Synergistic Flame Retardancy of Fullerene‑Anchored Reduced Graphene Oxide Hybrids in Epoxy Resin
Abstract
We synthesized a hybrid comprising fullerene (C60) covalently bonded to polyethyleneimine (PEI)‑modified reduced graphene oxide (rGO) and incorporated it into an epoxy (EP) matrix. Detailed structural analysis confirmed uniform C60 anchoring on PEI‑rGO. The resulting C60-PEI‑rGO/EP nanocomposites exhibited remarkable flame‑retardant performance: the time to ignition (tign) increased from 68 s (neat EP) to 89 s (1.0 wt % C60-PEI‑rGO), a 31 % improvement. Peak heat release rate (PHRR) and total heat release (THR) decreased by 40.0 % and 15.6 %, respectively. The hybrid’s effectiveness stems from three synergistic mechanisms: radical scavenging by C60, barrier protection from the layered rGO, and cross‑linking density enhancement via PEI. Consequently, the hybrid simultaneously boosts thermal stability and mechanical strength, offering a promising route to fire‑retardant thermosetting resins with superior multifunctionality.
Background
Polymeric materials dominate construction, electronics, and coatings due to their lightweight and versatility, yet their inherent flammability poses safety risks. Nanofillers have emerged as a powerful class of flame retardants, delivering high efficiency at low loadings while preserving base material properties. Their flame‑retarding action is evident in reduced peak heat release rate (PHRR), total heat release (THR), and increased limiting oxygen index (LOI).
While nanofillers markedly improve flame resistance in thermoplastics, their effect on thermosetting resins is limited. For instance, graphene oxide (GO) added to epoxy (EP) reduced PHRR by only 16 % at 1 wt % loading, and graphene achieved a 23 % PHRR drop under similar conditions. This muted response is attributed to the high cross‑link density of thermosets, which hampers significant structural alteration, and the limited barrier efficacy of nanofillers alone.
To enhance flame retardancy in thermosets, researchers have functionalized graphene with organic moieties or combined it with other additives. A notable example is the grafting of octa‑aminophenyl polyhedral oligomeric silsesquioxane (OapPOSS) onto graphene, producing OapPOSS‑rGO, which improved EP flame resistance. However, key metrics such as ignition time and peak heat release remain underreported, and the underlying synergistic mechanisms warrant deeper investigation.
Fullerene (C60) has attracted attention for its ability to quench free radicals, thereby delaying thermo‑oxidative degradation. Yet, its tendency to aggregate reduces its efficacy. Combining C60 with carbon nanofillers—particularly layered graphene—offers dual benefits: C60 provides radical scavenging, while graphene offers barrier protection and facilitates better dispersion. Prior studies have applied C60–graphene hybrids to thermoplastics, but no work has explored their impact on thermosetting resins.
In this study, we designed a C60-PEI‑rGO hybrid via a three‑step reaction and incorporated it into EP. The hybrid’s amino‑rich, loosely layered architecture promotes uniform dispersion, radical absorption, barrier action, and cross‑linking density enhancement, thereby aiming to simultaneously elevate flame retardancy, thermal stability, and mechanical performance.
Methods
Materials
Graphite (3000 mesh), sulfuric acid (H2SO4 98 %), sodium nitrate, potassium permanganate, hydrogen peroxide, ethanol, dimethyl sulfoxide (DMSO), toluene, acetone, and distilled water were analytical grade. Fullerene (C60, >99 % purity) was sourced from Henan Puyang Co. Ltd. Branched polyethyleneimine (PEI, 50 % aq., Mn = 70 000) was purchased from Sigma‑Adrich. Epoxy monomer diglycidyl ether of bisphenol A (DGEBA) and curing agent diethyltoluenediamine (DETDA) were obtained from Shanghai Resin Factory Co. Ltd. and Chongshun Chemical Co. Ltd., respectively.
Preparation of C60-PEI‑rGO
GO was synthesized via a modified Hummer’s method (see Additional file 1). PEI‑modified rGO (PEI‑rGO) was prepared by reacting PEI with GO. 150 mg of PEI‑rGO was dispersed in 300 mL DMSO using ultrasonication for 30 min. This dispersion, together with 300 mg C60, was added to a 350 mL DMSO‑toluene (4:3 v/v) mixture and sonicated for 30 min at room temperature, then stirred at 90 °C for 24 h. The product was washed with toluene and ethanol three times, then dried at 60 °C under vacuum for 12 h, yielding C60-PEI‑rGO (Scheme 1).
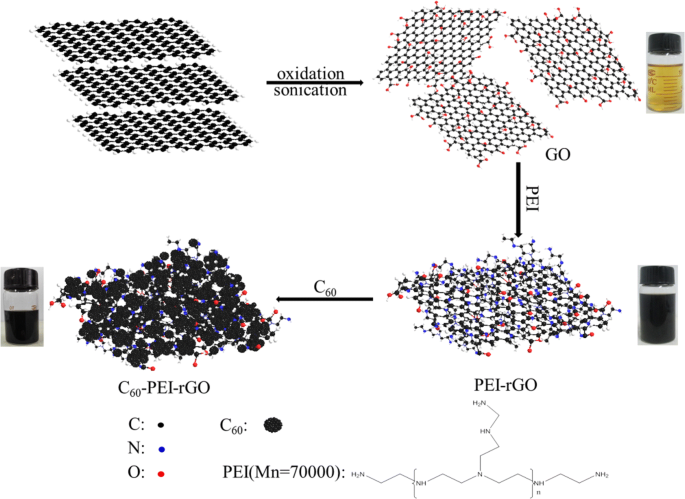
Schematic illustration of the preparation of C60-PEI‑rGO
Preparation of EP Resin and Nanocomposites
DGEBA and DETDA were blended at a weight ratio of 1:0.234, heated to 100 °C for 15 min, and vigorously stirred to form a light yellow prepolymer. The mixture was degassed at 110 °C for 30 min and poured into a preheated (100 °C) U‑type mold. Curing followed a two‑step schedule: 120 °C/1 h + 180 °C/2.5 h, then post‑curing at 190 °C/2 h, producing cured EP resin. For nanocomposites, PEI‑rGO, C60, or C60-PEI‑rGO (0.4–1.0 wt %) were dispersed in the EP prepolymer/ethanol mixture by sonication for 30 min, degassed at 60 °C, and cured with the same thermal cycle. The resulting samples were labeled PEI‑rGO1.0/EP, C601.0/EP, and C60-PEI‑rGOn/EP (n = 0.4, 0.6, 0.8, 1.0).
Apparatus and Experimental Method
Morphology and microstructure were examined by AFM (Veeco Instruments), TEM (JEOL JEM‑2010), SEM (HITACHI SU8010/EDX), and FTIR (AVATAR360N). TGA of nanofillers (TA Instruments STA449C) spanned 25–800 °C in N2 at 10 °C/min. EP and composites were thermally analyzed from 25–800 °C in air at 10–40 °C/min. DMA (TA DMA Q800) measured storage modulus from 25–250 °C at 1 Hz. Tensile tests followed ASTM D638 (5 mm/min). LOI values were determined per ASTM D2863/77. Cone calorimetry (FTT device) followed ISO 5660 with 35 kW/m² flux.
Results and Discussion
Characterization of GO, PEI‑GO, and C60-PEI‑rGO
PEI‑rGO and C60-PEI‑rGO dispersed readily in ethanol, forming stable colloids—a sign of successful PEI functionalization and C60 anchoring. Color change from yellow GO to black PEI‑rGO and C60-PEI‑rGO confirms reduction of GO.
FTIR spectra (Fig. 1) revealed diminished O–H and C=O bands in PEI‑rGO, indicating partial reduction and amide bond formation. In C60-PEI‑rGO, characteristic C60 peaks (1426, 1180, 574, 525 cm−1) and a new C60–H band at 2973 cm−1 confirm covalent attachment.
Raman XPS data (Fig. 2) showed C1s peaks at 286.7 and 532.6 eV for all samples. N1s peaks at 399.7 eV (PEI‑rGO) and 400.1 eV (C60-PEI‑rGO) confirm amide formation. Elemental analysis indicated C content rising to 86.55 at.% in C60-PEI‑rGO, implying a ~45 wt % C60 loading.
AFM and TEM images (Figs. 3–4) showed GO sheets (~0.9 nm thick) transformed into ~1.5 nm PEI‑rGO and further into a loosely layered C60-PEI‑rGO with ~20 nm C60 aggregates uniformly distributed. This architecture prevents restacking and enhances interfacial interaction with EP.
TGA (Fig. 5) demonstrated superior thermal stability of C60-PEI‑rGO: a 79.4 % residue at 600 °C, compared to 42.9 % for GO. Weight loss analysis suggested ~50 wt % C60 loading, consistent with XPS.
Microstructure of C60-PEI‑rGO/EP Nanocomposites
SEM of fractured surfaces (Fig. 6) confirmed homogeneous dispersion of PEI‑rGO and C60-PEI‑rGO without significant agglomeration, indicating strong interfacial adhesion. DMA curves (Fig. 7) revealed increased rubber plateau modulus for all hybrids, with C60-PEI‑rGO1.0/EP exhibiting the highest modulus—attributable to PEI cross‑linking and the rough, layered surface enhancing physical bonding.
Flame Retardancy and Mechanism
Cone calorimetry (Fig. 8, Table 1) showed that C60-PEI‑rGO1.0/EP extended tign by 21 s and delayed peak heat release by 28 s relative to neat EP. PHRR dropped by 40.0 % and THR by 15.6 %. LOI rose to 30.1 % for C60-PEI‑rGO0.8/EP, 1.18× that of neat EP. These improvements surpass those of PEI‑rGO or C60 alone, underscoring synergistic effects.
Thermal‑oxidation kinetics (Fig. 9–10, Tables 2–3) revealed increased activation energy (Ea) during the first decomposition stage for C60-PEI‑rGO/EP, confirming delayed chain scission. The presence of C60 scavenges radicals, while the layered rGO hinders volatile transport, collectively raising Ea and suppressing combustion.
Proposed mechanism (Fig. 11): (1) C60 aggregates trap radicals, raising decomposition energy; (2) the layered structure forms a physical barrier, limiting heat and mass transfer; (3) PEI‑induced cross‑linking densifies the matrix, further enhancing resistance.
Mechanical and Thermal Properties
Tensile testing (Fig. 12) demonstrated that all C60-PEI‑rGO/EP composites surpassed neat EP in strength and modulus. C60-PEI‑rGO1.0/EP achieved a Young’s modulus of 2810 MPa (1.35× EP) and tensile strength of 77.4 MPa (1.22× EP). Rough fractured surfaces indicate efficient stress transfer between filler and matrix.
DMA results (Fig. 13) showed a 53.7 % increase in storage modulus at 30 °C and a Tg rise of 11.3 °C for C60-PEI‑rGO1.0/EP, reflecting enhanced cross‑linking and restricted chain mobility.
TGA/DTG profiles (Fig. 14) revealed higher onset temperatures (ΔTonset = 28 °C) and maximum degradation temperatures (ΔTmax = 16 °C) for C60-PEI‑rGO1.0/EP, confirming superior thermal stability. The improved char microstructure and delayed oxidation further support the synergistic flame‑retardant action.
In summary, the C60-PEI‑rGO hybrid delivers multi‑faceted benefits—flame retardancy, mechanical reinforcement, and thermal resilience—through radical trapping, barrier formation, and cross‑linking enhancement.
Conclusions
The covalently anchored C60-PEI‑rGO hybrid exhibits a loosely layered, amino‑rich structure with uniformly dispersed ~20 nm C60 aggregates. Incorporation of 1.0 wt % of this hybrid into EP reduced PHRR by 40.0 % and THR by 15.6 %, while increasing tign and peak‑time by 21 s and 28 s, respectively. The hybrid’s efficacy originates from enhanced cross‑link density via PEI, barrier protection from the layered rGO, and radical scavenging by C60. Consequently, it represents a potent, multifunctional nanofiller for advanced fire‑retardant thermosetting resins.
Abbreviations
- AFM:
Atomic force microscope
- C60:
Fullerene
- DETDA:
Diethyltoluenediamine
- DGEBA:
Diglycidyl ether of bisphenol A
- DMA:
Dynamic mechanical analysis
- DMSO:
Dimethyl sulfoxide
- EP:
Epoxy
- FTIR:
Fourier transform infrared spectrometer
- GO:
Graphene oxide
- LOI:
Limiting oxygen index
- PEI:
Branched polyethyleneimine
- PHRR:
Peak heat release rate
- rGO:
Reduced graphene oxide
- SEM:
Scanning electron microscope
- TEM:
Transmission electron microscopy
- TGA:
Thermogravimetric analysis
- THR:
Total heat release
- TSR:
Total smoke release
Nanomaterials
- Elevate Your Home with Custom Epoxy Resin Art
- Titanate Nanotube-Enhanced Graphene Oxide Nanocomposites: Boosting Flame Retardancy and Photodegradation in Flexible PVC
- Assessing the Biosafety and Antibacterial Efficacy of Graphene and Graphene Oxide for Orthopedic Implant Applications
- Green Synthesis of Metal‑Oxide Nanoparticles with Gum Karaya and Their Ecotoxicological Impact on *Chlamydomonas reinhardtii*
- Enhancing Supercapacitor Energy Density with Anionic Surfactant‑Intercalated Thermally Reduced Graphene Oxide in Ionic Liquid Electrolytes
- Quaternized PVA/Graphene Oxide Composite Membrane Enhances Ethanol Barrier and Ionic Conductivity for Passive Alkaline DEFCs
- Reducing Cytotoxicity of Silver Nanoparticle Coatings with Graphene Oxide: Enhanced Antibacterial Performance and Biocompatibility of an Ag–GO Nanocomposite
- Enhancing Glass Fiber‑Aluminum Laminates: Surface Treatment & Graphene Oxide Interleaf Boost Interlaminar Toughness
- Eco‑Friendly, Cost‑Effective Synthesis of Tin Oxide Nanoparticles: A Comprehensive Review of Methods, Formation Mechanisms, and Applications
- Taurine-Functionalized Graphene Oxide for 5‑Fluorouracil Delivery: Fabrication, Characterization & Anticancer Efficacy



