Targeted Lipid Nanoparticles Functionalized with Transferrin Enhance Paclitaxel Efficacy in Leukemia Cells
Abstract
Leukemia, a hematologic malignancy marked by unchecked proliferation of white blood cells, continues to challenge clinicians with limited therapeutic options and high toxicity from conventional chemotherapy. Here we report the design, synthesis, and biological evaluation of transferrin‑decorated paclitaxel‑loaded solid lipid nanoparticles (TPLN) engineered to exploit transferrin receptor (TfR) over‑expression on HL‑60 leukemia cells. Compared to non‑targeted paclitaxel‑loaded nanoparticles (PLN), TPLN exhibited a markedly higher cytotoxicity (IC50 = 0.45 µg/mL versus 2.8 µg/mL) and induced extensive apoptosis (≈30 % apoptotic cells, 65 % viability). These findings underscore the potential of ligand‑conjugated lipid nanocarriers to improve therapeutic index and pave the way for next‑generation leukemia treatments.
Background
Leukemia is characterized by the aberrant proliferation of immature leukocytes, leading to bone marrow failure and immune dysfunction (1–3). Despite its prevalence, current chemotherapeutics often fall short due to poor bioavailability and off‑target toxicity (4–5). Paclitaxel (PTX) is a potent antimitotic agent used across multiple cancers, yet its systemic side effects and rapid clearance limit clinical utility (7,8). Lipid nanoparticles, particularly solid lipid nanoparticles (SLN), offer a biocompatible platform for sustained drug release and enhanced tumor accumulation via the enhanced permeation and retention (EPR) effect (9–14). However, passive targeting alone may be insufficient; receptor‑mediated strategies can further improve selectivity. Transferrin (Tf) binds TfR, which is markedly over‑expressed on leukemia cells, providing a natural targeting ligand (15–17). Our study integrates Tf onto SLN to generate TPLN, hypothesizing that this modification will elevate cellular uptake, reduce the effective dose, and trigger apoptosis more efficiently than conventional SLN.
Methods
Materials
Compritol 888 ATO, cholesterol, oleic acid, and PTX were sourced from Sigma‑Aldrich. Human Tf was obtained from the same supplier. All reagents were of analytical grade.
Preparation of Paclitaxel‑Loaded Transferrin‑Conjugated Solid Lipid Nanoparticles
Transferrin‑PEG‑oleic acid (Tf‑PEG‑OA) was synthesized via amide coupling (18). Briefly, oleic acid was reacted with NHS/DCC in a 1:2:2 molar ratio for 16 h at room temperature. Separately, NH2‑PEG‑COOH was dissolved in DMSO, activated with TEA, and reacted for 12 h under inert atmosphere. The resulting PEG‑OA was purified by dialysis and lyophilization. The Tf‑PEG‑OA conjugate was then prepared by activating PEG‑OA (1:2:2 NHS/DCC) and coupling to Tf in the presence of TEA for 12 h. The final conjugate was dialyzed (48 h) and lyophilized.
SLN were fabricated using a solvent‑evaporation technique (19). PTX, Compritol, cholesterol, and Tf‑PEG‑OA were dissolved in ethanol/chloroform (1:5) and slowly added to 1 % PVA aqueous phase. The mixture was homogenized at 12,000 rpm (T25 homogenizer, IKA) and sonicated (3 min). After 12 h stirring, the solvent was evaporated, and unencapsulated drug was removed by centrifugal filtration (Amicon Ultra‑4, 3,500 Da MWCO). Entrapment efficiency (EE) and loading efficiency (LE) were quantified by HPLC.
Physical Characterization of Nanoparticles
Particle size and polydispersity were measured by dynamic light scattering (Zetasizer ZS Nano, Malvern). Morphology was assessed by transmission electron microscopy (TEM, JEM‑2000EX, JEOL). Zeta potential was recorded to evaluate surface charge stability.
In Vitro Drug Release Study
Drug release was evaluated by the dialysis method. 1 mL of nanoparticle dispersion was placed in a 3,500 Da MWCO bag and submerged in 10 mL release medium, shaken at 100 rpm. At predetermined intervals, 1 mL samples were collected and analyzed by HPLC (C18 column, 10 %–90 % acetonitrile gradient, 214 nm detection).
Cytotoxicity Assay
MTT assays were performed on HL‑60 cells seeded at 8,000 cells/well in 96‑well plates. After 24 h incubation, cells were treated with free PTX, PLN, or TPLN at varying concentrations for an additional 24 h. MTT (5 mg/mL) was added for 4 h, followed by DMSO solubilization. Absorbance was read at 570 nm.
Cellular Uptake of Nanoparticles
For uptake studies, nanoparticles were loaded with rhodamine B. HL‑60 cells (3×105 per well) were incubated with dye‑laden particles for 3 h, washed, and imaged by confocal laser scanning microscopy (CLSM, Nikon).
Hoechst 33342 Assay
Apoptosis was qualitatively assessed by Hoechst 33342 staining. Treated cells were fixed with 4 % paraformaldehyde, stained, and examined under a fluorescence microscope.
Flow Cytometer‑Based Apoptosis Assay
Quantitative apoptosis was measured using Annexin V/PI staining. Post‑treatment, cells were incubated with Annexin V (2.5 µL) and PI (2.5 µL) for 15 min, diluted to 1 mL, and analyzed by flow cytometry (BD FACS).
Colony Formation Assay
1000 HL‑60 cells/well were seeded in 6‑well plates, treated with PTX‑equivalent doses (0.1 µg/mL), and incubated for 10 days. Colonies were fixed with methanol, stained with hematoxylin, and counted using AlphaEaseFCTM software.
Statistical Analysis
Data were analyzed by two‑tailed Student’s t test or one‑way ANOVA, with significance set at p < 0.05.
Results and Discussion
Physicochemical Characterization of Nanoparticles
PLN and TPLN displayed mean diameters of 140 nm and 160 nm, respectively, as determined by DLS (Fig. 2a). The modest size increase for TPLN reflects Tf conjugation. Both sizes fall within the optimal 100–200 nm window for tumor EPR-mediated accumulation (9,20,21). Zeta potentials were −22.5 ± 1.56 mV, indicating colloidal stability and potential for cellular interaction. TEM images (Fig. 2b) confirmed spherical morphology and uniform dispersion.
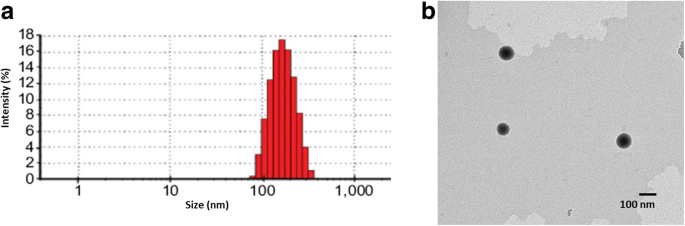
a Particle size distribution (DLS). b TEM micrograph.
Drug Loading and In Vitro Release
PTX loading achieved an EE of 92.5 ± 1.35 % and LE of 8.6 % w/w. Release studies (Fig. 3) showed a sustained profile: ~30 % cumulative release at 24 h and ~65 % at 75 h. TPLN released drug more slowly than PLN, likely due to the bulky Tf layer, which offers a diffusion barrier and may mitigate burst release.
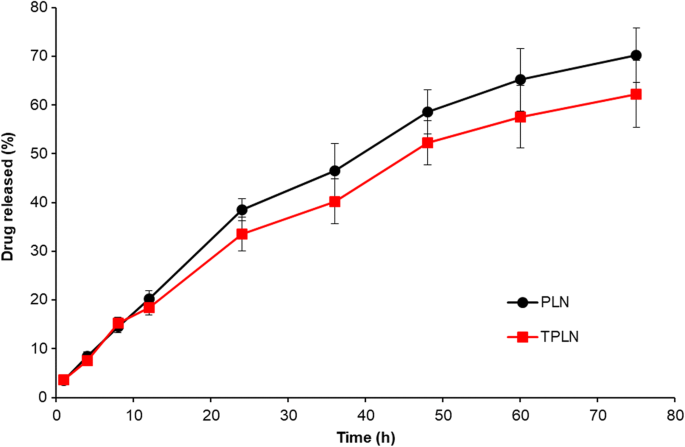
In vitro PTX release from PLN and TPLN (PBS pH 7.4).
Cellular Uptake of the Nanoparticles
CLSM images (Fig. 4) revealed markedly stronger red fluorescence in cells treated with TPLN compared to PLN, confirming Tf‑mediated uptake. The enhanced internalization is consistent with TfR over‑expression on HL‑60 cells (22).
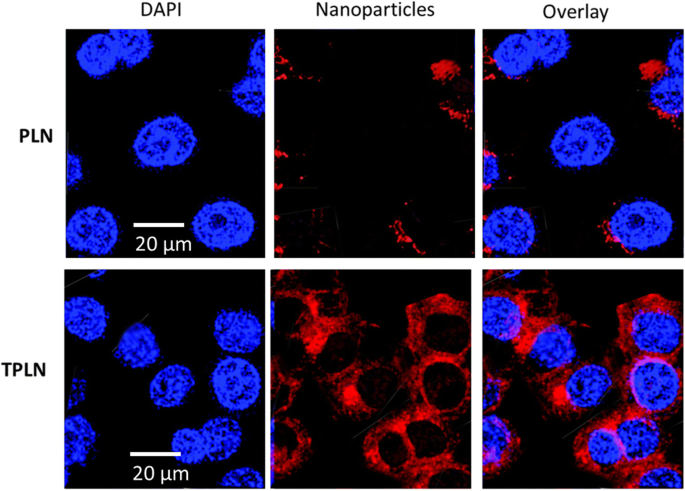
Confocal uptake of rhodamine‑loaded PLN vs. TPLN (scale bar 20 µm).
In Vitro Cytotoxicity Assay
MTT data (Fig. 5) indicated dose‑dependent cytotoxicity for all formulations. TPLN achieved an IC50 of 0.45 µg/mL versus 2.8 µg/mL for PLN, a six‑fold improvement attributable to enhanced cellular delivery and sustained release. The superior efficacy of TPLN aligns with prior reports on Tf‑decorated carriers reducing MDR and systemic toxicity (23).
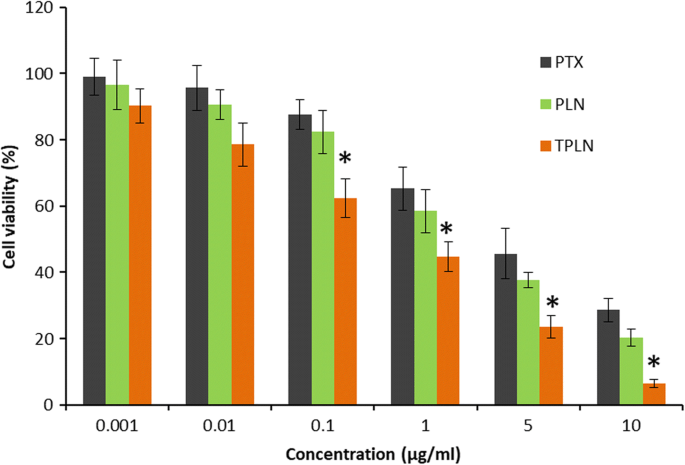
MTT cytotoxicity of free PTX, PLN, and TPLN (n = 6).
Hoechst 33342 Apoptosis Assay
Hoechst staining (Fig. 6) showed pronounced chromatin condensation and apoptotic body formation in TPLN‑treated cells, more evident than PLN or free PTX.
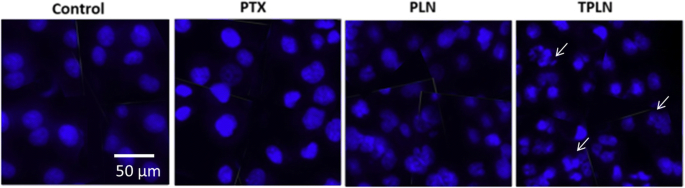
Hoechst 33342‑based apoptosis imaging (scale bar 50 µm).
Flow Cytometer‑Based Apoptosis Assay
Annexin V/PI analysis (Fig. 7) revealed 30 % apoptotic cells and 65 % viable cells for TPLN, compared to 10 % apoptosis for PLN and 90 % viability for free PTX. The significant increase in early/late apoptosis underscores the targeted delivery advantage (24,25).
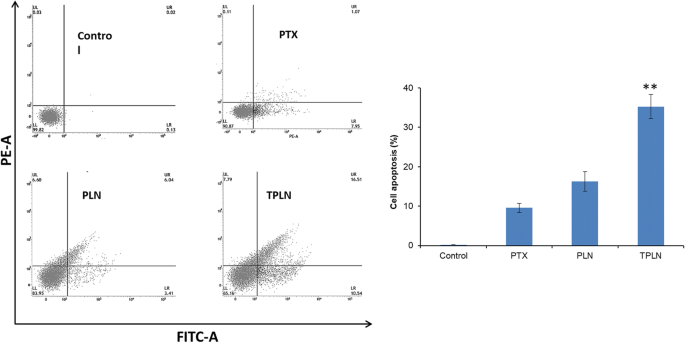
Flow cytometry apoptosis quantification (n = 3).
Colony Formation Assay
Colony suppression (Fig. 8) showed TPLN reduced colony count to ~20 % of control, whereas PTX and PLN achieved ~70 % and ~60 %, respectively. This suggests TPLN effectively targets leukemic progenitor cells, a critical step toward curative therapy.
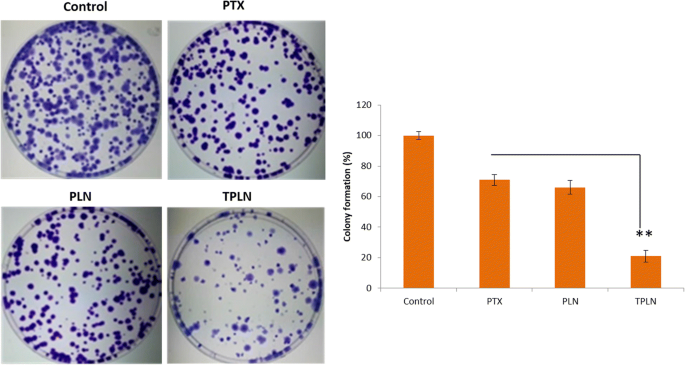
Colony formation after 10‑day treatment (p < 0.001 vs. PTX).
Conclusion
Transferrin‑decorated paclitaxel‑loaded solid lipid nanoparticles (TPLN) exhibit superior cellular uptake, potent cytotoxicity, and robust apoptosis induction in HL‑60 leukemia cells compared to non‑targeted PLN. These results validate TfR‑mediated targeting as a promising strategy to enhance the therapeutic index of PTX and mitigate systemic toxicity. Future studies will explore in vivo biodistribution, pharmacokinetics, and therapeutic efficacy in leukemia animal models.
Abbreviations
- EPR:
Enhanced permeation and retention effect
- OA:
Oleic acid
- PEG:
Polyethylene glycol
- PLN:
Paclitaxel‑loaded lipid nanoparticles
- PTX:
Paclitaxel
- SLN:
Solid lipid nanoparticles
- Tf:
Transferrin
- TPLN:
Transferrin‑conjugated PLN
Nanomaterials
- Using GaN/Fe Nanoparticles to Magnetically Guide Endothelial Cells in Vitro
- Ag Nanoparticle-Enhanced BiV1‑xMoxO4 Composites: Synergistic Photocatalytic Performance under Visible Light
- Gold Nanoparticle–Modified 6‑Mercaptopurine and Rabies‑Derived Peptide Enhance SH‑SY5Y Neural Cell Proliferation and Neurite Outgrowth
- gH625 Peptide vs. Folic Acid: Enhancing Fe3O4 Magnetic Nanoparticle Uptake Across the Blood–Brain Barrier
- Optimizing Perovskite Solar Cell Efficiency with Size‑Controlled Ag Nanoparticles in a TiO₂ Compact Layer
- Lithocholic Acid–Functionalized Gold Nanoparticles Induce Selective Apoptosis in Liver Cancer Cells
- Lanthanum Strontium Manganite Nanoparticles: Airway Epithelial Cell Toxicity and Mucus Secretion Impact
- Polydopamine Core–Shell Nanoparticles with Redox‑Responsive Polymer Shells for Targeted Drug Delivery and Synergistic Chemo‑Photothermal Therapy
- Enhanced Gene Delivery Using Gold Nanoparticles Functionalized with Chitosan, N‑Acylated Chitosan, and Chitosan Oligosaccharide
- Quantitative Study of PLGA Nanoparticle Uptake in Laryngeal Cancer and Immune Cells to Improve Drug Delivery



