Low‑Temperature UV‑Cured SnO₂ Electron Transport Layers Deliver 14.4% Efficiency in Planar Perovskite Solar Cells
Abstract
We introduce a facile ultraviolet (UV) ozone (UVO) treatment protocol for tin oxide (SnO2) electron transport layers (ETLs) fabricated at temperatures below 90 °C. By optimizing the SnO2 precursor concentration, we control film thickness, achieve high crystallinity, and remove organic residues, resulting in ETLs with superior conductivity and optical transmittance. When integrated into a planar perovskite solar cell (PSC) architecture (Glass/ITO/SnO2/MAPbI3/Spiro‑OMeTAD/Au), the devices attain a maximum power‑conversion efficiency (PCE) of 14.36 % at a 20 % SnO2 concentration. This low‑temperature, UV‑based approach offers a scalable, cost‑effective route to high‑performance PSCs.
Background
Perovskite solar cells (PSCs) have rapidly progressed from 3.8 % to 22.1 % PCE in recent years [1,2,3,4,5,6,7,8]. In both mesoporous and planar architectures, the perovskite absorber is sandwiched between electron transport layers (ETLs) and hole transport layers (HTLs), and the interfacial quality of these layers directly influences device performance [9,10,11]. Conventional fabrication of compact ETLs often requires high‑temperature annealing or sintering (up to 450 °C for TiO2 and 180 °C for SnO2) [24–31], which limits large‑scale, low‑cost production.
We present a spin‑coating protocol for SnO2 precursors followed by UVO treatment, achieving a fully compact ETL at temperatures below 90 °C. This method eliminates the need for high‑temperature steps, reduces processing complexity, and lowers manufacturing costs. Our PSCs, based on CH3NH3PbI3 (MAPbI3) and fabricated via a one‑step anti‑solvent (OSAS) approach [33–37], exhibit a planar heterojunction architecture: Glass/ITO/SnO2/MAPbI3/Spiro‑OMeTAD/Au. The optimized devices achieve a PCE of 14.5 %.
Methods
Materials and Precursor Preparation
Methylammonium iodide (MAI; Z99.5 %) and lead iodide (PbI2; Z99.9 %) were sourced from Xi’an Polymer Light Technology Corp. Tin oxide (SnO2; 15 % mass in H2O colloidal dispersion with trace organic solvents) was purchased from Alfa Aesar. 1,2‑Dichlorobenzene (DCB; 99.5 %) and other solvents (DMF, DMSO, Spiro‑OMeTAD, TBP, Li‑TFSI) were obtained from Sigma Aldrich. All reagents were used as received without further purification.
Fabrication of Devices
The device stack is ITO/SnO2/MAPbI3/Spiro‑OMeTAD/Au. ITO glass substrates (<15 Ω/□) were ultrasonically cleaned with acetone, ethanol, and DI water (15 min each) and dried with nitrogen. Subsequent UVO cleaning (15 min at ~60 °C) removed residual organics. SnO2 films were spin‑coated from precursor solutions (10 %, 15 %, 20 %, 30 % by volume) at 5000 rpm for 30 s, dried at 50 °C for 5 min, and then subjected to UVO (60 min, ~60 °C). A 1‑M MAPbI3 precursor (MAI:PbI2 = 1:1 M) was prepared in a 9:1 (v/v) DMF:DMSO mixture, stirred and heated at 50 °C overnight. The perovskite layer was spin‑coated at 4000 rpm for 30 s, with a drop of diethyl ether added 5 s before the end of spin. The film was annealed at 90 °C for 10 min in a glove‑box. The HTL consisted of 70 mM Spiro‑OMeTAD, 28.8 mM Li‑TFSI, and 55 mM TBP in DCB, spin‑coated at 5000 rpm for 20 s. Finally, 100 nm Au was thermally evaporated at ~5 Å/s under 5×10−4 Pa. The active area was 4 mm2.
Characterization and Measurements
Current–voltage (J‑V) curves were recorded with a Keithley 2400 under AM1.5G illumination (100 mW/cm2) using a Newport 94043A solar simulator, calibrated against a reference silicon cell. UV–vis transmission spectra were obtained with a Cary 5000 spectrometer. Surface morphology and cross‑sectional structure were examined by SEM (JSM‑7001F). X‑ray diffraction (XRD) was performed on an X‑ray diffractometer (DX‑2700) to confirm the rutile SnO2 phase. Energy‑dispersive X‑ray spectroscopy (EDS) assessed elemental composition.
Results and Discussion
UV/ozone emits photons at 185 and 254 nm (647 and 472 kJ/mol), sufficient to cleave C‑C, C‑O, and C‑H bonds (346–411 kJ/mol) [38–40]. EDS of a 20 % SnO2 film post‑UVO shows >99 % Sn and O, with <1 % residual C, confirming removal of organics and high‑purity ETLs (Table 1).
XRD of the UVO‑treated films reveals characteristic rutile peaks at 2θ = 26.5°, 34.0°, 38.1°, 51.6°, and 65.9° (planes (110), (101), (200), (211), (301)) matching JCPDS 41‑1445. Debye–Scherrer analysis yields an average crystallite size of 5.5 nm (Fig. 2).
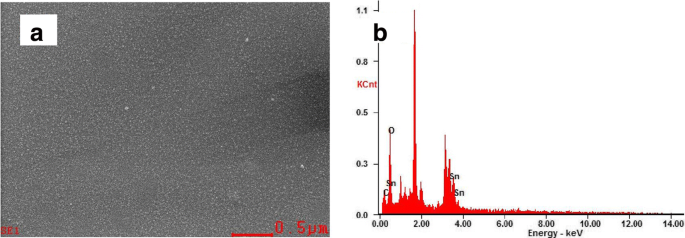
Surface SEM image of SnO2 (a) and the corresponding EDX spectra of ITO/SnO2 film
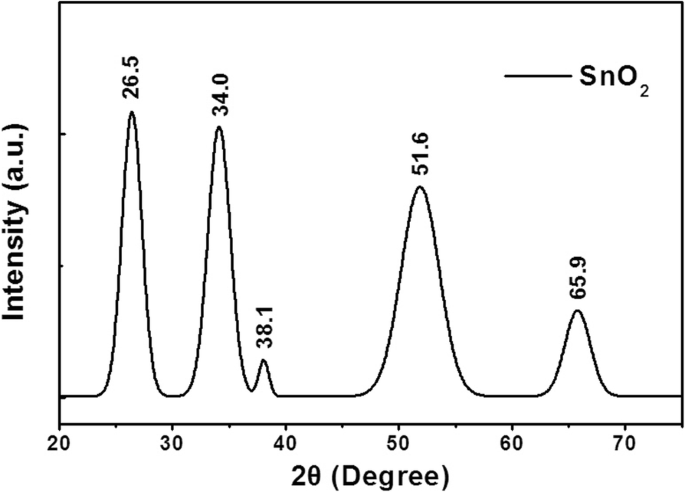
The X-ray diffraction (XRD) pattern of SnO2 after UV treatment
Figure 3a shows the device schematic. SEM of the active layer (Fig. 3b) reveals a continuous MAPbI3 film (~384 nm thick) with single crystals >1 µm, indicative of high‑quality perovskite formation. The SnO2 (20 %) layer is ~65 nm thick, providing an optimal balance between conductivity and light transmission.
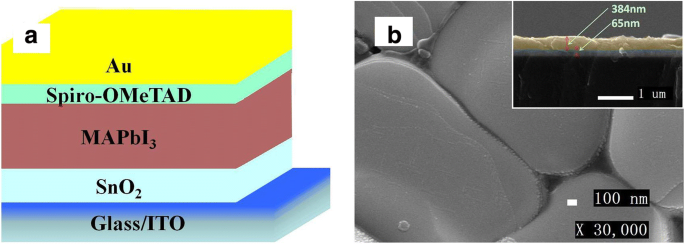
Structure diagram of the perovskite solar cell (a) and the SEM image of active layer (b)
J‑V characteristics (Fig. 4) and detailed parameters (Table 2) illustrate that Jsc peaks at 20.11 mA/cm2 for the 20 % SnO2 device, while Voc and fill factor (FF) also improve with increasing concentration up to 20 %. The corresponding PCE reaches 14.36 % with series resistance (Rs) of 232.8 Ω and shunt resistance (Rsh) of 15 868 Ω. Excessive SnO2 (30 %) reduces transparency, increasing recombination and lowering performance.
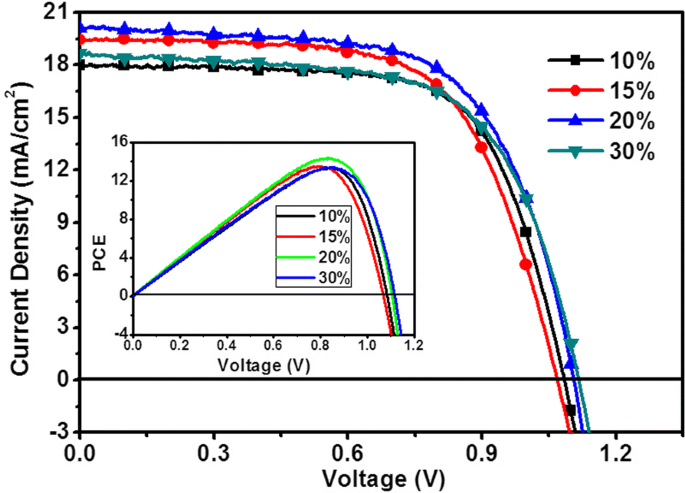
J‑V characteristics of the device. The inset shows the corresponding PCE‑Voc curve.
Cross‑sectional SEM (Fig. 5) confirms film thicknesses of 34 nm (10 %), 48 nm (15 %), 66 nm (20 %), and 97 nm (30 %). I-V measurements of ITO/SnO2/Au structures reveal that vertical resistance decreases from 98.6 Ω (10 %) to 33.7 Ω (20 %) before increasing to 50.8 Ω (30 %), correlating with film continuity and crystallinity.

Cross‑sectional SEM images of a ITO/SnO2 (10 %), b ITO/SnO2 (15 %), c ITO/SnO2 (20 %), and d ITO/SnO2 (30 %)
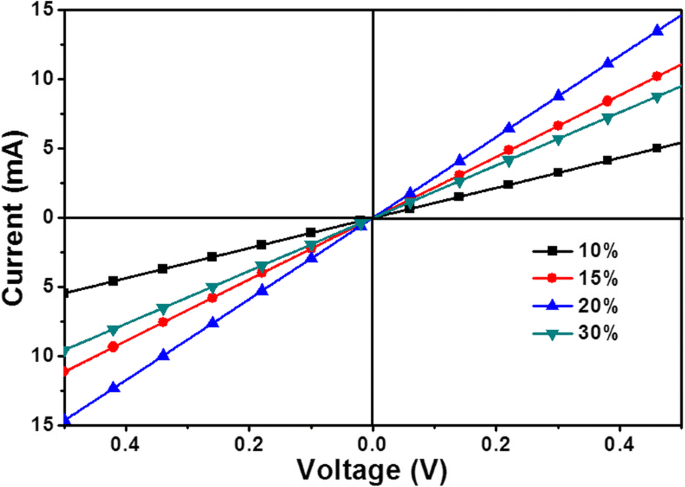
I‑V curves of ITO/SnO2(x)/Au, x = 10, 15, 20, and 30 %
Top‑view SEM (Fig. 7) shows uniform, smooth films across all concentrations, with crystallite sizes around 6.8 nm—consistent with XRD estimates. Minor island defects appear at 10 % (Fig. 7a, e), reducing conductivity, while 20 % provides optimal continuity. At 30 % the films exhibit slight coalescence, increasing resistance.

Top view SEM images of a–d the prepared ITO/SnO2(x) films at × 50,000 magnification, and e–h films at × 200,000 magnification
UV‑vis transmission spectra (Fig. 8) indicate >75 % transmittance across 400–800 nm. Peaks shift to longer wavelengths with increasing SnO2 thickness (10 % → 616 nm, 15 % → 662 nm, 20 % → 718 nm, 30 % → >800 nm). The MAPbI3 absorption band (300–760 nm) overlaps best with the 10–20 % SnO2 films, maximizing light harvesting. Excess thickness (30 %) reduces transmittance, leading to lower PCE.
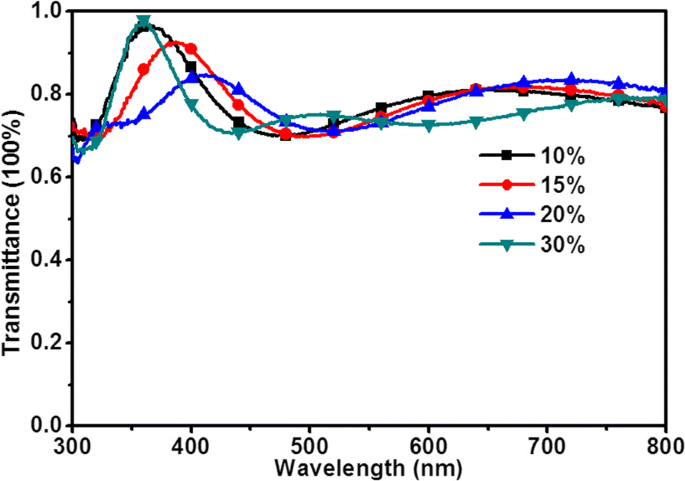
UV–vis transmission spectra of the ITO/SnO2(x) films
Conclusions
We demonstrate that low‑temperature UVO treatment of spin‑coated SnO2 precursors yields compact, high‑purity ETLs with excellent conductivity and transparency. Optimizing the SnO2 concentration to 20 % produces planar PSCs with a PCE of 14.36 %. The performance is governed by the balance between film thickness, uniformity, and optical transparency. This scalable, low‑cost process is well suited for industrial PSC manufacturing.
Abbreviations
- ETLs:
Electron transport layers
- FF:
Fill factor
- HTLs:
Hole transport layers
- Jsc:
Short‑circuit photocurrent
- OSAS:
One‑step anti‑solvent
- PCE:
Power conversion efficiency
- PSCs:
Perovskite solar cells
- UVO:
Ultraviolet ozone
- Voc:
Open‑circuit voltage
Nanomaterials
- Nano‑Heterojunctions: Boosting Solar Cell Efficiency with Colloidal Quantum Dots
- Electrodeposited SnO₂ Thin Films on FTO: A Scalable Electron Transport Layer for High‑Efficiency Planar Perovskite Solar Cells
- Superior Performance of Inverted Perovskite Solar Cells Using CdSe Quantum Dot and LiF Electron Transport Layers
- High‑Efficiency Planar Perovskite Solar Cells via Sequential Vapor‑Grown Hybrid Perovskite Layers
- Optimizing Titanium Precursors for High‑Performance Compact TiO₂ Layers in Perovskite Solar Cells
- UV‑Ozone‑Treated Reduced Graphene Oxide Enhances PEDOT:PSS Hole Transport Layer in Inverted Perovskite Solar Cells
- Boosting Planar Sb₂S₃ Solar Cell Efficiency with Cs₂CO₃‑Modified TiO₂ Electron Transport Layer
- In Situ-Formed, Low‑Temperature Deposited Nb‑Doped TiO₂ Compact‑Mesoporous Layer Enhances Hysteresis‑Free, High‑Efficiency Perovskite Solar Cells
- Gradient‑Engineered Light‑Absorbing Layer Boosts Carrier Separation and Efficiency in Perovskite Solar Cells
- Enhancing 2D Perovskite Solar Cell Performance Through Water-Assisted Crystallization



