Spherical Graphene/SiO₂ Supports Enable Uniform In Situ Polymerization of UHMWPE for High‑Performance Nanocomposites
Abstract
We present a novel spherical support fabricated from reduced graphene oxide (RGO) and SiO₂ that serves as a durable scaffold for Ziegler–Natta catalysts. The support exhibits a porous, sandwich‑like architecture that preserves graphene’s intrinsic properties while providing extensive surface area for catalyst anchoring. During ethylene polymerization, active sites distributed across both the outer surface and internal pores drive orderly chain growth, ensuring a homogeneous dispersion of graphene within the polyethylene matrix. Compared with conventional approaches, this spherical graphene/SiO₂ support markedly enhances nanocomposite performance and retains graphene’s intrinsic characteristics.
Background
Graphene, a single layer of carbon atoms arranged in a honeycomb lattice, has emerged as a leading material in advanced composites due to its exceptional mechanical strength, stiffness, flexibility, and cost‑effectiveness relative to carbon nanotubes. When incorporated into polymer matrices, graphene imparts not only superior mechanical and barrier properties but also functional attributes such as electrical and thermal conductivity. Successful graphene‑reinforced composites have been demonstrated in a wide range of polymers, including polyaniline, polystyrene, poly(vinylidene fluoride), polyurethane, epoxy, and polyethylene. However, achieving uniform dispersion of graphene in non‑polar polymers such as polyethylene (PE) or polypropylene (PP) remains a formidable challenge because graphene is chemically inert and poorly soluble in most solvents.
Solution mixing and melt mixing are the most common routes for fabricating polymer/graphene composites. While effective for many systems, both methods often fail to disperse graphene uniformly, leading to agglomeration and sub‑optimal properties. Graphene’s lack of functional groups hampers its interaction with the polymer matrix, and the high surface energy of GO can cause restacking during processing. Consequently, the pursuit of an effective filler that preserves graphene’s structure while enabling uniform dispersion is of great interest.
Graphene oxide (GO) has been explored as a catalyst support for in situ polymerization of polyolefins. GO’s abundant oxygen‑containing groups allow it to anchor catalytic species, facilitating polymer growth directly from its surface. However, the interaction between GO and catalytic components can introduce additional functional groups, leading to disorder and defect formation that compromise graphene’s mechanical, electrical, and thermal properties. Traditional catalyst supports often fail to provide a regular morphology, especially for spherical shapes, which are critical for scalable olefin polymerization processes.
To address these issues, we developed a micron‑scale spherical RGO/SiO₂ nanocomposite that serves as a high‑performance support for Ziegler–Natta catalysts. The composite features a 3D porous, sandwich‑like architecture that preserves graphene’s structure while providing a robust scaffold for catalyst loading. This design minimizes defect generation during in situ polymerization, ensures uniform graphene dispersion in the polyethylene matrix, and yields a round, granule‑like UHMWPE/graphene nanocomposite with superior mechanical and electrical properties.
Characterizations
X‑ray diffraction (XRD) patterns were recorded on a Rigaku D/Max‑2400 diffractometer using Cu Kα radiation. Raman spectra of GO, BuMgCl/GO, RGO, GO‑SiO₂, RGO‑SiO₂, BuMgCl/GO‑SiO₂, and BuMgCl/RGO‑SiO₂ were obtained with a Horiba Jobin Yvon LABRAM‑HR800, covering 0–4000 cm⁻¹. Fourier transform infrared (FT‑IR) spectra were acquired on a Nicolet NEXUS 670, with samples pressed into KBr tablets. Transmission electron microscopy (TEM) images were captured using a Hitachi H‑600 microscope. Thermogravimetric analysis (TGA) was performed on a Perkin–Elmer diamond analyzer from room temperature to 600 °C at 10 °C min⁻¹ under N₂. Scanning electron microscopy (SEM) coupled with energy dispersive X‑ray (EDX) analysis (Hitachi S‑4800) examined fracture surfaces and composition. Differential scanning calorimetry (DSC) measured thermal transitions on a Perkin‑Elmer Pyris diamond analyzer under N₂ flow. Molecular weights were determined using a Ubbelohde viscometer and the Mark–Houwink equation.
Results and Discussion
Morphology Evolution
Transmission electron microscopy revealed the progression from GO to GO‑SiO₂. Figure 1a displays the layered GO sheets; Figure 1b shows the spherical SiO₂ nanoparticles that tend to agglomerate due to high surface energy. In Figure 1c–d, SiO₂ is encapsulated within GO sheets, forming a dense sandwich structure. The formation mechanism is illustrated in Figure 2, where GO’s oxygen‑rich functional groups facilitate strong interactions with SiO₂, leading to a tightly wrapped composite that preserves electrical conductivity and mechanical flexibility.

TEM images for a GO, b SiO₂, and c, d GO‑SiO₂ hybrid
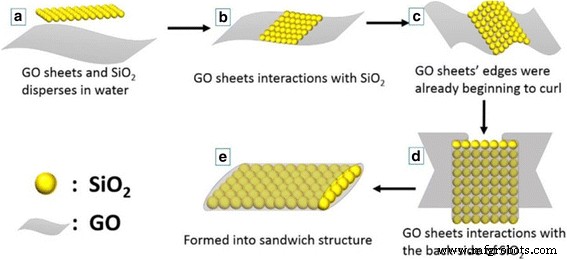
Schematic of the suggested mechanism for GO‑SiO₂ sandwich structure synthesis
SEM images in Figure 3a show that the RGO‑SiO₂ supports possess a narrow size distribution, regular spherical morphology, and high porosity. Statistical analysis (Figure 3b) indicates that 75 % of particles fall within the 30–70 µm range, with an average diameter of 46.8 µm, matching the desired catalyst support size. The spherical particles feature a porous outer layer (Figure 3c–d) and an internal network with an average pore size of 2.23 µm (Figure 3f), distinct from conventional silica supports.
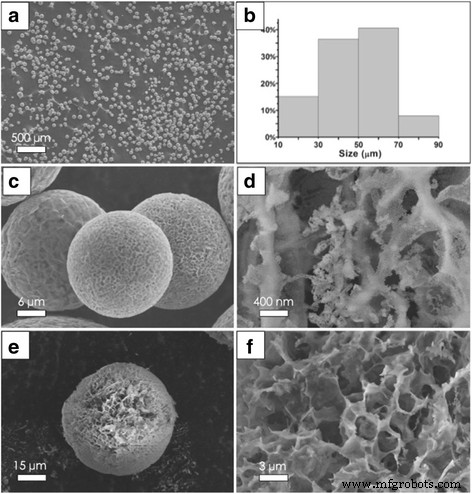
SEM images for a, c RGO‑SiO₂ spherical supports in different magnification. b Histograms of size distribution of a. d Surface magnification of c. e A broken sphere. f Magnification of e
Figure 4 illustrates how varying the SiO₂ content influences the spherical morphology. A 5:1 SiO₂/GO ratio maintains near‑spherical shape with minor surface pores, whereas lower ratios introduce thin flaps and reduced structural integrity. Supports devoid of SiO₂ exhibit a balloon‑like softness, underscoring SiO₂’s role in enhancing mechanical strength and maintaining spherical geometry.
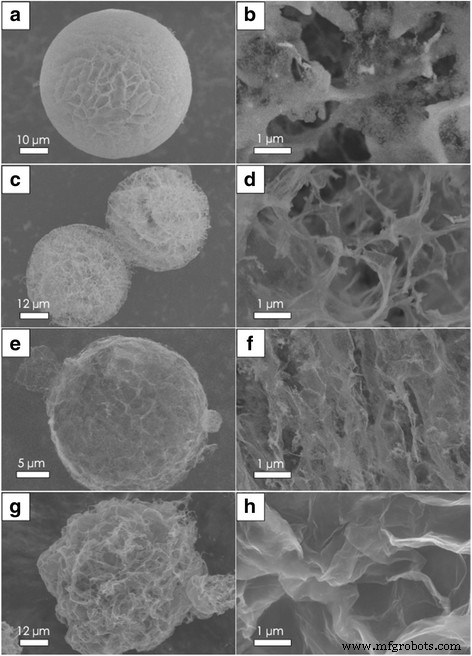
SEM images for RGO‑SiO₂ spherical supports in different SiO₂ content. a, b Quality ratio of SiO₂ and GO is 5:1, and magnification of a; c, d 1:1; and e, f 0.5:1. g, h Without SiO₂
SEM analysis of the catalyst–loaded supports (Figure 5) demonstrates that the spherical porous structure is largely preserved after impregnation with BuMgCl and TiCl₄. EDX spectra confirm the presence of Mg, Cl, Ti, and Si, indicating successful loading of the Ziegler–Natta catalytic components within both the surface and internal pores of the support.
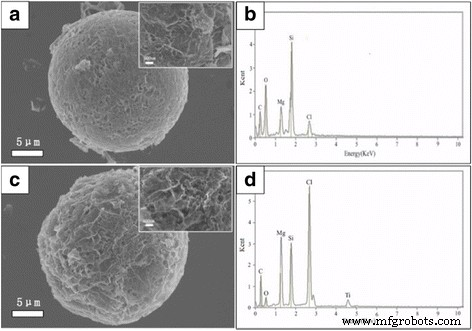
a SEM image of BuMgCl/RGO‑SiO₂, b the corresponding EDX spectrum of BuMgCl/RGO‑SiO₂, c SEM image of TiCl₄/(BuMgCl/RGO‑SiO₂) catalyst, and d EDS spectrum of TiCl₄/(BuMgCl/ RGO‑SiO₂) at an area indicated with a white line in part c
Defects and Disorder of Graphene in Supports
Raman spectroscopy (Figure 6) reveals the degree of disorder in the graphene layers. The D band (≈1350 cm⁻¹) and G band (≈1600 cm⁻¹) intensities change markedly upon interaction with BuMgCl. GO alone shows a high D/G ratio, indicative of defects. After thermal reduction to RGO, the D/G ratio improves, reflecting partial restoration of the sp² network. Importantly, RGO‑SiO₂ maintains a higher D/G ratio after BuMgCl treatment compared to GO‑SiO₂, suggesting that the sandwich architecture protects the graphene layers from excessive functionalization and preserves their crystalline order.
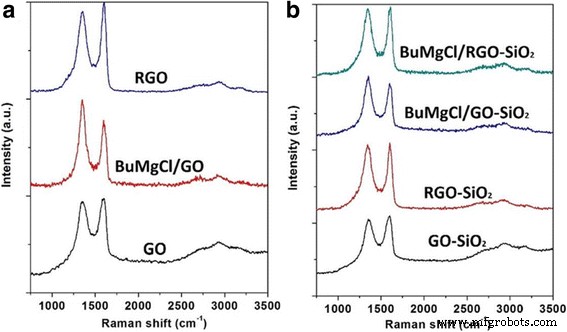
Raman spectra of GO, BuMgCl/GO, RGO, GO‑SiO₂, RGO‑SiO₂, BuMgCl/GO‑SiO₂, and BuMgCl/RGO‑SiO₂
In situ UHMWPE Polymerization with RGO‑SiO₂ Supported Ziegler–Natta Catalyst
Ethylene polymerization using the TiCl₄/(BuMgCl/RGO‑SiO₂) catalyst yielded activities ranging from 2.66 × 10⁵ to 4.2 × 10⁵ g PE mol⁻¹ Ti h⁻¹, with higher TEA/Ti ratios producing greater activity. DSC analysis confirmed a consistent melting temperature of ~142 °C for the resulting UHMWPE. The polymer’s viscosity‑average molecular weight decreased from 3.1 × 10⁶ to 2.0 × 10⁶ g mol⁻¹ as TEA content increased, satisfying UHMWPE specifications.
SEM images (Figure 7) show that the UHMWPE/RGO‑SiO₂ granules retain a uniform spherical morphology, with an average diameter of 150 µm—approximately three times the support size—reflecting the morphological replication of the catalyst support. The surface is densely covered with graphene sheets, indicating successful dispersion of RGO‑SiO₂ within the polymer matrix.
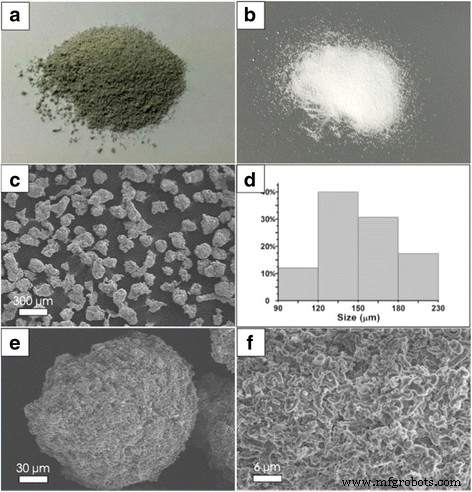
a UHMWPE/RGO‑SiO₂ composite powder as obtained after polymerization. b UHMWPE powder without RGO‑SiO₂. d Histograms of size distribution of c. e SEM images obtained from fracture surface of composites in different magnification. f Surface magnification of e
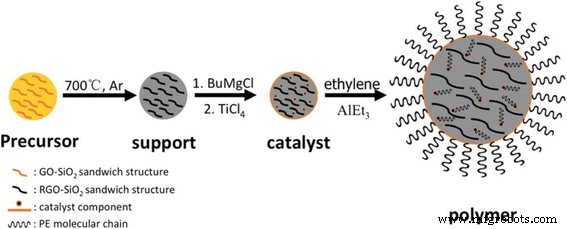
Schematic of nanocomposite synthesis
Electrical Conductivity
Melt‑compressed UHMWPE nanocomposites exhibit notable electrical conductivity. At 0.5 wt % GO loading, the conductivity reaches 6.46 × 10⁻⁴ S cm⁻¹, decreasing to 4.62 × 10⁻⁵ S cm⁻¹ at 0.28 wt %. Compared with direct graphene addition, the spherical support approach requires lower graphene content to achieve comparable conductivity because the internal graphene network remains intact and forms an efficient percolation pathway within the polymer matrix.
Conclusions
We have engineered a micron‑scale RGO/SiO₂ spherical support with a 3D porous sandwich architecture that serves as an efficient scaffold for Ziegler–Natta catalysts. This design minimizes defect formation during in situ polymerization, yields a uniform distribution of active sites on both the surface and internal pores, and reproduces the spherical morphology in the resulting UHMWPE/graphene nanocomposite. The approach offers a scalable route for producing high‑performance, electrically conductive UHMWPE composites with retained graphene properties. Beyond polymer composites, the RGO‑SiO₂ supports hold promise as catalysts, energy‑storage materials, and components in nanoelectronics and photo‑electronic devices.
Abbreviations
- (Z-N) catalyst:
Ziegler‑Natta catalyst
- CNT:
Carbon nanotube
- PE:
Polyethylene
- PP:
Polypropylene
- RGO/SiO₂:
Reduced graphene oxide/SiO₂
- TEA:
Triethyl aluminum
- UHMWPE:
Ultrahigh molecular weight polyethylene
Nanomaterials
- Graphene‑Based Loudspeakers and Earphones: Ultra‑Low‑Power, High‑Fidelity Sound
- Graphene‑Polymer Composites for High‑Performance Supercapacitors: A Comprehensive Review
- Titanate Nanotube-Enhanced Graphene Oxide Nanocomposites: Boosting Flame Retardancy and Photodegradation in Flexible PVC
- Tailoring Graphene Work Function via Size, Functionalization, Defects, and Doping: A First‑Principles Investigation
- Hybrid Graphene/WO₃ and Graphene/CeOx Electrodes for High‑Performance Supercapacitors
- Sodium Alginate–Sulfonated Graphene Oxide Membrane: Superior Proton Conductivity and Low Methanol Permeability for Direct Methanol Fuel Cells
- Enhanced Gas Separation Using Covalently Functionalized Graphene Oxide in PIM‑1 Mixed‑Matrix Thin‑Film Composite Membranes
- Oxidant‑Free Hydrothermal Anchoring of Ultrathin PEDOT on Reduced Graphene Oxide for High‑Performance Flexible Supercapacitors
- Synthesis, Structure, and Superior Capacitance of Sulfur‑Nitrogen Co‑Doped Graphene Quantum Dots
- Engineering High-Quality Graphene/TMD Heterostructures: Fabrication and Spectral Analysis



