Enhanced Photoelectrochemical Water Oxidation Using CdS-Modified α‑Fe₂O₃/TiO₂ Nanorod Array Photoanodes
Abstract
We report a streamlined successive ionic layer adsorption and reaction (SILAR) combined with a hydrothermal route to fabricate CdS nanoparticle‑modified α‑Fe₂O₃/TiO₂ nanorod arrays for photoelectrochemical (PEC) water oxidation. The integration of a CdS/α‑Fe₂O₃/TiO₂ ternary system significantly broadens visible‑light absorption and facilitates charge‑carrier separation, resulting in a pronounced enhancement of PEC performance. The engineered Fe₂O₃/TiO₂ interface exhibits an atypical type‑II band alignment, where the Fe₂O₃ conduction band lies above that of TiO₂. This configuration promotes rapid recombination of electrons from Fe₂O₃ with holes from TiO₂, effectively isolating electrons in Fe₂O₃ and holes in TiO₂ and improving charge‑carrier utilization. As a photoanode, the CdS/α‑Fe₂O₃/TiO₂ heterostructure delivers a photocurrent density of 0.62 mA cm⁻² at 1.23 V vs. RHE in alkaline medium, with an onset potential shifted 80 mV negatively. These findings provide a viable strategy for boosting TiO₂‑based photoanode performance in solar‑driven water splitting.
Background
Photoelectrochemical (PEC) water splitting is widely regarded as a leading technology for clean hydrogen production, addressing both pollution and finite fossil resources. Since the pioneering TiO₂‑based PEC water oxidation study [1], TiO₂ has attracted sustained interest due to its chemical stability, favorable band alignment, and robust optical response [2, 3]. Nonetheless, pristine TiO₂ suffers from sluggish water‑oxidation kinetics, largely because of limited light absorption and poor carrier‑separation efficiency [4, 5].
Numerous strategies—including surface functionalization [6], quantum‑dot sensitization, and heterojunction engineering [7, 8]—have been explored to overcome these limitations. Heterostructure formation, in particular, can facilitate efficient electron–hole separation. For example, TiO₂ coupled with Co₃O₄ or ZnIn₂S₄ (see [9], [10, 11]) demonstrates marked performance gains. Hematite (α‑Fe₂O₃) is a promising partner for TiO₂ because of its 2.0 eV band gap, excellent stability, and low cost [12]. Theoretical calculations suggest α‑Fe₂O₃ could reach 15.3% PCE, with a photocurrent density of 12.6 mA cm⁻² at 1.23 V vs. RHE under AM 1.5 G [13]. Thus, α‑Fe₂O₃/TiO₂ heterostructures can both extend light absorption and enhance carrier separation.
However, α‑Fe₂O₃ photoanodes face short electron–hole lifetimes and limited hole diffusion lengths (2–4 nm), leading to high recombination [12]. Coupling α‑Fe₂O₃ with narrow‑band semiconductors such as CdS [14, 15] or PbS [16] can further improve charge dynamics. CdS/Fe₂O₃/TiO₂ has emerged as a compelling architecture, combining matched band gaps with extended spectral response and efficient interfacial charge transfer. Moreover, one‑dimensional (1D) nanorod (NR) arrays outperform planar films due to enhanced light scattering and direct charge transport pathways [17–18]. Adding branched nanostructures further increases surface area, providing more active sites for water oxidation [19]. Consequently, a CdS‑modified Fe₂O₃/TiO₂ NR array presents an attractive design for high‑efficiency PEC water oxidation.
In this study, we present a facile SILAR‑hydrothermal synthesis of CdS‑modified Fe₂O₃/TiO₂ NR arrays and evaluate their PEC performance. UV‑vis spectroscopy confirms a broadened visible‑light absorption, while PL and EIS analyses demonstrate improved charge‑transfer dynamics. The resulting heterostructure achieves a photocurrent density of 0.62 mA cm⁻² at 1.23 V vs. RHE, nearly double that of bare TiO₂ (0.32 mA cm⁻²). These results highlight the potential of CdS/Fe₂O₃/TiO₂ NR arrays for advanced photovoltaic and photoelectrochemical devices.
Methods
Preparation of CdS/Fe₂O₃/TiO₂ NR Heterostructured Photoanode
Synthesis of TiO₂ NR Array
FTO glass (cut into rectangles) was sequentially sonicated in deionized water, acetone, and ethanol. The cleaned FTO was placed in an autoclave containing 20 mL deionized water, 20 mL 37 % HCl, and 1.1 mL titanium isopropoxide, then heated at 160 °C for 6 h. After washing with water and ethanol, the substrate was annealed at 450 °C for 30 min in air.
Synthesis of Fe₂O₃/TiO₂ NR Array
The TiO₂ NR array was immersed in a mixed solution of 15 mL 0.1 M FeCl₃ and 15 mL 0.5 M NaNO₃, then transferred to an autoclave and heated at 100 °C for 2 h. After cooling, the sample was washed and annealed at 450 °C for 1 h.
Synthesis of CdS/Fe₂O₃/TiO₂ NR
The Fe₂O₃/TiO₂ NR array was pretreated with 0.3 M mercaptopropionic acid (MPA) in ethanol at 50 °C overnight, followed by ethanol washing. CdS was deposited via SILAR: the substrate was sequentially immersed for 30 s in 0.1 M Cd(NO₃)₂·4H₂O (ethanol), pure ethanol, 0.2 M Na₂S·9H₂O (methanol), and pure methanol. Five SILAR cycles were performed, then the sample was washed with methanol.
Materials Characterization
XRD (2θ = 20–80°) identified phase composition. FE‑SEM combined with EDS revealed morphology and elemental distribution. TEM (Tecnai 20 U‑Twin) provided lattice imaging. UV‑vis absorption and PL (Hitachi U‑4100) were recorded to assess optical and recombination properties.
Photoelectrochemical Performance Characterization
PEC measurements were conducted in a CHI660E electrochemical workstation using a three‑electrode setup with 1 M NaOH as electrolyte. The system was purged with Ar for 30 min before measurement. Linear sweep voltammograms (LSV) and chronoamperometry were performed under 100 mW cm⁻² AM 1.5 G illumination. Mott–Schottky plots were recorded in the dark at 1 kHz. Potentials were converted to RHE using the Nernst equation: E_{RHE} =
Result and Discussion
Structure and Morphology Characterization
XRD patterns (Fig. 1a) confirm the formation of rutile TiO₂, α‑Fe₂O₃, and CdS phases. The TiO₂ peaks at 36.0°, 44.1°, 54.3°, 62.7°, 64.0°, 65.4°, and 69.8° match the (101), (210), (211), (002), (310), (221), and (112) planes of rutile TiO₂ (JCPDS 21‑1276). Fe₂O₃ introduces peaks at 32.9° and 45.2° corresponding to (222) and (332) planes (JCPDS 39‑0238). CdS deposition yields peaks at 26.4° and 28.2° for (002) and (101) planes (JCPDS 65‑3414). SEM images (Fig. 1b) reveal uniform TiO₂ NRs (~50 nm diameter) that become 60 nm after Fe₂O₃ growth and further enlarge with CdS decoration. EDS mapping (Additional files 1–2) shows uniform distribution of Ti, Fe, Cd, and S.
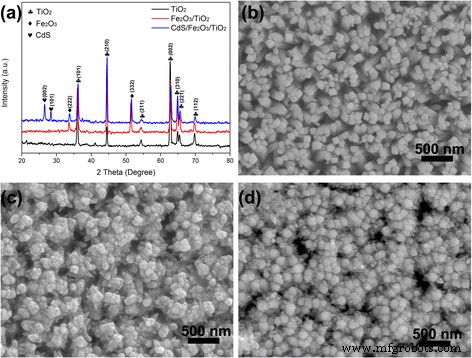
a XRD patterns and b SEM images of TiO₂ NR, Fe₂O₃/TiO₂ NR, and CdS/Fe₂O₃/TiO₂ NR.
HRTEM and SAED (Fig. 2) confirm the crystalline nature of TiO₂, Fe₂O₃, and CdS. Lattice spacings of 0.31, 0.27, and 0.21 nm correspond to CdS (101), Fe₂O₃ (222), and TiO₂ (210) planes, respectively.

a HRTEM of CdS/Fe₂O₃/TiO₂ NR; b SAED showing characteristic diffraction rings.
XPS analysis (Fig. 3) verifies the presence of Ti, Fe, O, Cd, and S. Ti 2p peaks at 458.2/464.2 eV (TiO₂), Fe 2p peaks at 710.6/724.1 eV (α‑Fe₂O₃), O 1s peaks at 531.2/531.9 eV (Ti–O/Fe–O), Cd 3d₅/₂ at 405.2 eV, and S 2p peaks at 161.5/162.6 eV (CdS).
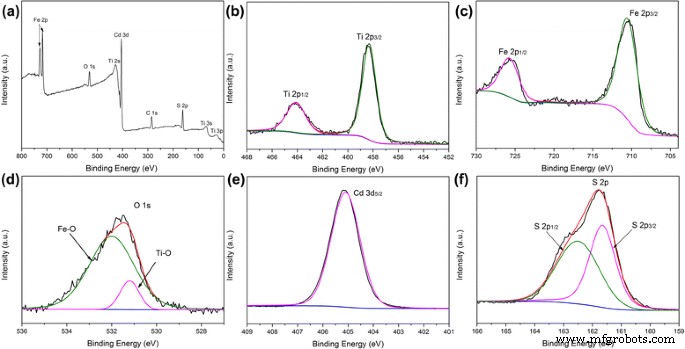
a XPS survey; b Ti 2p; c Fe 2p; d O 1s; e Cd 3d; f S 2p.
UV‑vis absorption (Fig. 4a) shows TiO₂’s band edge at 400 nm. Fe₂O₃/TiO₂ extends absorption to ~540 nm, and CdS/Fe₂O₃/TiO₂ further to ~580 nm, confirming successful visible‑light tuning. PL spectra (Fig. 4b) reveal reduced recombination: TiO₂ > Fe₂O₃/TiO₂ > CdS/Fe₂O₃/TiO₂. Time‑resolved fluorescence (Additional file 3) indicates progressively longer carrier lifetimes with each modification, underscoring improved charge separation.
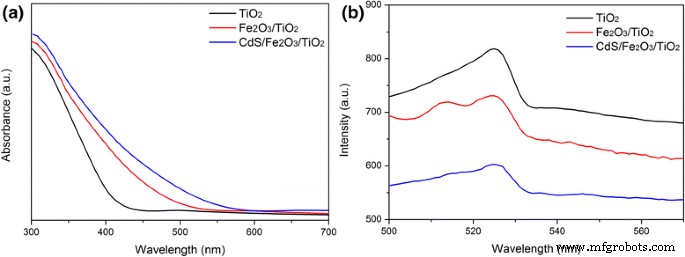
a UV‑vis absorption; b PL of TiO₂ NR, Fe₂O₃/TiO₂ NR, and CdS/Fe₂O₃/TiO₂ NR.
Charge‑transport pathways are illustrated in Fig. 5. In the CdS/Fe₂O₃/TiO₂ ternary, CdS’s higher conduction and valence bands drive electrons into Fe₂O₃ and holes into CdS. The abnormal type‑II alignment between Fe₂O₃ and TiO₂ further channels electrons from Fe₂O₃ to TiO₂’s valence band, enhancing hole separation and electron injection into TiO₂. This architecture reduces recombination and boosts PEC efficiency.
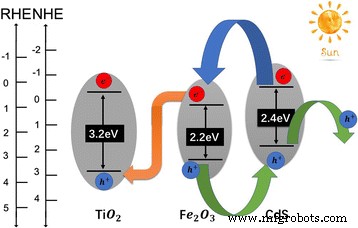
Band alignment of the CdS/Fe₂O₃/TiO₂ heterostructure.
LSV curves (Fig. 6a) and chronoamperometry (Fig. 6b) confirm that the CdS/Fe₂O₃/TiO₂ NR achieves 0.61 mA cm⁻² at 1.2 V vs. RHE, nearly twice that of bare TiO₂. The photocurrent remains stable under chopped illumination. EIS (Fig. 7a) shows reduced charge‑transfer resistance (R_ct2) from 1079.5 Ω (TiO₂) to 679.5 Ω (CdS/Fe₂O₃/TiO₂), indicating enhanced interfacial kinetics. Mott–Schottky analysis (Fig. 7b) reveals a cathodic shift of the flat‑band potential from 0.44 V (TiO₂) to 0.36 V (CdS/Fe₂O₃/TiO₂), reflecting increased electron accumulation and lower recombination.
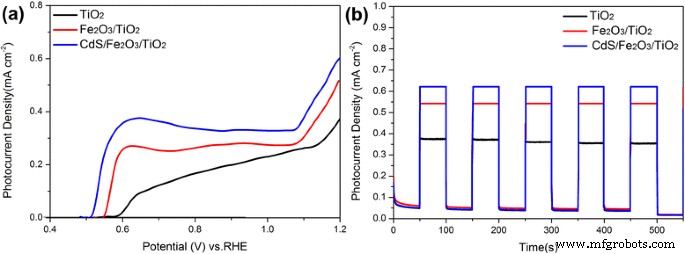
a LSV; b chronoamperometry of TiO₂ NR, Fe₂O₃/TiO₂ NR, and CdS/Fe₂O₃/TiO₂ NR.
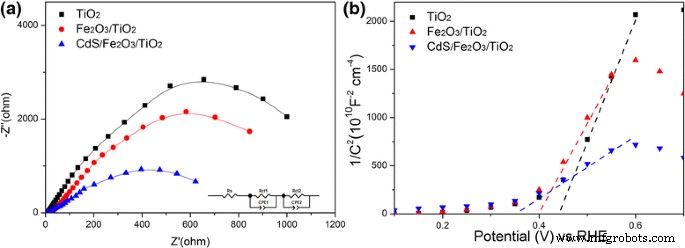
a EIS; b Mott–Schottky plots of TiO₂ NR, Fe₂O₃/TiO₂ NR, and CdS/Fe₂O₃/TiO₂ NR.
Comparative literature shows that the CdS/Fe₂O₃/TiO₂ NR’s photocurrent (0.62 mA cm⁻² at 1.23 V vs. RHE) surpasses reported Fe₂O₃/TiO₂ systems (e.g., 0.5 mA cm⁻² for Fe₂O₃/TiO₂ nanotubes) and rivals other advanced heterojunctions such as Fe‑TiO₂/Zn‑Fe₂O₃ thin films (0.262 mA cm⁻² at 0.95 V vs. SCE) [27, 28]. Thus, our design delivers outstanding and reliable PEC performance.
Conclusions
We have developed a simple SILAR‑hydrothermal approach to synthesize CdS‑modified α‑Fe₂O₃/TiO₂ NR arrays that exhibit enhanced visible‑light absorption and superior charge‑carrier separation. As photoanodes, these heterostructures deliver a photocurrent density of 0.62 mA cm⁻² at 1.23 V vs. RHE in alkaline electrolyte, outperforming pristine TiO₂ (0.32 mA cm⁻²). The strategy offers a promising route for next‑generation TiO₂‑based photoanodes in solar water splitting and related photoelectronic applications.
Nanomaterials
- S,N‑Co‑Doped Graphene Quantum Dot/TiO₂ Composites for High‑Efficiency Visible‑Light Photocatalytic Hydrogen Generation
- High‑Sensitivity Fe³⁺ Detection Using Ag‑Functionalized TiO₂ Nanotube Arrays via Anodic Stripping Voltammetry
- Boosting Planar Sb₂S₃ Solar Cell Efficiency with Cs₂CO₃‑Modified TiO₂ Electron Transport Layer
- Significant Efficiency Boost in CdS/CdSe Quantum Dot-Sensitized Solar Cells Using (001)-Oriented TiO2 Nanosheet Photoanodes
- Simple Fabrication and Performance of Polyaniline/CeO₂‑Co‑Decorated TiO₂ Nanotube Arrays for Efficient Photoelectrocatalytic Degradation of TBBPA
- PdO‑CeO₂ Rod‑Like Nanoporous Catalysts with Superior CO Oxidation and Methane Combustion Performance
- Enhanced Photoelectrochemical Water Splitting with TiO₂ Nanosheet Arrays, Layered SnS₂, and CoOx Nanoparticles
- p-Type Silicon Nanowires: Powerful Catalyst for Efficient Ozone Decomposition
- Enhancing Hematite Photoanodes: Tin and Oxygen‑Vacancy Co‑Doping for Superior Photoelectrochemical Performance
- Enhanced Dye Degradation & Antibacterial Performance of Graphene Oxide‑Doped MgO Nanostructures



