Carbon Nanomaterials for Treating Heavy‑Metal‑Contaminated Water and Advancing Environmental Remediation
Abstract
Nanotechnology, by manipulating matter at the nanoscale, offers powerful solutions for environmental challenges. Carbon‑based nanomaterials—fullerene, carbon nanotubes (CNTs), graphene, graphene oxide (GO), and activated carbon—exhibit exceptional physicochemical traits: non‑toxic chemistry, ultra‑high surface area, facile chemical modification, and straightforward regeneration. These attributes make them ideal for removing heavy‑metal ions such as lead, cadmium, arsenic, chromium, mercury, nickel, and zinc from water. This review surveys recent advances in employing these nanomaterials for heavy‑metal removal, highlights their role in broader environmental remediation, and discusses their toxicological profiles.
Introduction
Environmental pollution—stemming from industrial activity, rapid urbanization, and population growth—poses a serious threat to ecosystems and public health. Water contamination by organics, microbes, dyes, and particularly non‑biodegradable heavy‑metal ions (Pb, Cd, Zn, Ni, As, Cr, Hg) is a global crisis, causing cancer, kidney failure, neurological disorders, and developmental abnormalities (WHO).
Conventional remediation methods—ion exchange, reverse osmosis, precipitation, and biosorption—are often costly or inefficient at trace concentrations. Adsorption, by contrast, is cost‑effective, scalable, and capable of achieving sub‑ppb removal. Recent nanotechnological breakthroughs have introduced carbon nanomaterials as highly efficient adsorbents, offering tunable surface chemistry and superior mechanical properties. This article reviews their application in heavy‑metal removal, addressing both efficacy and environmental safety.
Graphical abstract illustrating graphene‑mediated purification of heavy‑metal‑contaminated water.
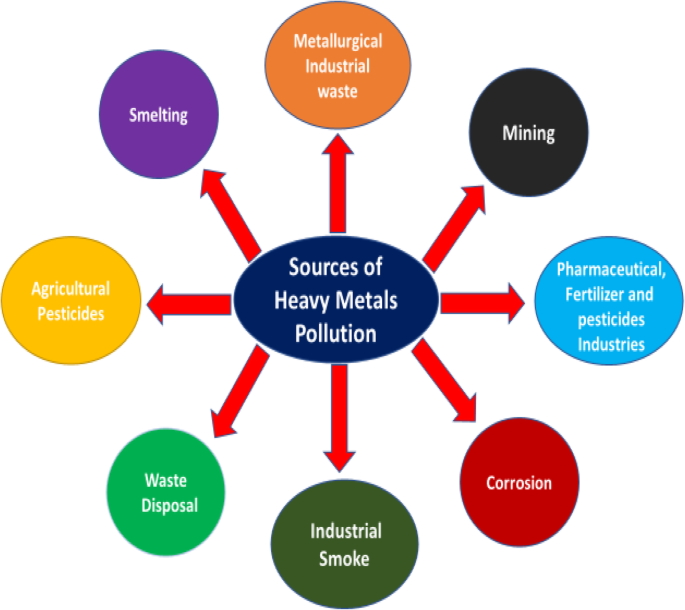
Key environmental sources of heavy‑metal contamination.
Carbon nanomaterials—graphene, GO, CNTs, fullerenes, and activated carbon—are prized for their large surface areas, mechanical strength, and chemical versatility. Their nanoscale dimensions enable high adsorption capacities and rapid kinetics, while functional groups allow selective targeting of specific metal ions. The following sections delve into each material’s unique properties, synthesis routes, adsorption mechanisms, and real‑world applications.
Human Health and Heavy Metal Toxicity
Heavy metals are defined by their atomic weights and toxicological profiles. While some, such as iron and Cr(III), are essential, others—including Pb, Cd, Hg, As, and Cr(VI)—pose significant health risks. Exposure routes (ingestion, inhalation, dermal contact) and dose determine toxicity, often leading to irreversible organ damage or carcinogenesis.
Mercury (Hg)
Mercury, a liquid d‑block element (atomic number 80), originates from mining, battery production, and chemical manufacturing. Methylmercury and mercuric chloride are classified as carcinogens by the EPA. Chronic exposure impairs renal, neurological, and reproductive systems (WHO).
Lead (Pb)
Lead, released by battery recycling, pigments, and smelting, is a recognized neurotoxin and carcinogen. It induces cognitive deficits, renal dysfunction, and cardiovascular complications (EPA).
Arsenic (As)
Arsenic—primarily as arsenate and arsenite—arises from pesticide use and mineral deposits. It disrupts cellular respiration by binding sulfhydryl groups, leading to carcinogenic and systemic effects.
Cadmium (Cd)
Cadmium, produced in electroplating and alloy manufacturing, is nephrotoxic and carcinogenic, ranking seventh on the ATSDR toxicity list.
Chromium (Cr)
While Cr(III) is essential, Cr(VI) is a potent carcinogen. Industrial effluents frequently exceed WHO limits of 50 µg/L for Cr(VI).
Zinc (Zn)
Excessive zinc exposure can disrupt iron absorption and is toxic to aquatic organisms.
Classification of Carbon Nanomaterials Based on their Dimensions
Nanomaterials are categorized by dimensionality: 0‑D (fullerenes, quantum dots), 1‑D (CNTs), 2‑D (graphene), and 3‑D (graphite). Each dimension imparts distinct surface chemistry and mechanical properties.
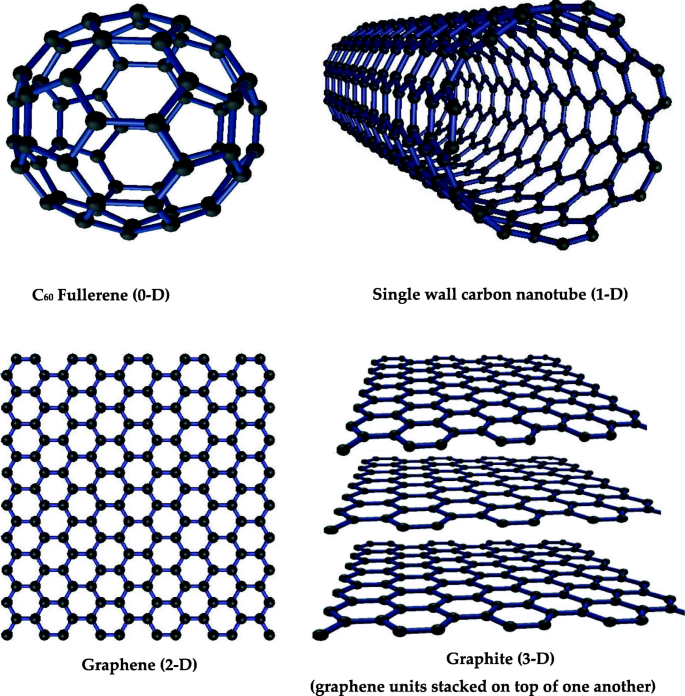
Illustration of carbon nanomaterials across dimensions.
Fullerenes in Environmental Remediation
Fullerenes, discovered in 1985, are closed‑cage carbon molecules (C₆₀, C₇₀). Their high surface area, electron affinity, and ability to form functionalized derivatives make them attractive for adsorption and photocatalysis.
Functionalized fullerenes (fullerols) act as visible‑light photogenerated ROS sources, degrading organic pollutants while remaining recoverable. They also adsorb heavy metals via defect‑site interactions, following Langmuir isotherms (e.g., Cu²⁺ removal). Though pristine fullerenes are costly, they can be incorporated into composites—activated carbon, lignin, zeolites—to enhance performance and lower cost.
Biocompatibility
Extensive in‑vitro and in‑vivo studies confirm fullerene’s low cytotoxicity and suitability for environmental applications.
Carbon Nanotubes (CNTs)
CNTs, comprising single‑walled (SWCNTs) or multi‑walled (MWCNTs) graphene cylinders, exhibit specific surface areas up to 1500 m²/g and tunable functional groups, enabling selective heavy‑metal capture.
Environmental Applications
CNTs enhance solar‑thermal collectors, photovoltaic devices, supercapacitors, and sensors while maintaining biocompatibility. In water treatment, oxidized or plasma‑treated CNTs adsorb Cr(VI), Pb²⁺, Cd²⁺, and Ni²⁺ with capacities ranging from 10–186 mg/g. Functionalization (COOH, NH₂, OH) boosts adsorption by up to 200 % (e.g., COOH‑SWCNTs: 96 mg/g for Pb²⁺).
MWCNTs in Water Purification
MWCNTs, often combined with metal oxides (Fe₃O₄, ZrO₂, MnO₂), exhibit superior adsorption for Cr(VI), As(III), Ni²⁺, Pb²⁺, and Cu²⁺. Their performance follows Langmuir or Freundlich models, and plasma‑oxidized variants display the highest capacities (≈186 mg/g for Cr(VI)). Reusability is achieved by pH‑driven desorption.
Biocompatibility of CNTs
CNTs are well‑documented as biocompatible, enabling safe deployment in environmental remediation.
Graphene and Graphene Oxide in Heavy‑Metal Removal
Graphene’s single‑atom thickness confers a theoretical strength 200× that of steel and an ultrahigh surface area. Functionalized graphene and GO composites (e.g., Fe₃O₄/GO, ZnO/GO) achieve removal efficiencies exceeding 90 % for Pb²⁺, Cu²⁺, Ni²⁺, Cd²⁺, and Cr³⁺, with capacities up to 689 mg/g.
Graphene‑based microbots, combining GO, Ni, and Pt layers, demonstrate 80 % Pb²⁺ removal via self‑propulsion or magnetic actuation, enabling simultaneous extraction and recovery (see example).
Biocompatibility
Graphene derivatives exhibit low cytotoxicity in vitro and in vivo, supporting their safe use in water treatment.
Activated Carbon
Activated carbon, produced from coal, biomass, or waste, offers >3000 m²/g surface area and is inexpensive to manufacture. Functionalized AC (e.g., sulfonated, ZnO‑loaded) removes Pb²⁺, Cd²⁺, Cu²⁺, and Ni²⁺ with capacities ranging from 52–238 mg/g. Its mechanical robustness allows repeated regeneration with minimal loss in performance.
Biocompatibility
AC is inert, non‑toxic, and widely used as an oral sorbent for toxin removal, confirming its environmental safety.
Conclusion
Carbon nanomaterials—fullerenes, CNTs, graphene, GO, and activated carbon—exhibit exceptional adsorption capacities, rapid kinetics, and recyclability for heavy‑metal removal. Their high surface area, tunable chemistry, and biocompatibility render them superior to conventional adsorbents. Future research should focus on scalable synthesis, cost reduction, and life‑cycle assessment to fully realize their potential in global water remediation.
Availability of Data and Materials
All data are fully available without restriction.
Abbreviations
- AC:
Activated carbon
- GO:
Graphene oxide
- MWCNTs:
Multi‑wall carbon nanotubes
- r‑GO:
Reduced graphene oxide
- SWCNTs:
Single‑wall carbon nanotubes
Nanomaterials
- Fluorescent Nanomaterials: Advances, Synthesis, and Biomedical Applications
- Comprehensive Atomic & Electronic Analysis of Stacked-Cup Carbon Nanofibers
- Graphene‑Polymer Composites for High‑Performance Supercapacitors: A Comprehensive Review
- Comparative Analysis of Electrochemical, Biomedical, and Thermal Properties of Natural vs. Synthetic Nanomaterials
- Enhancing CL‑20 Safety: Lowering Impact Sensitivity via Carbon Nanomaterial‑Assisted Thermal Conductivity
- Nanomaterials and Nanotechnologies in Wastewater Treatment: Insights from a Bibliometric Analysis (1997‑2016)
- Advances in Carbon Nanotube Assembly and Integration for Next‑Generation Applications
- Affordable Green Fluorescent Carbon Dots: Enhanced Bioimaging & Targeted Drug Delivery
- Advanced Wastewater & Water Treatment Solutions: Sustainable Technologies for Clean Water
- Harnessing IoT for Smarter Wastewater Treatment and Management



