Graphene Oxide‑Integrated nHAC/PLGA Scaffolds Promote Osteoblast Proliferation and Bone Regeneration
Abstract
Biodegradable porous scaffolds are central to bone tissue engineering. In this study, nano‑hydroxyapatite/collagen (nHAC) blended with poly(lactic‑co‑glycolic acid) (PLGA) and varying amounts of graphene oxide (GO) were fabricated by freeze‑drying. The resulting composites exhibited a continuous three‑dimensional pore network. GO slightly enhanced hydrophilicity and significantly increased Young’s modulus, with the 1.5 wt% GO sample reaching 10.20 ± 1.28 GPa compared to 7.53 ± 1.25 GPa for the GO‑free scaffold. In vitro, MC3T3‑E1 osteoblasts displayed markedly higher adhesion and proliferation on the 1.5 wt% GO scaffold, as confirmed by CCK‑8 assays and SEM imaging. These findings demonstrate that nHAC/PLGA/GO scaffolds combine excellent cytocompatibility with superior mechanical performance, positioning them as promising candidates for bone regeneration applications.
Background
Three‑dimensional porous scaffolds that emulate the bone extracellular matrix are essential for successful bone repair. Conventional fabrication techniques—electrospinning, computational topology design (CTD) combined with solid free‑form fabrication (SFF), and freeze‑drying—offer varying degrees of structural control and scalability. Freeze‑drying, in particular, provides a straightforward route to generate highly interconnected pores by sublimating a frozen solvent under vacuum.
Native bone consists of a hierarchical assembly of collagen and hydroxyapatite (HA). Nano‑hydroxyapatite/collagen (nHAC) composites mimic this natural architecture, delivering favorable biocompatibility and bioresorbability. However, collagen’s intrinsic brittleness and rapid degradation limit its use. Poly(lactic‑co‑glycolic acid) (PLGA) introduces mechanical robustness and tunable degradation rates, while GO offers unique physicochemical properties—high surface area, mechanical reinforcement, and functional groups that can modulate cell behaviour.
Previous work has shown GO‑enhanced scaffolds improve osteogenic differentiation and cell proliferation. Here, we systematically evaluated how incremental GO loading (0.0, 0.5, 1.0, and 1.5 wt%) affects the structural, mechanical, hydrophilic, and biological performance of nHAC/PLGA composites.
Results and Discussion
Structure of nHAC/PLGA/GO Composite Scaffolds
The fabrication process involved dissolving type I collagen in acetic acid, co‑precipitating Ca and P ions to form nHAC, mixing with PLGA and dispersed GO, followed by freeze‑drying. Scanning electron microscopy (SEM) revealed a highly porous architecture with interconnected pores ranging from 0 to 200 nm. The pore distribution was quantified by an automated surface area analyzer (Fig. 2f), confirming a predominance of sub‑100 nm pores, a size range conducive to osteoblast infiltration.
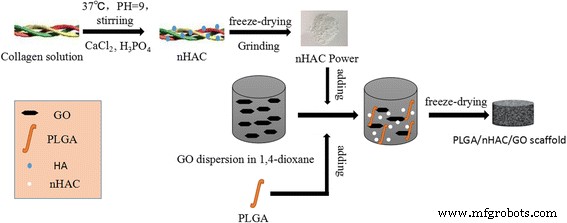
Schematic of scaffold fabrication via freeze‑drying.
Physicochemical and Mechanical Characterization
Powder X‑ray diffraction (XRD) confirmed the presence of hexagonal HA; GO signals were not discernible due to its low loading. FT‑IR spectra displayed characteristic collagen amide bands and phosphate peaks, with subtle shifts indicating chemical interaction between collagen, HA, PLGA, and GO.
Quantitative nano‑mechanical AFM measurements (QNM‑AFM) revealed a monotonic increase in Young’s modulus with GO content: 7.53 ± 1.25 GPa (0 wt%), 8.34 ± 1.00 GPa (0.5 wt%), 9.15 ± 0.85 GPa (1.0 wt%), and 10.20 ± 1.28 GPa (1.5 wt%). The 1.5 wt% GO scaffold displayed a statistically significant stiffness advantage over the GO‑free counterpart, reflecting GO’s reinforcing effect.
Contact angle measurements showed a gradual decrease from 125.1° (GO‑free) to 101.4° (1.5 wt%), indicating improved surface wettability due to GO’s hydrophilic functional groups (Fig. 4f).
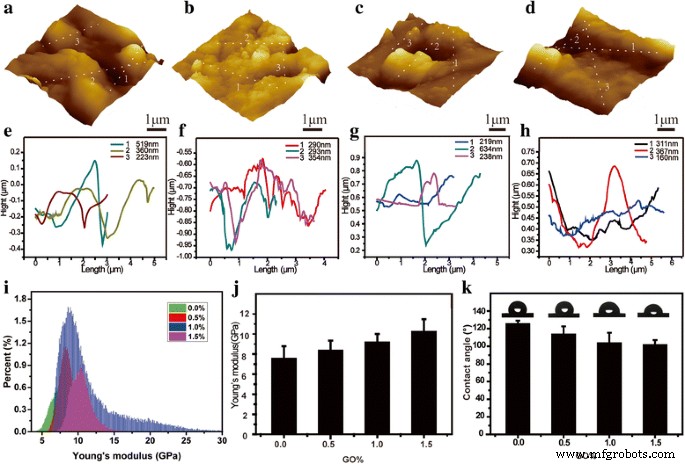
AFM images (a‑d) and stiffness maps (i) of the four scaffold variants; contact angles (k) measured by sessile drop method.
Cell Culture and Cytotoxicity
MC3T3‑E1 osteoblasts seeded on the scaffolds exhibited increasing metabolic activity over 7 days (CCK‑8 assay). While early proliferation on the 0.5 wt% and 1.0 wt% GO scaffolds was slightly lower than the GO‑free control, the 1.5 wt% GO scaffold matched and surpassed the control by days 3, 5, and 7 (Fig. 5). SEM images confirmed extensive cell spreading and network formation, with cells on the GO‑laden scaffolds displaying larger, more flattened morphologies (Fig. 6).
NIH‑3T3 fibroblasts exposed to scaffold suspensions for 24 h showed no significant cytotoxicity; viability remained above 97 % across all GO loadings, affirming the biocompatibility of the composites (Fig. 7).
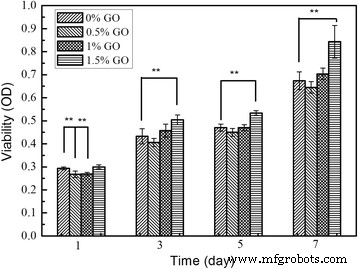
Cell viability of MC3T3‑E1 on the four scaffold variants (*** p < 0.01).
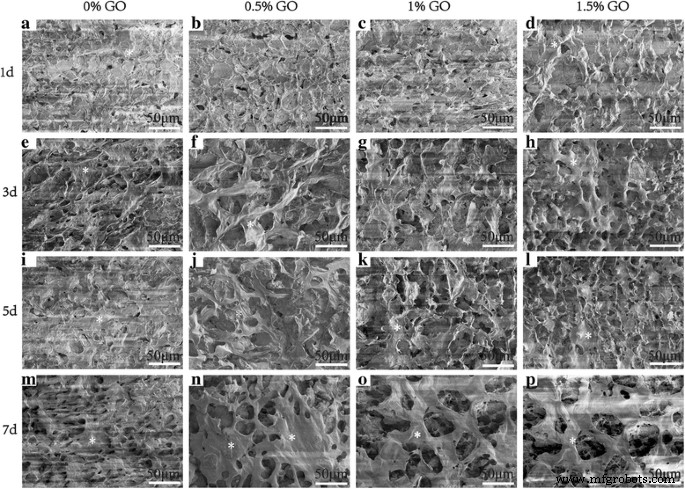
SEM images of MC3T3‑E1 cells at 1, 3, 5, and 7 days on the four scaffold types.
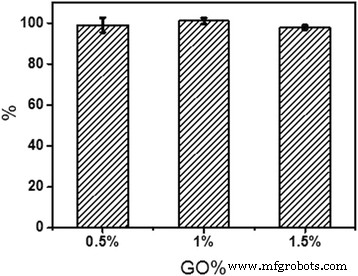
Relative viability of NIH‑3T3 cells after 24 h exposure to scaffold suspensions.
Conclusions
Freeze‑drying enabled the fabrication of nHAC/PLGA/GO scaffolds with interconnected porosity, enhanced mechanical stiffness, and improved hydrophilicity. The 1.5 wt% GO scaffold demonstrated superior osteoblast adhesion and proliferation while maintaining excellent cytocompatibility, underscoring its potential as a bone tissue engineering scaffold.
Methods
Materials
Type I collagen, PLGA (75:25, Mw = 95 kDa), GO, and cell culture reagents were sourced from established suppliers. MC3T3‑E1 osteoblasts and NIH‑3T3 fibroblasts were obtained from reputable cell banks.
Scaffold Preparation
Collagen was dissolved in 0.5 M acetic acid (4 g L⁻¹). Calcium and phosphate solutions (Ca/P = 1.66) were added dropwise at 100 drops min⁻¹, stirred, and titrated to pH 9 with ammonia at 37 °C. After 24 h, the precipitate was collected, freeze‑dried, and ground to produce nHAC powder.
GO was dispersed in dioxane (0.0, 0.5, 1.0, 1.5 g L⁻¹) via ultrasonication, then mixed with PLGA (10 % m/v) and stirred 12 h. nHAC was added at a 1:1 weight ratio to PLGA, followed by 4 h ultrasonication. The mixture was frozen at –20 °C overnight and lyophilized to yield the final scaffolds.
Characterization
SEM, AFM (QNM mode), FT‑IR, XRD, contact angle, and porosity measurements were conducted using standard protocols. All data were collected in triplicate.
Cell Culture and Assays
MC3T3‑E1 cells were seeded at 2.5 × 10⁴ cells mL⁻¹ on scaffold discs and cultured in DMEM +10 % FBS +3 % antibiotics at 37 °C, 5 % CO₂. Proliferation was assessed by CCK‑8 at days 1, 3, 5, and 7. For SEM, cells were fixed with 4 % paraformaldehyde, dehydrated, critical‑point dried, and sputter‑coated with gold.
Cytotoxicity Test
NIH‑3T3 cells were exposed to 100 mg mL⁻¹ scaffold suspensions for 24 h, followed by CCK‑8 assay. Cell viability was calculated relative to control cultures.
Statistical Analysis
Data are expressed as mean ± SD. Student’s t‑test was applied; p < 0.05 was considered significant.
Nanomaterials
- High‑Efficiency Graphene Solar Cells: 9% Power Conversion with TFSA Doping
- Improved Osteoinductivity with rhBMP‑2-Loaded Silk Fibroin Scaffolds for Bone Tissue Engineering
- High-Sensitivity Graphene Oxide Fluorescent Aptasensor for Rapid Detection of CCRF-CEM Leukemia Cells
- Enhancing Supercapacitor Energy Density with Anionic Surfactant‑Intercalated Thermally Reduced Graphene Oxide in Ionic Liquid Electrolytes
- Synergistic Flame Retardancy of Fullerene‑Anchored Reduced Graphene Oxide Hybrids in Epoxy Resin
- Quaternized PVA/Graphene Oxide Composite Membrane Enhances Ethanol Barrier and Ionic Conductivity for Passive Alkaline DEFCs
- Reducing Cytotoxicity of Silver Nanoparticle Coatings with Graphene Oxide: Enhanced Antibacterial Performance and Biocompatibility of an Ag–GO Nanocomposite
- Optimizing Niobium-Doped Titanium Oxide Layers for Enhanced Silicon Quantum Dot Solar Cells
- SOX2 Modulates the CCAT1/miR‑185‑3p/FOXP3 Pathway to Control Proliferation and Self‑Renewal of Cervical Cancer Stem Cells
- AgO X Wetting Layer Boosts Metal Oxide Electrodes for Higher Efficiency CIGS Solar Cells



