Cellular Uptake and Intracellular Trafficking of Nanoparticles: Mechanisms, Physicochemical Drivers, and Clinical Implications
Abstract
Nanoparticle (NP) science is transforming research across disciplines and enabling new technological platforms, particularly in nanomedicine where NPs serve as diagnostic and therapeutic tools. While the benefits are substantial, concerns about short‑ and long‑term human effects persist. This review synthesizes key studies on NP cellular uptake pathways, intracellular trafficking, and the influence of physicochemical properties—size, shape, charge, and surface chemistry—on uptake mechanisms and biological outcomes.
Introduction
Nanoparticles have become central to cutting‑edge research due to their unique properties and vast applicability across fields [1–4]. According to the Global NP Market Outlook 2020, the NP market is projected to grow at a CAGR of 16% between 2015 and 2020, underscoring its expanding influence [5]. In biomedicine, NPs are employed for drug and gene delivery, pathogen detection, protein imaging, tissue engineering, tumour imaging, hyperthermia therapy, and MRI contrast enhancement [6–15].
NPs’ small size enables them to traverse cellular and tissue barriers via endocytosis, yet this very capability raises safety concerns, as NP uptake can influence biodistribution, therapeutic efficacy, and toxicity [16–19]. The pathway of cellular entry dictates NP fate and cytotoxic potential, making it essential to understand endocytic routes and intracellular trafficking for safe nanomedicine design [20–23].
Optimizing NP design—by tuning size, shape, surface charge, and chemistry—can enhance targeting, reduce toxicity, and improve therapeutic outcomes [24]. This review details cellular uptake pathways, intracellular routes, and how NP physicochemical traits shape these processes, providing a foundation for rational NP design in biomedical applications.
Cellular Uptake Pathways of NPs
The plasma membrane (PM) safeguards intracellular components and regulates molecular traffic. Small, non‑polar molecules cross passively, while polar or charged molecules require active transport, predominantly via endocytosis [25–36]. Endocytosis splits into phagocytosis—engulfing large debris by specialized phagocytes—and pinocytosis—engulfing extracellular fluid and dissolved molecules by all cell types [37–49].
Pinocytosis subdivides into clathrin‑mediated, caveolae‑mediated, clathrin‑ and caveolae‑independent, and macropinocytosis pathways [48–49]. Clathrin‑mediated uptake involves ligand‑receptor binding, clathrin coat formation, and vesicle fusion with early endosomes, eventually reaching lysosomes via the endo‑lysosomal pathway [50–60]. Many NP classes, including PLGA, polylactide, PEG‑coated lactide, and silica, preferentially enter cells via clathrin‑mediated routes [61–62].
Caveolae‑mediated endocytosis employs flask‑shaped caveolae (~50–80 nm) rich in caveolin‑1, enabling cargo to bypass lysosomes and avoid degradation—a feature exploited by pathogens and nanomedicines [64–73]. Clathrin‑ and caveolae‑independent routes, used by molecules such as folic acid, enable alternative entry pathways [58–79]. Macropinocytosis allows uptake of large volumes of fluid and micron‑sized particles, active in most cells except brain microvascular endothelium [82–89].
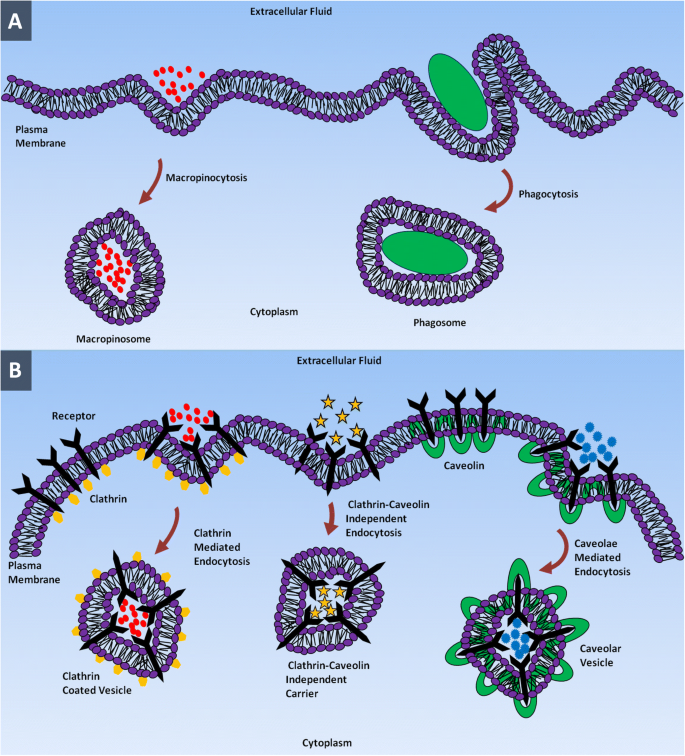
Illustration of NP entry via major endocytic pathways: (a) macropinocytosis and phagocytosis; (b) clathrin‑mediated, clathrin‑/caveolin‑independent, and caveolae‑mediated routes.
Effect of Physicochemical Properties of NP on Cellular Uptake
Size, shape, surface charge, hydrophobicity, and surface chemistry collectively govern NP interaction with the PM, determining uptake efficiency, route, and cytotoxicity [90–91]. Figure 2 visualizes these relationships.
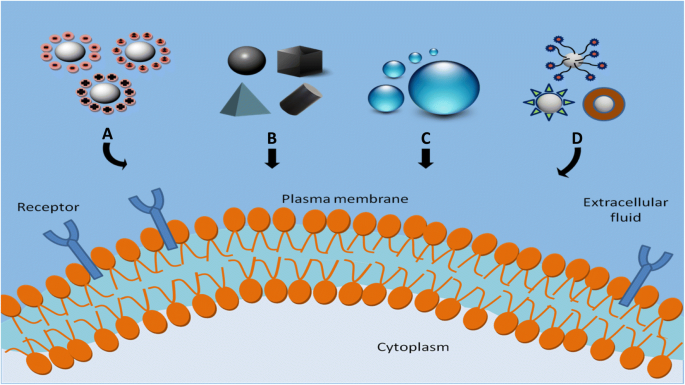
Key physicochemical factors influencing NP uptake: (a) surface charge; (b) shape; (c) size; (d) surface chemistry.
Size
NP size dictates uptake mode and cytotoxic potential. Particles <200 nm favor clathrin‑ or caveolae‑mediated uptake, whereas larger particles (>250 nm) are taken up via phagocytosis or macropinocytosis [47,93]. Optimal uptake often occurs around 50 nm; sizes below 15–30 nm or above 70–240 nm show reduced internalization [94–99]. In drug delivery, larger NPs clear faster via the reticuloendothelial system, reducing circulation time [100–105]. However, experimental inconsistencies arise from aggregation effects and cell‑type variations [106–109].
Shape
Shape strongly influences uptake rates. Spherical NPs consistently exhibit higher internalization than rod‑shaped counterparts across multiple cell lines [112,94,106,116]. Filomicelles—flexible filaments—show prolonged circulation and reduced uptake compared to spheres in vivo [115]. Computational and experimental studies confirm that aspect ratio, rod versus disk or cube geometry, and surface curvature modulate membrane wrapping energy and uptake efficiency [117–119].
Surface Charge
Positively charged NPs interact strongly with the negatively charged PM, enhancing uptake via macropinocytosis and caveolae/clathrin pathways but also increasing membrane disruption and cytotoxicity [47,120–124]. Negatively charged NPs exhibit lower uptake yet may induce membrane gelation; neutral NPs show intermediate behavior [110,125–127]. Surface charge density dictates binding strength and membrane wrapping, as demonstrated by MD simulations [132–133].
Hydrophobicity
Hydrophobic NPs embed within the bilayer core, whereas hydrophilic NPs adsorb on the surface, influencing membrane packing and uptake [138–141].
Surface Modification
Functionalization with PEG, carboxyl, hydroxyl, or amine groups tunes NP charge, stability, and cellular interaction [142]. Amino or carboxyl groups enhance positive or negative charge, respectively, boosting uptake via clathrin‑mediated or clathrin‑independent pathways [143–149]. Functionalized NPs also improve targeting efficiency and reduce toxicity, as shown with polydopamine–aptamer conjugates and folic acid‑coated particles [147–150].
Elasticity
NP stiffness influences membrane wrapping energy. Stiffer particles (high Young’s modulus) are generally internalized more readily than softer ones, though some studies report the opposite or intermediate optimal stiffness [151–156]. Adjusting elasticity can therefore fine‑tune uptake for drug delivery, as demonstrated by enhanced tumor accumulation of nanolipogels with tailored stiffness [157].
Intracellular Trafficking of NPs
Following endocytosis, NPs progress through early endosomes, late endosomes, recycling endosomes, and lysosomes, where degradative enzymes reside [158–166]. Some NPs evade lysosomal degradation by escaping into the cytoplasm or by exploiting autophagic pathways [167–170]. Autophagy, which sequesters cytoplasmic content into autophagosomes for lysosomal degradation, can be induced by certain NPs, affecting cell homeostasis [171–178].
Dynamic imaging of Tat‑conjugated quantum dots revealed macropinocytic uptake, perinuclear trafficking along microtubules, and filopodial release events [179]. Size‑dependent intracellular fate was observed for fluorescent polystyrene particles in hepatocytes, with mitochondrial localization and biliary excretion pathways [180]. Silver NPs aggregated near the nucleus in mesenchymal stem cells, while titania nanotubes crossed nuclear membranes in neural stem cells, illustrating diverse trafficking routes [181–183].
Conclusions
NP applications are expanding across consumer products, raising exposure and safety concerns. A deep understanding of cellular uptake mechanisms and intracellular trafficking—shaped by NP size, shape, charge, hydrophobicity, surface chemistry, and elasticity—is essential to design safe, effective nanomedicines and to mitigate potential risks.
Abbreviations
- ATP:
Adenosine triphosphate
- CAGR:
Compound annual growth rate
- CM:
Cell membrane
- FITC:
Fluorescein isothiocyanate
- GPI:
Glycosylphosphatidylinositol
- LDH:
Layered double hydroxide
- MTOC:
Microtubule‑organizing center
- NP:
Nanoparticle
- PEG:
Polyethylene glycol
Nanomaterials
- FePO4 Nanoparticles: Biocompatible Carriers for Doxorubicin Delivery, RNA Stabilization, and Enhanced Anticancer Activity
- Amphiphilic Hyperbranched Polyglycerol Enables Size‑Controlled, Stable Gold Nanoparticles for Hydrocarbon‑Based Nanofluids
- Polypyrrole‑Coated FePt Nanoparticles: A Multifunctional Platform for Photothermal Therapy and Photoacoustic Imaging
- Green Synthesis of Metal‑Oxide Nanoparticles with Gum Karaya and Their Ecotoxicological Impact on *Chlamydomonas reinhardtii*
- PEG‑PCCL Nanoparticles: Low‑Toxicity, Sustained Paclitaxel Delivery and Enhanced Anti‑Tumor Efficacy in Hepatocarcinoma Models
- Shape-Dependent Cytotoxicity & Cellular Uptake of Green Tea‑Reduced Gold Nanoparticles in Cancer Cells
- Green Synthesis of Gold Nanoparticles with Mimosa tenuiflora Bark Extract: Cytotoxicity, Cellular Uptake and Catalytic Degradation of Methylene Blue
- Green Synthesis of Silver Nanoparticles with Yeast Extract: Controlled Morphology and Enhanced Antibacterial Activity
- Optimizing Fe3O4‑Au Core‑Shell Nanoparticle Uptake for Targeted Endocytosis in Colorectal Cancer Cells
- In‑Situ Synthesis of Silver Nanoparticles on Amino‑Grafted Polyacrylonitrile Fibers for Enhanced Antibacterial Performance



