Dynamic Hierarchical Self‑Assembled Hexabenzocoronene for High‑Performance Lithium‑Ion Battery Anodes
Abstract
In this work we investigate the dynamic, hierarchical self‑assembly of hexabenzocoronene (HBC) nano‑graphene and its impact on lithium‑ion storage. The molecules re‑organise under their own internal pressure, creating water‑rippling d‑spacing patterns. Comprehensive material‑characterisation shows that the assembly is governed solely by van der Waals forces within layers and π–π interactions between layers, with no covalent bonds linking the molecules. This ordered arrangement yields multiple lithium‑ion shuttle channels—both interlayer galleries and intrananosheet pathways—significantly shortening diffusion distances and mitigating irreversible capacity loss.
Introduction
As the demand for sustainable energy storage grows, researchers are turning to graphene‑based materials for next‑generation lithium‑ion battery (LIB) anodes. Nanographene and its composites have shown promising electrochemical performance, and core–shell architectures that encapsulate silicon or metallic nanostructures have further enhanced anode behaviour. Graphene remains the benchmark for replacing graphite, owing to its exceptional conductivity, mechanical strength, and large surface area. Its discovery by Geim and Novoselov in 2004 via the simple Scotch‑tape method has spurred extensive exploration, including liquid‑phase exfoliation, thermal annealing, chemical vapour deposition, and epitaxial growth on SiC.
Hexabenzocoronene (HBC) is a prototypical nanographene molecule whose modular size and tunability make it an ideal model for studying self‑assembly. Each HBC unit consists of sp2‑hybridised carbon atoms arranged in a hexagonal honeycomb lattice, forming a nano‑graphene sheet. While the chemistry of HBC is well established, the mechanisms that drive its overlap and aggregation into complex morphologies remain underexplored. Understanding how individual HBC molecules stack and interact is essential for designing materials with tailored electronic and ionic transport properties.
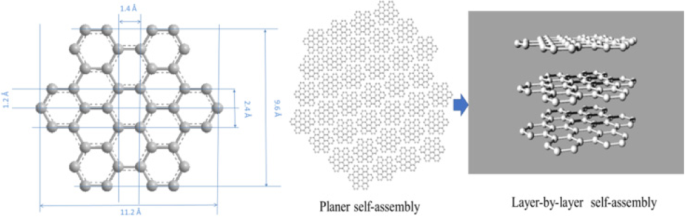
Hexabenzocoronene structure and self‑assembly diagram
This paper elucidates the dynamic, hierarchical self‑assembly of HBC, linking the observed d‑spacing variations at the molecular level to the resulting nanostructural clusters. We provide an in‑depth analysis of the factors that govern the internal architecture of nano‑graphene, revealing how these structures influence lithium‑ion transport.
Methods/Experimental
Materials
Hexabenzocoronene was synthesized following established protocols. Solvents were freshly distilled and dried under argon; all reagents were analytical grade and sourced from Shanghai Chemical Corp. Thin‑layer chromatography (TLC) was performed on silica gel 60 F254 (Merck DGaA, Germany). The electrolyte solution (0.1 M tetra‑n‑butylammonium perchlorate, TBAP) was purchased from Shanghai Annaiji Technology Co., Ltd. Deionised water was used throughout.
Characterisation
Morphology and lattice fringes were examined using a JEOL JCM‑6000Plus scanning electron microscope (SEM), a JEOL H‑7000 transmission electron microscope (TEM), and a JEOL JEM‑2100 high‑resolution TEM (HRTEM).
Electrochemical Measurements
Electrochemical testing employed a Shanghai Chenhua CHI660e system with a three‑electrode configuration: platinum wire counter, platinum plate working electrode, and a saturated calomel reference. The supporting electrolyte (0.1 M TBAP) and acetonitrile (ACN) were used. The glassy‑carbon electrode was polished with 0.05 µm alumina slurry, rinsed, sonicated in acetone, and dried. HBC suspension was drop‑cast onto the electrode and allowed to dry. Cyclic voltammetry was performed at 0.1 mV s−1 in the Li+/Li vs Ag/AgCl potential window.
Results and Discussion
Hexabenzocoronene’s conjugated π‑system confers strong π–π stacking, yet the synthesis yields are modest due to multiple reaction steps (Sonogashira coupling, Diels‑Alder, Lewis‑acid catalysed cyclisation, and deprotonation). The final product is isolated as a yellowish solid after repeated methanol precipitation and methanol/acetone washing.
Despite extensive study of HBC as a material, its self‑assembly behaviour has received limited attention. Here we focus on the dynamic aggregation process, dissecting the internal distribution of nano‑graphene sheets and elucidating the factors that govern self‑assembly and, consequently, electrochemical performance.
Transmission electron microscopy revealed that HBC molecules self‑assemble into regular thin sheets that stack into intermittent fragments held tightly together. Under mechanical stress, these stacks rearrange, forming gear‑like patterns that maintain the overall nanoscale architecture. The resulting morphology resembles a fingerprint (Fig. 2).

Nano‑graphene dynamic hierarchical assembly and rearrangement
SEM images confirm that particle size remains unchanged during rearrangement, with uniformly distributed nanoparticles and daisy‑like clusters ranging from 20 nm to 200 nm (Fig. 3). Two self‑assembly pathways are evident: edge‑overlap and lateral overlap, both facilitating close packing.

SEM and TEM images for hexabenzocoronene
High‑resolution TEM demonstrates a coherent layer spacing of 0.34 nm, with most particles comprising ~14 layers—an ultrathin, polycrystalline architecture (Fig. 4). The selected‑area diffraction pattern confirms the crystalline nature of HBC.

HRTEM image of hexabenzocoronene with dynamic hierarchical assemblies
Electrochemical testing shows a reversible capacity of 200 mAh g−1 after 100 cycles at 100 mA g−1, with a coulombic efficiency exceeding 98% (Fig. 5). The galvanostatic charge–discharge profiles remain stable over successive cycles, highlighting the structural integrity of the self‑assembled layers.
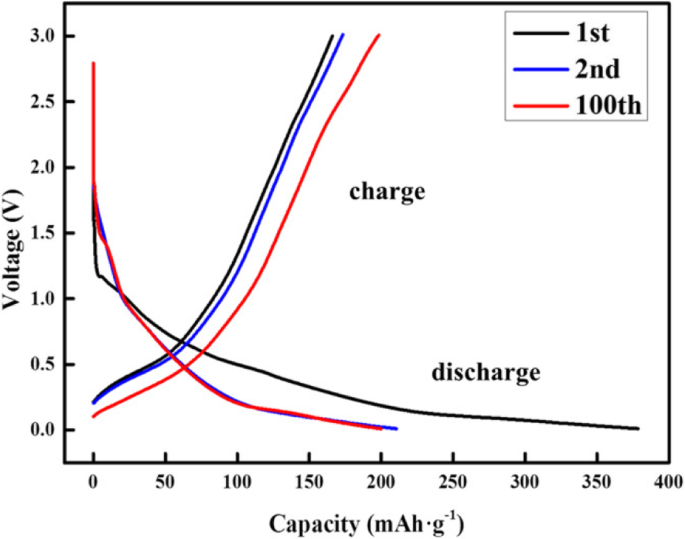
The galvanostatic discharge–charge voltage profiles of hexabenzocoronene anode as a function of cycling numbers
CV measurements at 0.1 mV s−1 display redox peaks that shift slightly with scan rate, yielding a quasi‑rectangular shape characteristic of fast lithium‑ion diffusion (Fig. 6). The HOMO energy analysis (Eq. 1) confirms the contribution of oxidation and reduction processes to the overall capacity.
Equation (1):
−V1 + V2 + V3 = HOMO(V)
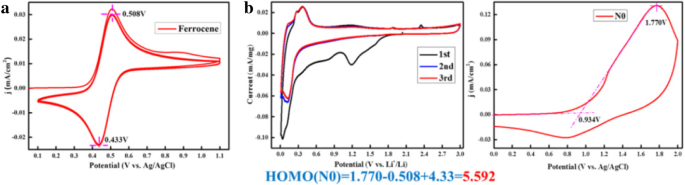
Cyclic voltammograms (CVs) of ferrocene current collector disc vs. silver metal in the electrolyte (a) without additive, and b oxidation energy HOMO values in acetonitrile using tetrabutylammonium perchlorate as electrolyte
The presence of electron‑donating functional groups on the HBC framework results in a uniform electron distribution, enhancing Li+ uptake. Calculated HOMO stabilization energies for the radical anion are 5.592 V, underscoring the material’s readiness for charge transfer.
Figure 7’s inset shows the sequential arrangement and rearrangement of the multilayer structure, revealing optimal d‑spacing for lithium diffusion. TEM images confirm that lithium ions can move both interlayer and intrananosheet, dramatically improving diffusion efficiency. BET analysis indicates a surface area of 81.16 m2 g−1 and a pore volume of 18.647 cm3 g−1, with no hysteresis in the isotherms—evidence of a highly accessible, non‑porous network.

TEM image of nano‑graphene multi‑stage self‑assembly structure
Conclusion
Hexabenzocoronene demonstrates robust structural stability and high electrochemical performance when assembled into dynamic, hierarchical nanostructures. The optimized d‑spacing within the self‑assembled layers promotes efficient lithium‑ion transport, resulting in a high reversible capacity (200 mAh g−1) and excellent cycling stability. These findings establish a clear structure–property relationship between functional‑group chemistry and Li storage capability. Future work will focus on unraveling the precise mechanisms of hierarchical assembly and exploiting these insights to design next‑generation anode materials.
Abbreviations
- CV:
Cycle voltage
- HBC:
Hexabenzocoronene
- HOMO:
Highest occupied molecular orbital
- HRTEM:
High‑resolution transmission electron microscope
- SAED:
Selected area electron diffraction
- SEM:
Scanning electron microscope
- TBAP:
Tetra‑n‑butylammonium perchlorate
- TEM:
Transmission electron microscope
- TLC:
Thin‑layer chromatography
Nanomaterials
- IBM and Warwick Scientists Capture First High‑Resolution Image of Triangulene, a Highly Reactive Triangular Molecule
- Monodisperse Carbon Nanospheres with Hierarchical Porosity Deliver Superior Supercapacitor Performance
- Hybrid Graphene/WO₃ and Graphene/CeOx Electrodes for High‑Performance Supercapacitors
- High-Sensitivity Graphene Oxide Fluorescent Aptasensor for Rapid Detection of CCRF-CEM Leukemia Cells
- From Photovoltaic Si Sludge to High‑Performance Li‑Ion Battery Anodes via Oxygen Diffusion
- Enhancing Supercapacitor Energy Density with Anionic Surfactant‑Intercalated Thermally Reduced Graphene Oxide in Ionic Liquid Electrolytes
- Efficient Solution-Scale Synthesis of Red Phosphorus Nanoparticles for High‑Performance Lithium‑Ion Battery Anodes
- High‑Performance Perovskite Photodetector via Trilayer EA/TiO₂ Architecture
- Hexagonal Boron Arsenide as a Highly Sensitive SO₂ Gas Sensor: A First‑Principles Study
- Optimized Rosin Transfer Technique Minimizes Residue on Graphene Surfaces



