Mechanism of Helical Nanofiber Formation via Co‑Electrospinning of Cellulose Acetate and Thermoplastic Polyurethane
Abstract
To investigate how cellulose acetate (CA) and thermoplastic polyurethane (TPU) combine to produce helical nanofibers, we systematically varied the electrospinning parameters and analysed the interfacial interactions that drive fibre morphology. Our experiments show that a CA solution of 14 wt % in a DMAc/acetone (1:2) mixture paired with a TPU solution of 18 wt % in DMAc/acetone (3:1) – referred to as TPU2 – consistently yields well‑defined helical fibres when processed by co‑electrospinning. By characterising the blend with differential scanning calorimetry (DSC) and Fourier‑transform infrared spectroscopy (FTIR), we demonstrate that the observed morphology stems from a combination of enhanced solution conductivity (due to LiCl addition), favourable interfacial tension, and hydrogen‑bond‑mediated miscibility between CA and TPU. This study clarifies the mechanistic role of polymer structure and intrinsic properties in the formation of CA/TPU helical nanofibres, and expands the material toolbox for advanced helical fibre applications.
Background
Helical nanostructures are prized for their high surface‑to‑volume ratio and intrinsic porosity, making them attractive for sensors, filtration media, oil sorbents, and photovoltaic devices. The addition of a helical geometry to micro‑ or nanofibres enhances mechanical resilience and creates a three‑dimensional, porous mat that can be tailored for specific performance requirements. Natural examples – from plant tendrils to wool fibres – illustrate how differential shrinkage or extension during growth can produce tight helices, a principle that has inspired synthetic fabrication strategies.
Co‑electrospinning has emerged as a simple yet powerful route to generate composite fibres with complex morphologies. Unlike techniques such as chemical vapour deposition, sol‑gel processing, or hydrothermal synthesis, co‑electrospinning allows two polymers to be spun simultaneously, yielding core‑shell or side‑by‑side architectures that can evolve into helical shapes. Prior work has demonstrated helical nanofibres from combinations such as PAN/TPU, Nomex/TPU, and PU/Nomex, highlighting the importance of polymer stiffness, miscibility, and hydrogen bonding in driving curvature.
While previous studies have shown that Nomex/TPU can produce fine helices, the non‑hydrophilic nature of Nomex limits its applicability in biomedical or adsorption contexts. Our work therefore focuses on a CA/TPU system, where CA provides rigidity and TPU offers elastomeric flexibility, to explore how solution chemistry and interfacial forces govern helix formation.
Experimental
Materials
Cellulose acetate (CA, Mw ≈ 100 kDa) was purchased from Acros Organics. Thermoplastic polyurethane (TPU, Desmopan DP 2590A) was obtained from Bayer Materials Science. Solvents – N,N‑dimethylacetamide (DMAc), acetone, tetrahydrofuran (THF) – and lithium chloride (LiCl) were supplied by Shanghai Chemical Reagents Co., Ltd. All reagents were used as received. Experiments were conducted at ~25 °C and 40–50 % RH.
Co‑electrospinning
CA solutions of 14 wt % were prepared by dissolving CA and LiCl in DMAc/acetone (1:2) and stirring for 5 h at room temperature. TPU solutions (18 wt %) were dissolved in either DMAc/THF (3:1; TPU1) or DMAc/acetone (3:1; TPU2). A custom off‑centered co‑electrospinning setup (Figure 1) used syringes and pumps to deliver core and shell solutions to a high‑voltage supply (15–20 kV). The collector rotated at 14.24 cm s⁻¹. The core‑shell configuration mimics plant tendril formation, where the core component contracts more rapidly than the shell, driving helix curvature.
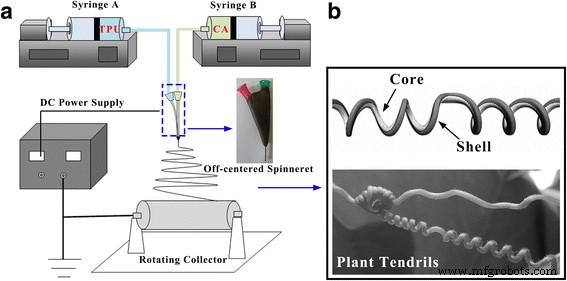
a Schematic of the off‑centered co‑electrospinning system. b Formation mechanism of helical nanofibres.
Characterisations
Fiber Morphology
Resultant fibres were examined by SEM (JSM‑5600LV) after gold sputtering.
DSC
Glass transition temperatures were measured on a DSC‑4000 under nitrogen (scan rate 10 °C min⁻¹, −80 → 300 °C). Samples (5–10 mg) were rapidly cooled to −80 °C after the first melt scan.
FTIR
Infrared spectra were recorded on a Bruker Vector 33 FTIR spectrometer (32 scans, 1 cm⁻¹ resolution). Films were thin enough to satisfy the Beer‑Lambert law. Temperature‑dependent spectra were obtained using a temperature‑controlled cell.
Experimental Results
We first optimized single‑spinning conditions for CA and TPU, then explored two co‑electrospinning systems: CA/TPU1 and CA/TPU2. Figures 2 and 3 illustrate the impact of solution concentration and solvent composition on fibre morphology.
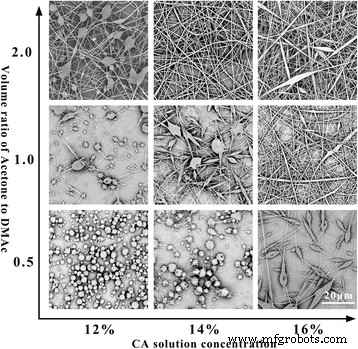
SEM images of CA single‑spinning at varying concentrations (x‑axis) and DMAc/acetone ratios (y‑axis). Applied voltage: 15 kV; working distance: 10 cm; flow rate: 0.15 ml h⁻¹.

SEM of single‑spinning TPU1 (a) and TPU2 (b) at 18 wt % (15 kV, 10 cm, 0.15 ml h⁻¹). TPU2 shows reduced fibre adhesion relative to TPU1.
Co‑electrospinning experiments varied LiCl content in the CA shell and employed either TPU1 or TPU2 as the core. Figure 4 shows that without LiCl, no helices form. At 0.5–1 wt % LiCl, CA/TPU2 fibres begin to bend, while CA/TPU1 remains largely straight or forms bundles. Increasing LiCl to 2 wt % produces many helices in CA/TPU2 but compromises fibre uniformity due to excessive conductivity.
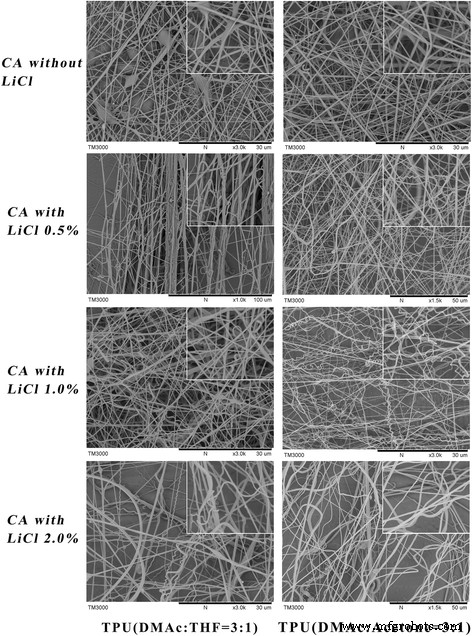
SEM of CA/TPU1 and CA/TPU2 at 0–2 wt % LiCl (20 kV, 15 cm, 0.15 ml h⁻¹).
Across all trials, CA/TPU2 consistently produced a higher density of helices than CA/TPU1, underscoring the importance of solvent compatibility and interfacial tension.
Results and Discussion
Our analysis focuses on three interrelated factors: solution chemistry, hydrogen‑bonding, and miscibility. Together, they explain why the CA/TPU2 system favours helical formation.
Mechanism of CA/TPU Helical Fibres
LiCl plays a dual role. It increases solution conductivity, enhancing jet stability, and it interacts with CA chains via chloride‑hydrogen bonding, aligning the rigid CA backbone and increasing its stiffness relative to TPU. This differential stiffness generates longitudinal stress during jet elongation, promoting a coiled morphology. Figure 5 illustrates the proposed LiCl‑mediated dissolution of CA in DMAc.
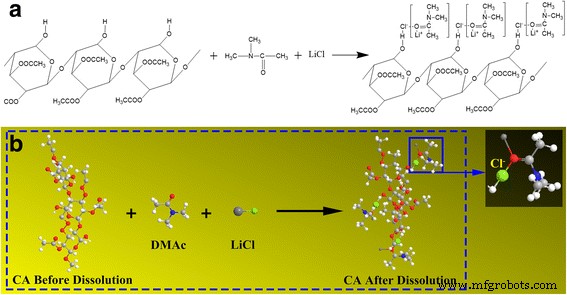
Proposed mechanism for CA dissolution in DMAc/LiCl: (a) molecular formula; (b) 3‑D structure.
Solution Properties
Co‑electrospinning demands a delicate balance of viscosity, vapor pressure, and interfacial tension. TPU2’s lower surface tension (25.34 N m⁻¹) and compatible solvent mix with CA reduce interfacial tension, preventing fibre collapse and enabling smooth core‑shell integration. In contrast, TPU1’s higher surface tension (34.45 N m⁻¹) and solvent mismatch with CA lead to fibre adhesion and poor helix formation.
Hydrogen Bonding in Blends
FTIR spectra (Figure 6) reveal a pronounced 3400 cm⁻¹ band in the CA/TPU blend, indicative of –NH···O hydrogen bonds between TPU and CA. This interaction not only stabilises the core‑shell interface but also enhances viscous drag during jet stretching, encouraging the two components to intertwine into helices.
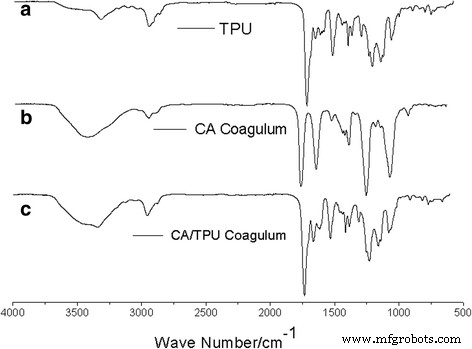
FTIR of pure CA, TPU, and CA/TPU blends: (a) TPU; (b) CA; (c) CA/TPU.
Miscibility Behaviour
DSC thermograms (Figure 8) show two distinct glass‑transition temperatures for the CA/TPU blend, positioned between the individual polymers’ Tg values (−31.24 °C for TPU, 193.74 °C for CA). This intermediate behaviour confirms partial miscibility, which amplifies interfacial stresses when the core and shell experience differential shrinkage, further driving helix formation.
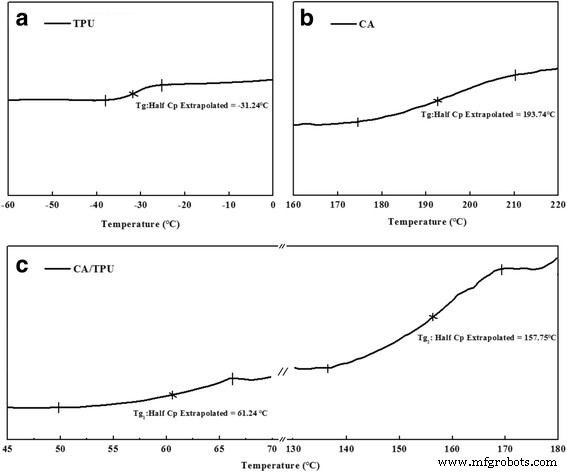
DSC of TPU, CA, and CA/TPU blends: (a) TPU; (b) CA; (c) CA/TPU.
Conclusions
The CA/TPU2 co‑electrospinning system, leveraging solvent‑matched interfacial tension and LiCl‑mediated conductivity, reliably produces helical nanofibres. Enhanced hydrogen bonding and partial miscibility between CA and TPU generate strong interfacial stresses, while the large stiffness differential promotes curvature. These findings elucidate the mechanistic underpinnings of CA/TPU helix formation and broaden the range of materials suitable for advanced helical fibre applications.
Abbreviations
- CA
Cellulose acetate
- DMAc
N,N‑dimethylacetamide
- DSC
Differential scanning calorimetry
- HSPET
Poly(ethylene glycol terephthalate)
- LiCl
Lithium chloride
- Nomex
Poly(m‑phenylene isophthalamide)
- PAN
Polyacrylonitrile
- PS
Polystyrene
- PTT
Poly(ethylene propanediol terephthalate)
- PU
Polyurethane
- THF
Tetrahydrofuran
- TPU
Thermoplastic polyurethane
- TPU1
TPU dissolved in DMAc/THF (3:1)
- TPU2
TPU dissolved in DMAc/acetone (3:1)
- FTIR
Fourier transform infrared spectroscopy
Nanomaterials
- Mesh Analysis Explained: Procedure, Examples, and Applications
- Nanofiber & Filament-Based Nanocarriers: Advancing Precision Drug Delivery
- Synthesis and Characterization of Monodisperse ZnO Nano‑Clips via a Simple Polyol Process
- High‑Efficiency Trilayer Phosphorescent OLEDs Without Electrode Modification Layers: Design, Mechanism, and Performance
- Scalable Fabrication of Gate‑Controlled UV Sensors Using Self‑Assembled ZnO Nanowire Networks
- Temperature‑Dependent Evolution of HfO₂/Si Interfaces: Crystallization Mechanisms and Optimal Annealing Conditions
- Synthesis and Enhanced Photocatalytic Degradation of Rhodamine B by Ag3PO4/T‑ZnO Whisker Heterostructures
- Water Jet Cutting: Precision Machining & Fabrication Solutions
- How Metal Fabricators Can Support COVID-19 Efforts: Automation & Safety Solutions
- Understanding LCD Pricing: Cost Drivers, Market Trends, and Future Outlook



