Hexagonal Boron Arsenide as a Highly Sensitive SO₂ Gas Sensor: A First‑Principles Study
Abstract
Using density functional theory (DFT), we examined the adsorption of nine atmospheric gases—N2, O2, CO2, H2O, CO, NO, NO2, NH3, and SO2—on pristine hexagonal boron arsenide (BAs). We evaluated multiple adsorption sites, calculated adsorption energies, charge transfer, and work functions, and analyzed electronic density of states. SO2 displayed the strongest binding (−0.92 eV), shortest equilibrium distance (2.46 Å), and significant charge transfer (−0.179 e). These findings suggest that monolayer BAs could serve as a highly sensitive and selective SO2 sensor.
Introduction
Boron arsenide (BAs) is a III–V semiconductor that combines outstanding photo‑electrical performance, robust mechanical properties, and a sizable band gap. Recent work on 2D III–V materials—including BN, AlN, GaN, GaAs, and BP—has highlighted their potential for detecting biomolecules, pollutants, and gases. 1–17 For example, AlN(0001) has been identified as a powerful catalyst for ammonia synthesis, and GaAs nanowires show strong CO2 and N2 adsorption. 27–29
Despite experimental synthesis and theoretical progress, the gas‑sensing capabilities of BAs remain largely unexplored. In this study, we use first‑principles DFT calculations to systematically assess the adsorption of common atmospheric gases on BAs and identify its suitability as a gas sensor, with a focus on SO2.
Theory and Method of Simulations
The model consists of a 4 × 4 BAs supercell with a single gas molecule adsorbed. Calculations were carried out in DMol3 using the GGA‑PBE exchange‑correlation functional. A 5 × 5 × 1 Monkhorst–Pack k‑point grid and Methfessel–Paxton smearing (0.01 Ry) were employed. Atomic positions were relaxed until the total energy converged to 1 × 10−5 eV and forces were below 0.06 eV/Å. 31–33
The adsorption energy is defined as:
Ead = EBAs+gas − (EBAs + Egas)
Charge transfer was evaluated by Mulliken population analysis.
Result and Discussion
We considered three adsorption sites: atop a boron atom (B), atop an arsenic atom (As), and the center of the hexagonal ring (center). Figure 1a shows the geometry.
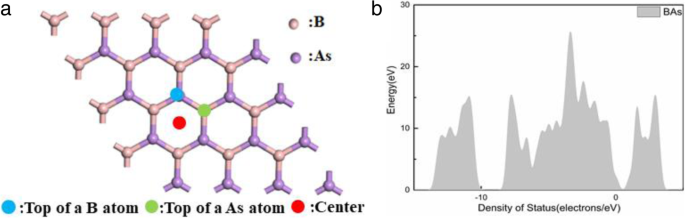
a Schematic of adsorption sites on BAs. b Density of states of pristine BAs.
After geometry optimization, the B–As bond length was 1.967 Å and the indirect band gap was 1.381 eV, consistent with previous reports. 34–35
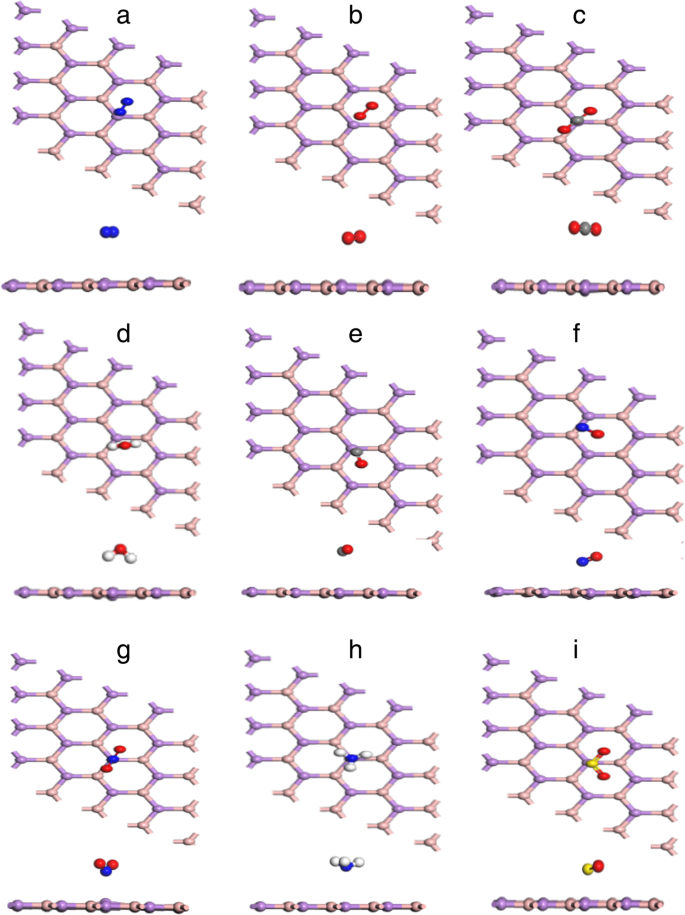
Most stable adsorption configurations for each gas on BAs.
The adsorption energies, charge transfers, and equilibrium distances for all gases are summarized in Table 1.
| Gas | Site | Distance (Å) | Ead (eV) | Charge Transfer (e) |
|---|---|---|---|---|
| N2 | center | 3.65 | −0.24 | +0.014 |
| O2 | center | 2.90 | −0.35 | −0.172 |
| CO2 | top‑As | 3.55 | −0.28 | −0.018 |
| H2O | top‑As | 3.63 | −0.38 | −0.03 |
| CO | top‑As | 3.50 | −0.27 | −0.024 |
| NO | top‑B | 2.86 | −0.18 | +0.01 |
| NO2 | top‑As | 2.47 | −0.43 | −0.09 |
| NH3 | top‑As | 3.27 | −0.34 | +0.007 |
| SO2 | center | 2.46 | −0.92 | −0.179 |
Key observations:
- SO2 shows the strongest adsorption (−0.92 eV) and the shortest binding distance (2.46 Å).
- NO2 has the largest charge transfer (≈−0.09 e) and a pronounced effect on the electronic structure, turning the semiconductor into a metallic state.
- O2 and CO2 exhibit moderate adsorption, while N2 and CO interact weakly.
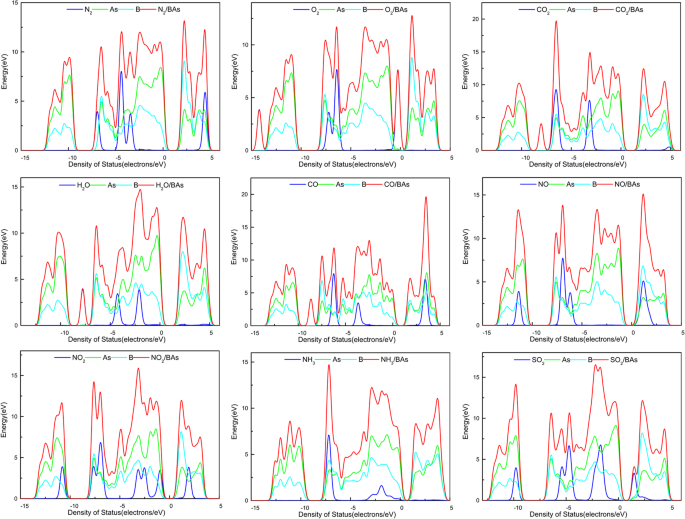
Density of states for each gas‑BAs complex (a–i).
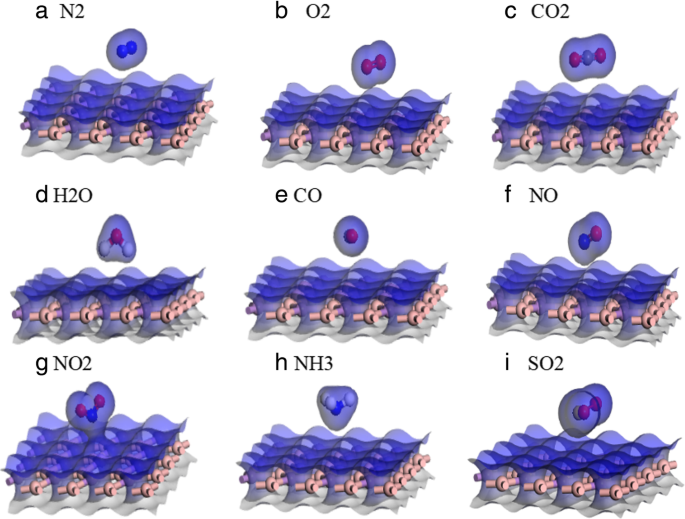
Electron density maps for the nine gas–BAs systems (a–i).
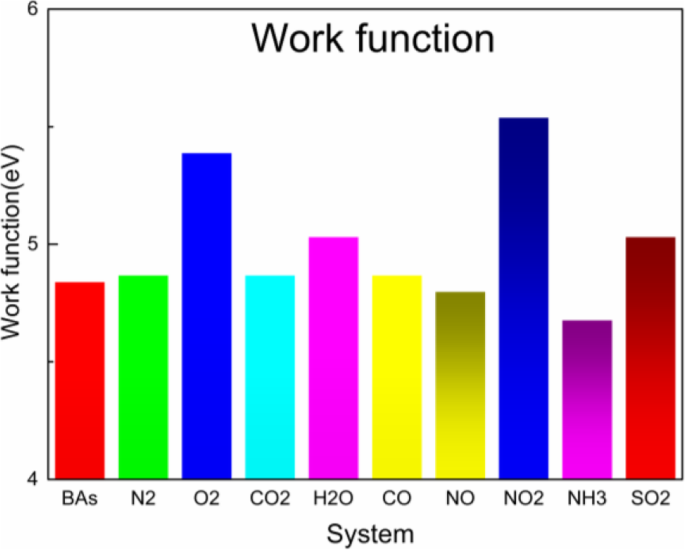
Work function of BAs with each adsorbed gas.
Work‑function analysis further corroborates the strong interaction of SO2 with BAs: the pristine work function is 4.84 eV; adsorption of NO and NH3 reduces it, while O2, NO2, and SO2 increase it. These variations align with the observed charge transfer trends.
Conclusion
Our DFT study demonstrates that hexagonal boron arsenide exhibits exceptional sensitivity to SO2 adsorption, characterized by the most negative adsorption energy, the shortest equilibrium distance, and significant charge transfer. In contrast, other gases show weaker interactions. These properties—combined with measurable changes in electronic conductivity and work function—highlight BAs as a promising, selective platform for SO2 gas sensing.
Abbreviations
- BAs
Hexagonal boron arsenide
- DOS
Density of states
- WF
Work function
Nanomaterials
- IBM and Warwick Scientists Capture First High‑Resolution Image of Triangulene, a Highly Reactive Triangular Molecule
- Ultra‑Narrowband Perfect Absorber with 95% Efficiency for Visible‑Range Plasmonic Sensing
- Penta‑Graphene: A Next‑Generation NOx Gas Sensor with Superior Sensitivity
- Methane Adsorption on MoX₂ (S, Se, Te) Monolayers: Insights from DFT
- Complete Arduino & Intel Edison Starter Kit with Sensors, Webcam, and Essential Accessories
- Choosing the Ideal Gas for MIG Welding: Tips for Strong, Consistent Results
- How to Select the Ideal Grinding Wheel for Your Specific Application
- MQ-3 Gas Sensor: Comprehensive Guide to Alcohol Detection
- Choosing the Ideal Gas for MIG Welding: A Comprehensive Guide
- Choose the Perfect Positioner for Your Welding Application



