Boosting Planar Sb₂S₃ Solar Cell Efficiency with Cs₂CO₃‑Modified TiO₂ Electron Transport Layer
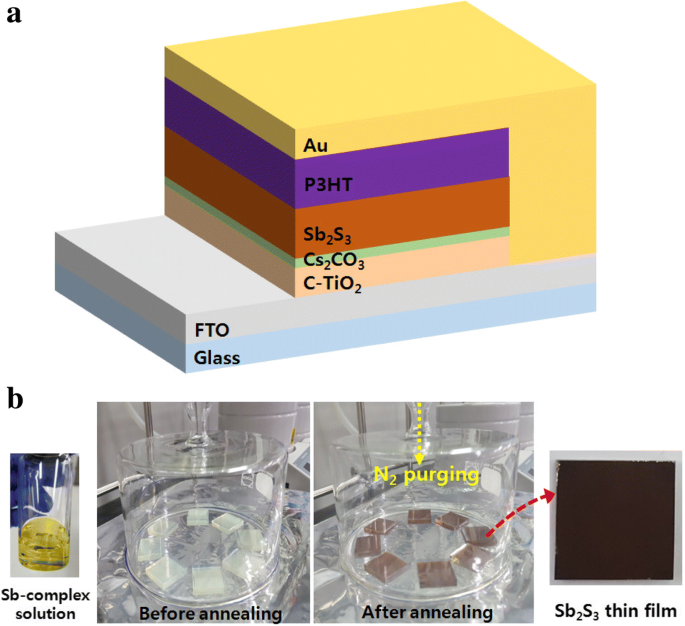
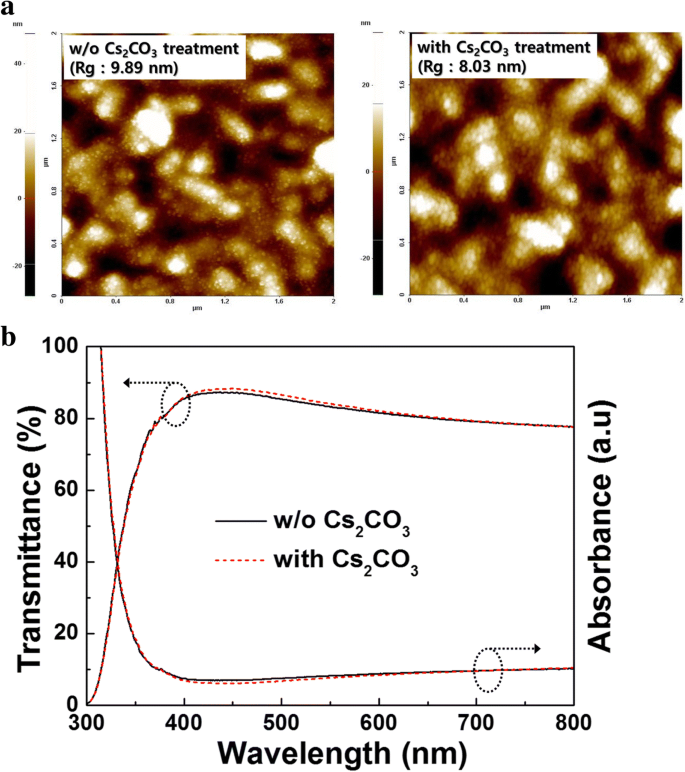
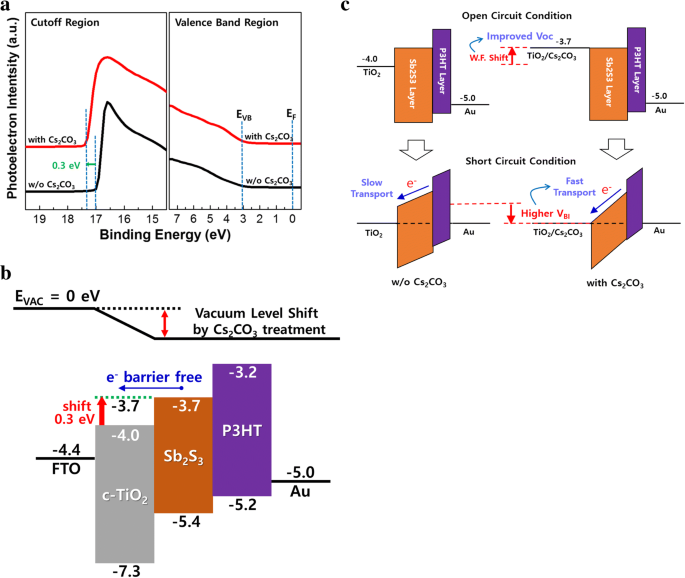
We demonstrate a straightforward, solution‑processed surface modification of compact TiO₂ (c‑TiO₂) with Cs₂CO₃ that raises its work function, reduces surface roughness, and increases the power conversion efficiency (PCE) of planar‑type Sb₂S₃ solar cells from 2.83 % to 3.97 %—a 40 % improvement. This work establishes Cs₂CO₃ as a simple, scalable route to enhance electron transport layers for high‑performance, earth‑abundant absorber materials. In the quest for low‑cost, non‑toxic thin‑film photovoltaics, earth‑abundant chalcogenides such as CuZnSnSe₂, PbS, Cu₂S, SnS, and Sb₂S₃ are being explored as alternatives to CIGS and CdTe. While CuZnSnSe₂ and PbS suffer from toxic precursors or complex stoichiometry control, Sb₂S₃ stands out with a suitable bandgap (~1.65 eV), a high absorption coefficient (>10⁵ cm⁻¹), a high dielectric constant that facilitates exciton dissociation, and excellent stability and low toxicity. Planar Sb₂S₃ devices (FTO/c‑TiO₂/Sb₂S₃/HTL/Au) are attractive for industrial scaling because of their simpler architecture compared to sensitized devices that use mesoporous TiO₂. However, planar cells currently lag in efficiency (2.5–5.8 %) because charge transport within the Sb₂S₃ layer depends on grain size, crystallinity, and interfacial contact. Previous efforts have focused on optimizing Sb₂S₃ film morphology via chemical bath deposition, thermal evaporation, ALD, or nanoparticle inks. The fast chemical approach (FCA) using a butyldithiocarbamic acid precursor has recently achieved large grains in a single spin‑coating step, yielding high‑quality absorbers. In electron transport layers, surface engineering can lower the energy barrier and improve charge extraction. Cs₂CO₃ has proven effective in organic photovoltaics, OLEDs, and perovskite cells by lowering the work function of TiO₂. Yet its application to Sb₂S₃ devices has not been reported. All reagents were used as received from Sigma‑Aldrich. The Sb complex was prepared by reacting Sb₂O₃ with CS₂, n‑butylamine, and ethanol, yielding an antimony butyldithiocarbamate precursor that was diluted for spin‑coating. Planar devices followed the stack FTO/c‑TiO₂/Sb₂S₃/P3HT/Au. c‑TiO₂ was deposited by spin‑coating a TTIP/ethanol solution and annealed at 500 °C. Cs₂CO₃ (1–10 mg mL⁻¹) dissolved in methanol was spin‑coated onto UV‑ozone treated c‑TiO₂, then annealed at 150 °C before Sb₂S₃ deposition. Sb₂S₃ films were spin‑coated from the FCA precursor and annealed at 200 °C/1 min and 350 °C/2 min. P3HT (10 mg mL⁻¹ o‑DCB) was spin‑coated and annealed at 100 °C, followed by thermal Au deposition under 5 × 10⁻⁶ Torr. Active area: 0.16 cm². Morphology: FE‑SEM (Hitachi S‑4800) and AFM (Park NX10). Optical properties: UV‑Vis (Lambda 750). Photovoltaic performance: J–V curves under AM 1.5G (100 mW cm⁻²) using a Keithley 2400 and Newport 91192. EQE: Newport QuantX‑300. XRD: PANalytical Empyrean. XPS and UPS: Thermo Scientific ESCALAB 250Xi with He I (21.2 eV) and Al Kα (1486.6 eV). XPS depth profiling used Ar⁺ clusters at 1 Å s⁻¹. Figure 1a shows the device schematic: a compact TiO₂ layer on FTO acts as the electron transport layer, Sb₂S₃ absorbs light, and P3HT transports holes to the Au electrode. a Device schematic of planar Sb₂S₃ solar cells. b SBA fabrication process via FCA. The FCA precursor yields a highly crystalline Sb₂S₃ film with large grains, as confirmed by SEM (Fig. 2). The grains are comparable to those reported by Wang et al., indicating high film quality. a Top view and b cross‑section SEM of Sb₂S₃ after annealing at 350 °C. Optimizing Cs₂CO₃ concentration is critical: 1 mg mL⁻¹ leads to incomplete coverage, while 5–10 mg mL⁻¹ behaves as a dielectric, raising series resistance. The optimum, 3 mg mL⁻¹, yields the best performance (Fig. 3a, Table 1). a J–V curves and b EQE spectra for devices with/without Cs₂CO₃ treatment. Without treatment, the cell shows V_OC = 0.549 V, J_SC = 10.71 mA cm⁻², FF = 48.14 % (PCE = 2.83 %). After 3 mg mL⁻¹ Cs₂CO₃, V_OC rises to 0.596 V, J_SC to 11.71 mA cm⁻², FF to 56.89 %, boosting PCE to 3.97 % (≈ 40 % gain). EQE improvements across the full spectrum confirm more efficient light‑to‑current conversion. XRD shows no new phases after Cs₂CO₃ treatment, indicating that the c‑TiO₂ crystallinity remains unchanged (Fig. 4). XPS depth profiling reveals a Cs₂CO₃ layer of 2–3 nm (Fig. 5d). AFM demonstrates a reduction in root‑mean‑square roughness from 9.89 nm to 8.03 nm, which correlates with a decrease in series resistance (11.14 Ω cm² → 8.82 Ω cm²) and a higher FF (48.14 % → 56.89 %) (Fig. 6a). XRD patterns of c‑TiO₂ with/without Cs₂CO₃ treatment. a XPS survey and Cs 3d peak. b Ti 2p peak. c O 1s peak. d Cs 3d depth profile showing 2–3 nm thickness. a AFM images (2 µm × 2 µm). b UV‑Vis absorption and transmission of c‑TiO₂ with/without Cs₂CO₃. UPS measurements reveal a 0.3 eV drop in the work function of c‑TiO₂ after Cs₂CO₃ treatment (Fig. 7a). This shift aligns the conduction band of c‑TiO₂ closer to that of Sb₂S₃, enhancing the built‑in potential (V_BI) and reducing the electron extraction barrier. Consequently, both V_OC and J_SC increase. The energy band diagram (Fig. 7b) and proposed operating principle (Fig. 7c) illustrate how Cs₂CO₃‑induced band bending improves charge collection. a UPS spectra of c‑TiO₂. b Energy level diagram. c Device operation with/without Cs₂CO₃. Cs₂CO₃ surface modification of c‑TiO₂ effectively lowers its work function, smooths the surface, and improves physical contact with Sb₂S₃. These changes reduce series resistance, increase V_OC, J_SC, and FF, culminating in a 40 % PCE enhancement (2.83 % → 3.97 %). This study demonstrates that inorganic surface modifiers like Cs₂CO₃ can play a pivotal role in advancing planar‑type Sb₂S₃ solar cells toward higher efficiencies and scalable production. Atomic force microscopy Compact TiO₂ External quantum efficiency Electron transport layers Fast chemical approach Fill factor Fluorine‑doped tin oxide Hole transport layers Short‑circuit current density Current density–voltage Mesoporous TiO₂ Poly(3‑hexylthiophene) Power conversion efficiency Series resistance Shunt resistance Scanning electron microscopy Ultraviolet photoelectron spectroscopy Ultraviolet–visible spectrometer Built‑in potential Open‑circuit voltage X‑ray photoelectron spectroscopy X‑ray diffractionAbstract
Background
Methods/Experimental
Materials and Sb Complex Synthesis
Device Fabrication
Characterization
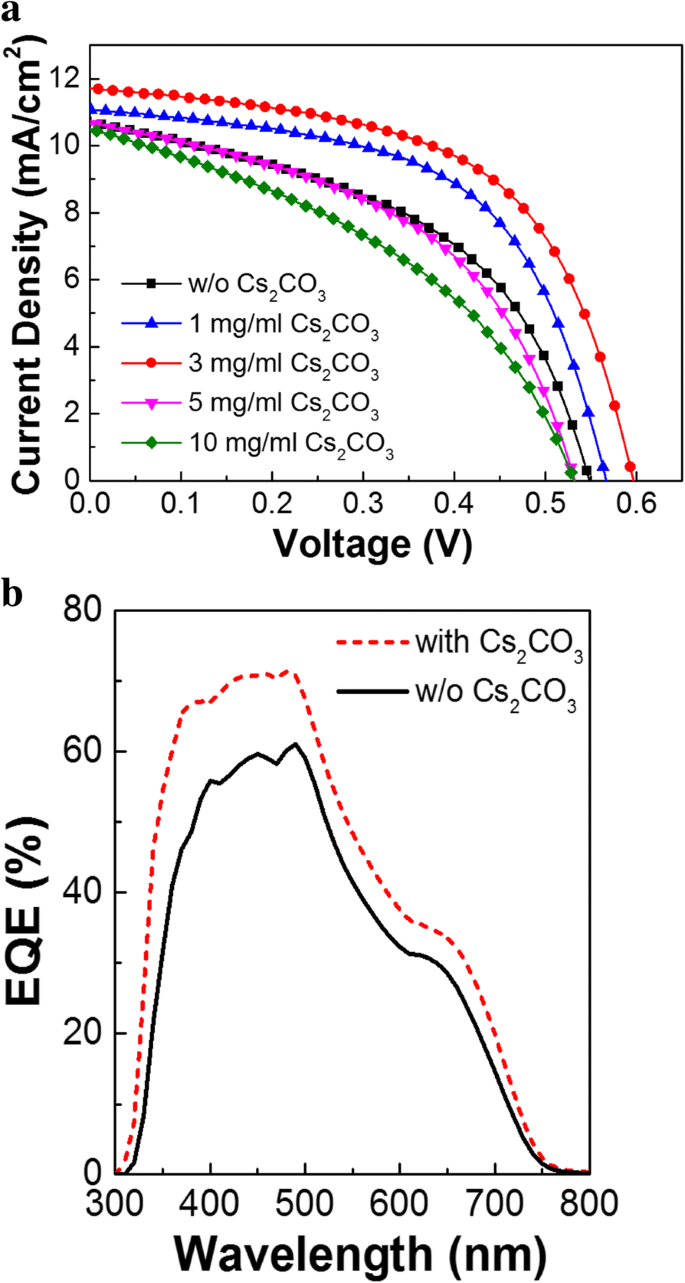
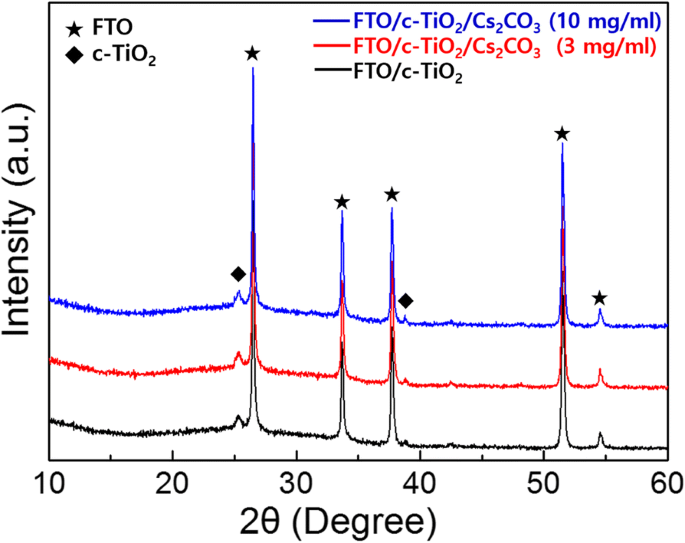
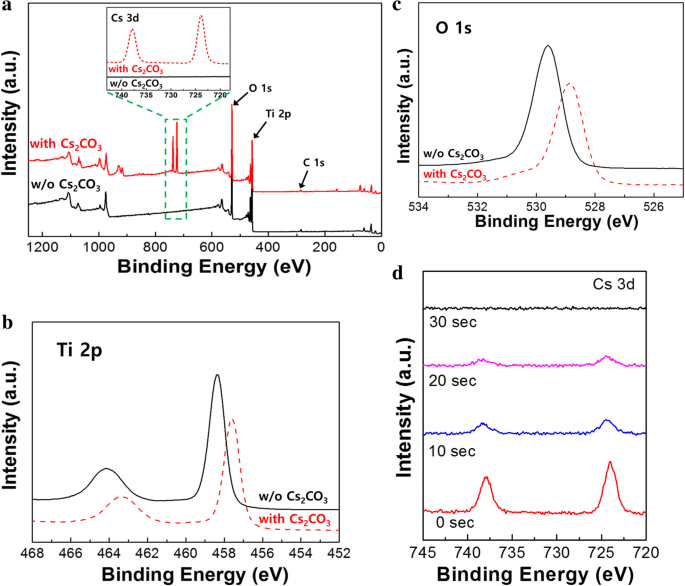
Results and Discussion

Conclusions
Abbreviations
Nanomaterials
- Nano‑Tree ZnO Nanowires Boost Dye‑Sensitized Solar Cell Efficiency
- Nano‑Heterojunctions: Boosting Solar Cell Efficiency with Colloidal Quantum Dots
- Optimizing Titanium Precursors for High‑Performance Compact TiO₂ Layers in Perovskite Solar Cells
- Low‑Temperature UV‑Cured SnO₂ Electron Transport Layers Deliver 14.4% Efficiency in Planar Perovskite Solar Cells
- Designing Plasmonic Nanoparticle Strategies for Enhanced Organic Solar Cell Performance
- Efficient Ambient‑Air Fabrication of Mesoporous Perovskite Solar Cells Using N‑Butyl‑Amine‑Enhanced PbI₂ Precursors
- High‑Performance Dye‑Sensitized Solar Cells Using Screen‑Printed Multi‑Walled Carbon Nanotube Counter Electrodes
- Thickness‑Dependent Photocurrent and Optoelectronic Dynamics in TiO₂/Sb₂S₃/P3HT Planar Hybrid Solar Cells
- In Situ-Formed, Low‑Temperature Deposited Nb‑Doped TiO₂ Compact‑Mesoporous Layer Enhances Hysteresis‑Free, High‑Efficiency Perovskite Solar Cells
- High-Performance MoIn₂S₄@CNT Counter Electrodes for Dye‑Sensitized Solar Cells



