13.3° for TRGS (d = 0.66 nm), and a broader peak for TRGSE, suggesting further expansion. Small‑angle X‑ray scattering (SAXS) confirmed a 3.92 nm interlayer spacing in TRGSE, consistent with the formation of SDS/IL aggregates (Figure 5).
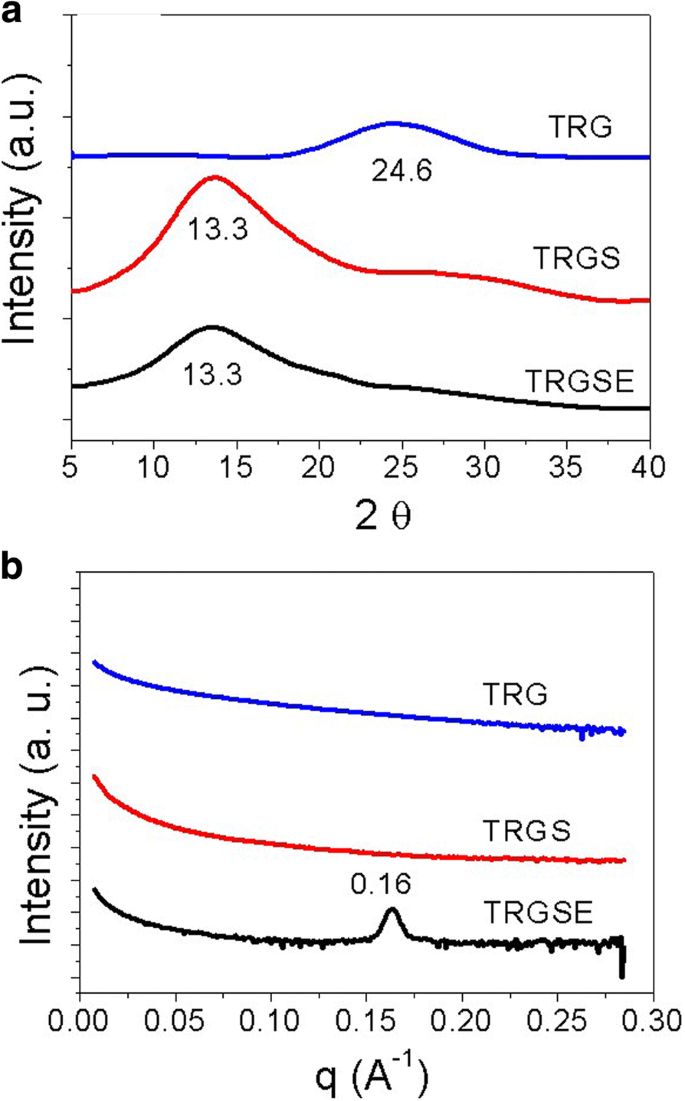
a XRD patterns. b SAXS profiles for TRG, TRGS, and TRGSE
Electrochemical Performance
Electrochemical impedance spectroscopy (EIS) showed that TRGS and TRGSE possess lower contact resistances (Rc ≈ 200–270 Ω cm⁻²) than pristine TRG (Rc ≈ 388 Ω cm⁻²). The larger interlayer spacing in TRGSE reduced the diffusion resistance (Rp = 11.2 Ω cm⁻²) compared to TRGS (21.3 Ω cm⁻²), albeit at the cost of slightly higher overall internal resistance (R ≈ 285 Ω cm⁻²) (Figure 6).
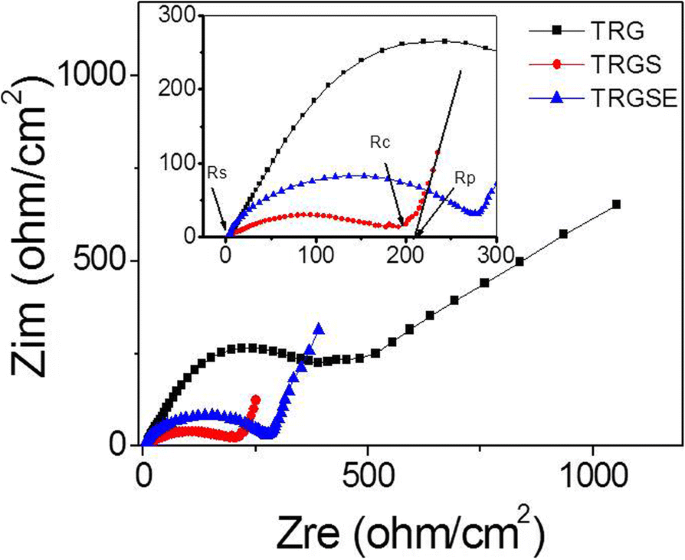
The impedance spectra of TRG, TRGS, and TRGSE EDLC cells
Cyclic voltammetry (CV) demonstrated near‑rectangular curves for TRGS and TRGSE, indicating ideal capacitive behavior, whereas TRG deviated markedly (Figure 7). Specific capacitance values at 1 A g⁻¹ were 43.1, 112.6, and 200.5 F g⁻¹ for TRG, TRGS, and TRGSE, respectively—TRGSE’s capacitance was 1.78 × higher than TRGS (Figure 8).
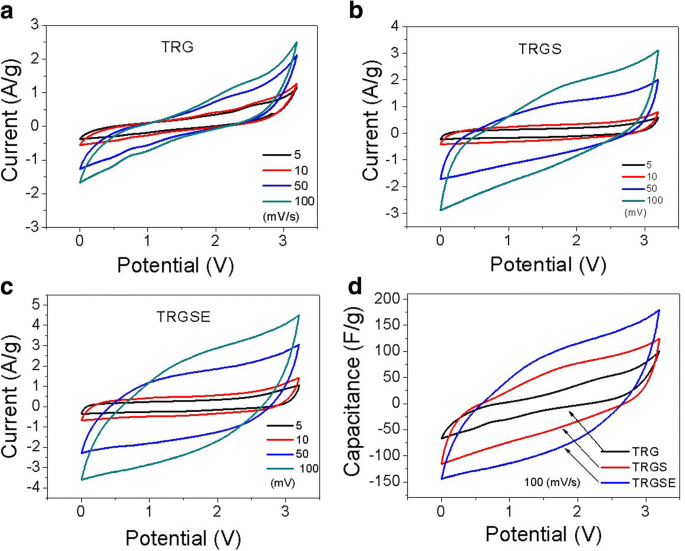
Cyclic voltammetry curves of a TRG, b TRGS, and c TRGSE at various scan rates. d Specific capacitance versus scan rate
Galvanostatic discharge curves confirmed that TRGSE maintained the highest energy density—61.8 Wh kg⁻¹ at 1 A g⁻¹—compared to TRGS (34.9 Wh kg⁻¹) and TRG (15.3 Wh kg⁻¹). At higher current densities, TRGSE retained 4.1 Wh kg⁻¹ at 18 A g⁻¹, while TRG fell below 0.5 Wh kg⁻¹ (Figure 9). Ragone plots illustrated the superior trade‑off between energy and power for TRGSE (Figure 10).
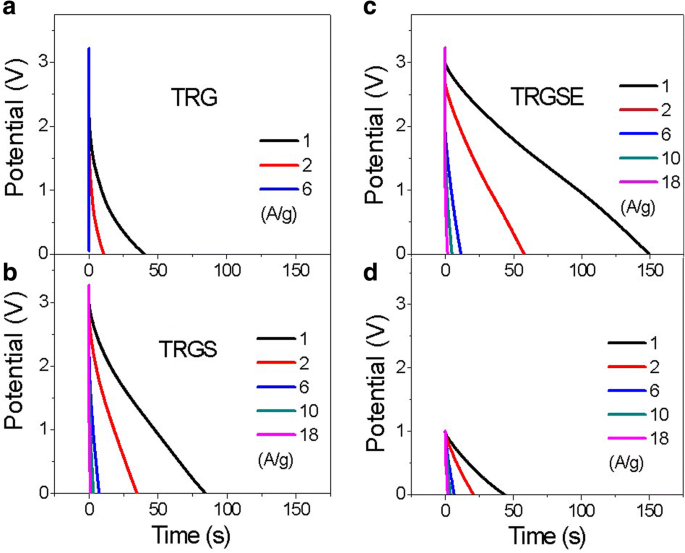
Galvanostatic discharge for a TRG, b TRGS, c TRGSE with IL, and d TRGSE with 2 M H₂SO₄
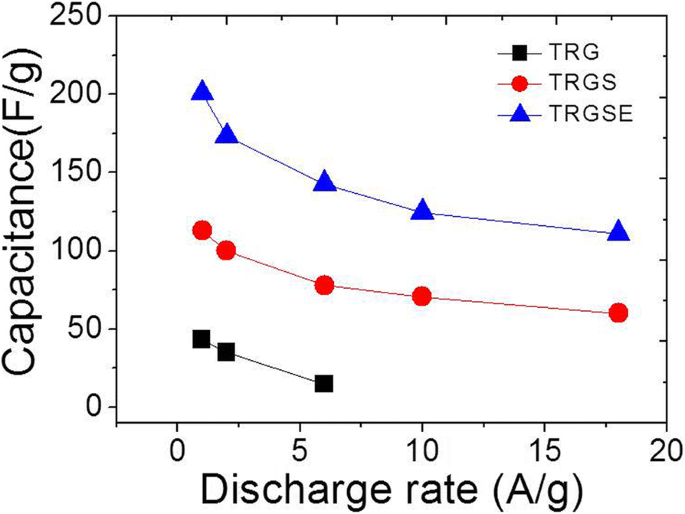
Specific capacitance versus discharge rate
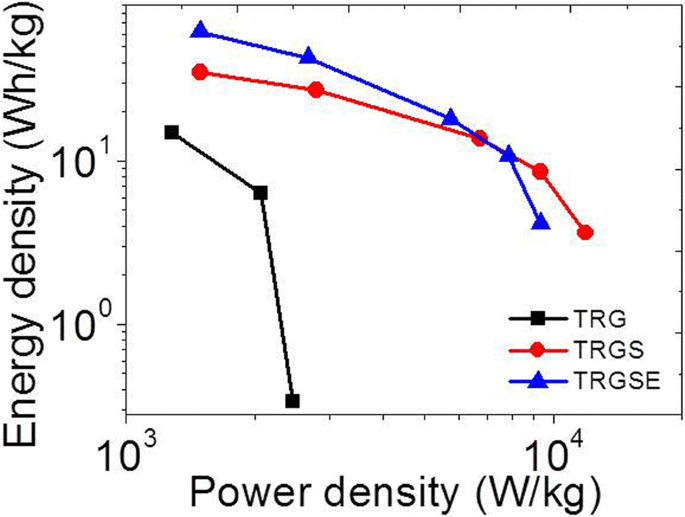
The Ragone plot of TRG, TRGS, and TRGSE
Conclusion
By intercalating SDS into thermally reduced graphene oxide and subsequently introducing EMI‑TFSI, we achieved a controllable interlayer expansion from 0.66 to 3.92 nm. This structural tuning substantially increased the accessible surface area for the IL electrolyte, leading to a 1.78‑fold rise in specific capacitance and a 1.77‑fold increase in energy density at 1 A g⁻¹. The approach demonstrates a viable pathway to high‑performance EDLCs that combine the structural advantages of graphene with the electrochemical benefits of ionic liquids.
Methods
TRG was synthesized from natural graphite via a modified Hummers method followed by thermal reduction. For TRGS, 10 mg of TRG was dispersed in 30 mL of 0.1 M SDS and ultrasonicated for 12 h, then vacuum‑filtered onto a Celgard 3500 separator. The resulting film was rinsed with 15 mL of 0.2 M EMI‑TFSI in ethanol during filtration to obtain TRGSE. Control TRG electrodes were prepared by dispersing 10 mg TRG in 20 mL of 20 wt % ethanol and filtering identically. Electrodes (1 cm²) were pressed onto 304 Stainless Steel current collectors. Two‑electrode EDLC cells were assembled in sealed bags filled with EMI‑TFSI electrolyte and tested on a VersaSTAT 4 potentiostat. Morphology was examined by JEOL‑6700 SEM, structural analysis by Bruker‑AXS D8 XRD and Nanostar U SAXS, thermal stability by TA Q50 TGA (10 °C min⁻¹, N₂), and vibrational spectra by Bruker Vertex 70v FTIR (4000–400 cm⁻¹).
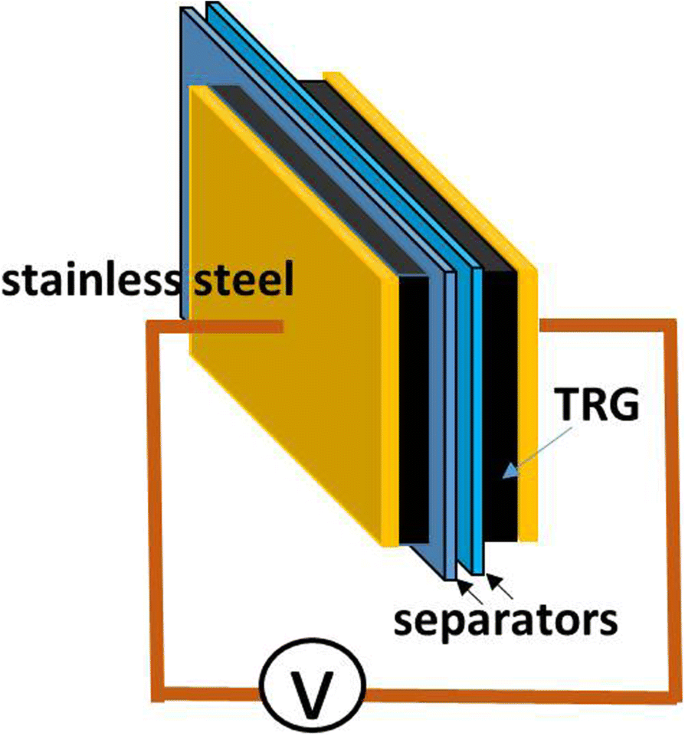
The schematic structure of the EDLC cell
Abbreviations
- EDLC
Electrical double‑layer capacitor
- EMI‑TFSI
1‑Ethyl‑3‑methylimidazolium bis(trifluoromethylsulfonyl)imide
- FTIR
Fourier‑transform infrared spectroscopy
- GO
Graphene oxide
- RGO
Reduced graphene oxide
- SAXS
Small‑angle X‑ray scattering
- SDS
Sodium dodecyl sulfate
- SEM
Scanning electron microscope
- TGA
Thermal gravimetric analysis
- TRG
Thermally reduced graphene oxide
- TRGS
SDS‑intercalated TRG composite
- TRGSE
TRGS rinsed with EMI‑TFSI during filtration
- XRD
X‑ray diffraction
Nanomaterials
- Scalable Ball‑Milling Production of Nanoscale CL‑20/Graphene Oxide Composites with Reduced Particle Size and Sensitivity
- Millstone Exfoliation: High-Yield, Large-Size Few-Layer Graphene Oxide via True Shear Force
- Synergistic Flame Retardancy of Fullerene‑Anchored Reduced Graphene Oxide Hybrids in Epoxy Resin
- Magnetic MnFe₂O₄/Reduced Graphene Oxide Nanocomposite: Efficient and Recyclable Adsorption of Tetracycline from Water
- Quaternized PVA/Graphene Oxide Composite Membrane Enhances Ethanol Barrier and Ionic Conductivity for Passive Alkaline DEFCs
- Dynamic Hierarchical Self‑Assembled Hexabenzocoronene for High‑Performance Lithium‑Ion Battery Anodes
- Ultra‑Sensitive, Rapid‑Response Ammonia Gas Sensor Based on Reduced Holey Graphene Oxide Thin Films
- High‑Performance Flexible Microsupercapacitors Based on 3‑D rGO/PEDOT Open‑Network Architectures
- Sulfur‑Doped Alkylated Graphene Oxide: A Superior Green Anti‑Wear Lubricant Additive
- Efficient Photocatalytic Wastewater Treatment Using Silver-Decorated Reduced Graphene Oxide Nanoflakes



