Synthesis and Characterization of Monodisperse ZnO Nano‑Clips via a Simple Polyol Process
Abstract
Monodisperse ZnO nano‑clips were successfully fabricated by a single‑step polyol route using zinc acetate hydrate (Zn(OAc)₂·nH₂O, n < 2) and ethylene glycol (EG) as solvent, without the addition of water or alkali. At 170 °C and a Zn(OAc)₂·nH₂O concentration of 0.01 M, the reaction proceeds via complexation and polymerization, yielding polycrystalline wurtzite ZnO nano‑clips with aggregated nanocrystals (5–15 nm) and a high specific surface area of 88 m²/g. Annealing at 400–600 °C preserves the clip morphology while improving crystallinity and slightly increasing nanocrystal size. Systematic variation of precursor concentration revealed that <0.0125 M favors clip formation, whereas ≥0.015 M leads to spherical or elliptical particles, indicating a competitive balance between complexation and alcoholysis pathways. This study presents the first report of ZnO nano‑clips synthesized via a water‑free polyol process and proposes a plausible growth mechanism based on the interplay of precursor chemistry and reaction conditions.
Background
Zinc oxide (ZnO) is a wide‑band‑gap semiconductor (Eg = 3.37 eV) with a large exciton binding energy (60 meV), making it attractive for photocatalysis, photovoltaics, and optoelectronic devices. Its nanostructured forms—nanospheres, nanorods, nanowires, and nanoflowers—have been extensively explored, and a variety of synthesis routes have been reported. Among them, the polyol method stands out for its ability to generate metal oxides with controlled morphology, owing to the high boiling point, reducing capability, and surfactant‐like behavior of polyols such as EG. Parameters such as solvent, temperature, precursor concentration, and additives critically influence the size and shape of ZnO particles. Prior work has predominantly produced spherical ZnO particles (20–500 nm) using Zn(OAc)₂·2H₂O and EG, but no reports have described the formation of nanoscopic “clip”‑shaped ZnO structures by a simple, additive‑free polyol route. This study fills that gap by demonstrating the synthesis of monodisperse ZnO nano‑clips and elucidating the underlying growth mechanism.
Methods
All reagents were analytical grade and used without further purification. A 0.01 M solution of Zn(OAc)₂·nH₂O (9.2 mg dissolved in 5 mL EG) was heated to 170 °C on a hot plate under magnetic stirring. Turbidity and milky flocculates appeared after 6–7 min, indicating nucleation. After 1–3 h, the mixture was cooled, the precipitate collected by centrifugation, and washed several times (ethanol : deionized water = 4:1) at 2000–3000 rpm. The product was dried at room temperature overnight. Selected samples were annealed at 400 and 600 °C (2 h, 2 °C/min ramp) in air. To probe concentration effects, solutions ranging from 0.005 to 0.2 M were prepared under identical conditions.
Structural characterization was performed using X‑ray diffraction (Rigaku D/max 2000, Cu kα, λ = 1.5405 Å), scanning electron microscopy (SEM, Quanta 50, FEI), and transmission electron microscopy (TEM, Tecnai G2 F20, Philips). Thermal stability was assessed by thermogravimetric/differential thermal analysis (TG‑DTG, STA 409 PC, Netzsch) at 20 °C/min in air. Fourier‑transform infrared spectra (FTIR, PerkinElmer Spectrum) were recorded in the 4000–400 cm⁻¹ range using KBr pellets. Brunauer–Emmett–Teller (BET) surface areas were measured with a Micromeritics TriStar‑3000. UV–visible–NIR absorbance spectra (Shimadzu UV‑3600) were obtained to evaluate optical properties.
Results and Discussion
Morphology of ZnO Nano‑Clips
SEM images (Fig. 1a) reveal that the as‑prepared ZnO adopts a distinctive clip‑like morphology, with widths of 50–100 nm, lengths of 1–3 µm, and diameters of 10–30 nm. TEM and HRTEM (Fig. 1c) confirm that each clip is a polycrystalline aggregate of nanocrystals (3–15 nm). The morphology remains unchanged after annealing at 600 °C (Fig. 1b), underscoring the structural robustness of the nano‑clips. To date, such a morphology has not been reported for ZnO, particularly via a simple polyol route.

SEM images of (a) as‑prepared and (b) 600 °C annealed ZnO nano‑clip samples. TEM images of (c) 400 °C annealed ZnO nano‑clips with HRTEM inset. (d) Sketch of a ZnO nano‑clip.
Structure of ZnO Nano‑Clips
XRD patterns (Fig. 2a) show the dominant wurtzite ZnO phase (JCPDS 36‑1451) with peaks from (100), (002), (101), (102), (110), (103), (112), and (201) planes, confirming the polycrystalline nature observed by HRTEM. Annealing sharpens the diffraction peaks, indicating improved crystallinity. Using the Scherrer equation, average crystallite sizes increase from 11.5 nm (as‑prepared) to 21.0 nm (400 °C) and 24.8 nm (600 °C). TG‑DTG curves (Fig. 2b) display weight losses at 118, 180, and 400 °C, corresponding to acetic acid, EG, and ester decomposition, respectively; only ~7 % loss occurs up to 600 °C, after which the mass stabilizes, confirming complete removal of organics. FTIR spectra (Fig. 2c) corroborate this: as‑prepared samples show bands for Zn–O (400–600 cm⁻¹), OH (≈800 cm⁻¹), C–OH (1020–1090 cm⁻¹), C–O (≈1260 cm⁻¹), and acetate (1587 and 1413 cm⁻¹). The Δ of 174 cm⁻¹ indicates bridging acetate complexes. After 600 °C annealing, only the Zn–O stretch remains, confirming phase purity.
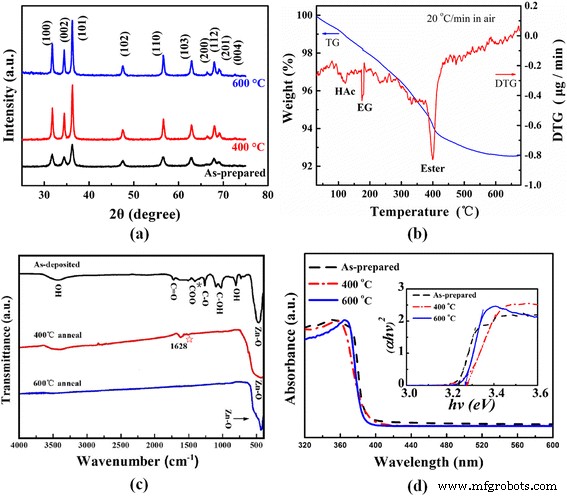
(a) XRD patterns of as‑prepared, 400 and 600 °C annealed ZnO nano‑clip samples. (b) TG‑DTG curves of as‑prepared ZnO nano‑clips heated in air. (c) FTIR spectra of as‑prepared, 400 and 600 °C annealed ZnO nano‑clip samples. (d) UV–visible absorbance spectrum of as‑prepared, 400 and 600 °C annealed ZnO nano‑clip samples. Inset: (αhv)² versus hv.
Optical measurements (Fig. 2d) show a direct band gap of 3.24 eV (as‑prepared), increasing to 3.28 eV after 400 °C annealing and slightly decreasing to 3.27 eV at 600 °C. The initial rise reflects enhanced crystallinity and reduced carbon impurities, while the subsequent decline is attributed to the grain‑size effect. BET analysis indicates a specific surface area of 88 m²/g for the as‑prepared nano‑clips, decreasing to ~59 m²/g after 400 °C annealing due to grain growth and pore collapse.
Effect of Solution Concentration on ZnO Morphology
Systematic variation of Zn(OAc)₂·nH₂O concentration (0.005–0.2 M) revealed a clear morphological transition. At ≤0.0125 M, ZnO nano‑clips dominate (Fig. 3a). At 0.015 M, only elliptical particles (~435 × 200 nm) form, consistent with literature. Higher concentrations (0.05 and 0.2 M) yield a mixture of ellipsoids, spheres, and irregular aggregates, with the reaction becoming faster (turbidity onset drops from 7 min to 4.5 min). These observations support the notion that low precursor concentrations favor complexation and polymerization, whereas higher concentrations promote alcoholysis and polycondensation.

SEM images of ZnO samples under (a) 0.015 M, 5 mL, 170 °C and (b) 0.05 M, 5 mL, 170 °C.
The Possible Growth Mechanism of ZnO Nano‑Clips
Early-stage SEM images (12 min) show feather‑like aggregates and nascent nano‑rings, while 2.5 h images reveal fully formed nano‑clips (Fig. 4). This evolution suggests that Zn(OAc)₂·nH₂O first complexes with EG to form alkoxyacetate species (Eq. 1). Subsequent polymerization yields a line‑shaped template that directs anisotropic growth of ZnO nanowires and rings via slow hydrolysis or thermal decomposition (Eqs. 2–5). After sufficient time, these intermediates assemble into the observed nano‑clip architecture. When the precursor concentration increases, alcoholysis dominates, leading to isotropic nucleation and spherical/elliptical particles.

SEM images of ZnO samples from 0.01 M Zn(OAc)₂·nH₂O at 170 °C with reaction times of (a–c) 12 min and (d–f) 2.5 h. Inset of (c) shows a nano‑ring.
The growth model is illustrated in Fig. 5, contrasting the clip‑forming route with the conventional spherical particle pathway. The competition between complexation/polymerization and alcoholysis/polycondensation determines the final morphology, governed by precursor concentration, reaction temperature, and the intrinsic water content of the Zn source.
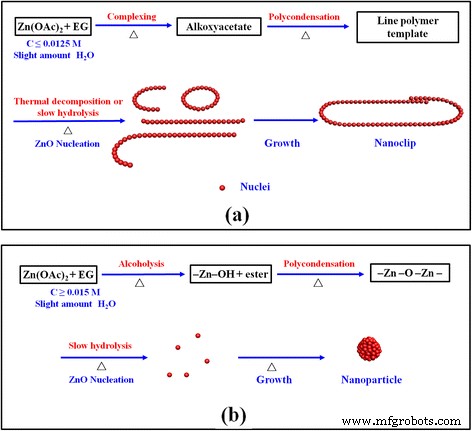
Evolution schematics of (a) ZnO nano‑clip and (b) ZnO particle formation via two polyol‑mediated pathways.
Additional experiments (Supplementary File 1) confirm that adding PVP, increasing water content, or raising temperature shifts the balance toward isotropic growth, underscoring the delicate interplay of reaction parameters.
Conclusions
We have demonstrated that a simple, water‑free polyol route can produce highly monodisperse ZnO nano‑clips with a polycrystalline wurtzite structure. The key is maintaining a Zn(OAc)₂·nH₂O concentration below 0.0125 M and reacting at 170 °C without added alkali or surfactant. Annealing up to 600 °C preserves the clip morphology while enhancing crystallinity and slightly increasing the band gap. A mechanistic model based on the competition between complexation/polymerization and alcoholysis/polycondensation explains the concentration‑dependent morphology. The resulting nano‑clips, with a large surface area (≈88 m²/g) and robust structure, hold promise as photocatalysts for pollutant degradation, anode materials for Li‑ion batteries or supercapacitors, and gas sensors.
Abbreviations
- BET:
Brunauer‑Emmett‑Teller
- DEG:
Diethylene glycol
- EG:
Ethylene glycol
- FTIR:
Fourier‑transform infrared spectra
- FWHM:
Full width at half maximum
- HRTEM:
High‑resolution transmission electron microscopy
- NIR:
Near infrared ray
- PVP:
Polyvinyl pyrrolidone
- SEM:
Scanning electron microscopy
- TEM:
Transmission electron microscopy
- TG‑DTG:
Thermogravimetry‑differential thermal analysis
- UV:
Ultraviolet
- XRD:
X‑ray diffraction
Nanomaterials
- Sintering of Refractory Metal Powders: Processes, Classifications, and Performance Outcomes
- PDMS Fabrication: A Professional Guide for Modern Microfluidic Manufacturing
- Sheet Metal Fabrication: From Design to Delivery – A Complete Process Guide
- Optimizing Emulsion Droplet Size and PVA Surfactant to Enhance Stability of Quantum‑Dot Micellar Nanocrystals
- Solution‑Derived ZnO Nanoshells for Flexible 3D Periodic Structures: A Low‑Cost, Non‑Vacuum Templating Approach
- Atomic-Scale Insights into Slip Deformation and Nanometric Machinability of 6H‑SiC
- Efficient ICP Etching for Black GaAs Nanoarrays: Simple Fabrication & Low Reflectance
- Bronze Casting Explained: Process, Applications, and Industrial Uses
- The Complete Guide to Plastic Processing and Fabrication
- Carbon Fiber Varieties & Production Processes



