Ag Nanoparticle-Enhanced BiV1‑xMoxO4 Composites: Synergistic Photocatalytic Performance under Visible Light
Abstract
In recent years, bismuth vanadate (BiVO₄) has emerged as a leading visible‑light photocatalyst due to its strong absorption and chemical stability. Here, we present Ag‑decorated BiV₁₋ₓMoₓO₄ composites synthesized by a simple hydrothermal route followed by photo‑induced reduction of Ag⁺ under nearly neutral pH. Metallic Ag nanoparticles selectively nucleate on the (040) facet of Mo‑doped BiVO₄. X‑ray diffraction (XRD) and scanning electron microscopy (SEM) confirm the phase purity and surface morphology. Photocatalytic tests, based on rhodamine B (RhB) degradation under visible light (λ > 420 nm), reveal that the Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄ composite delivers the highest activity, achieving complete decolorization within two hours. These findings highlight the synergistic role of Ag and Mo in enhancing charge separation and visible‑light response, advancing BiVO₄‑based photocatalysts for environmental remediation.
Background
As global environmental concerns and energy demands intensify, the development of robust, efficient photocatalysts has become a research priority [1, 2]. Semiconductors such as Bi₂WO₆, BiPO₄, Ag₃PO₄, and BiVO₄ have attracted attention for their roles in organic pollutant degradation and water‑splitting applications [3–13]. Nonetheless, many oxide photocatalysts suffer from narrow visible‑light response windows and rapid electron–hole recombination, limiting their practical utility [14–18].
BiVO₄, with a bandgap of 2.42 eV, offers wide‑bandgap visible‑light absorption, excellent chemical stability, and non‑toxic composition, making it a promising n‑type semiconductor photocatalyst [19–21]. However, intrinsic carrier mobility is limited, leading to high recombination rates that constrain photocatalytic efficiency. Various strategies—including element substitution and metal doping—have been employed to modulate the crystal lattice and enhance charge transport [22–28]. For instance, substituting V⁵⁺ with hexavalent Mo⁶⁺ can reinforce n‑type behavior and improve photo‑oxidation performance [29, 30]. Additionally, facet‑specific deposition of noble metals such as Ag, Cu, and Au has demonstrated significant activity enhancements [31–33]. Li et al. successfully anchored Ag nanoparticles onto the (040) facets of BiVO₄ via hydrothermal synthesis and photoreduction, achieving superior photoelectrochemical performance [34].
Building on this approach, we employed a mild hydrothermal method to synthesize BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄ under weakly alkaline conditions, followed by photoreduction of Ag⁺ to deposit Ag nanoparticles on the (040) facets. The resulting Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄ composites exhibit markedly enhanced photocatalytic degradation of rhodamine B (RhB) under visible‑light irradiation (λ > 420 nm) compared with non‑composite counterparts. This study details the synthesis, characterization, and photocatalytic performance of BiVO₄, Ag/BiVO₄, BiV₁₋ₓMoₓO₄, and Ag/BiV₁₋ₓMoₓO₄ composites.
Experimental
Synthesis of BiVO₄ and BiV₁₋ₓMoₓO₄ Powders
Analytical‑grade bismuth nitrate pentahydrate (Bi(NO₃)₃·5H₂O), ammonium metavanadate (NH₄VO₃), ammonium carbonate, and ammonium molybdate ((NH₄)₂MoO₄) were sourced from Sigma–Aldrich and used without further purification. In a typical synthesis, 3.7 mmol of Bi(NO₃)₃·5H₂O, 3.7 mmol of NH₄VO₃, and 12 mmol of (NH₄)₂CO₃ were dissolved in 75 mL of 1 M HNO₃, stirred for 30 min at room temperature, and the pH adjusted to 8 with 2 M NaOH. The resulting solution was transferred to a 150‑mL Teflon‑lined stainless‑steel autoclave and heated at 180 °C for 12 h under autogenous pressure. After cooling, the precipitate was filtered, washed with distilled water and ethanol, and dried at 60 °C for 12 h.
For Mo‑doped samples, the NH₄VO₃ quantity was partially replaced by ammonium molybdate to achieve nominal 0.5 %, 0.75 %, and 1 % atomic substitution of V. The resulting powders were labeled BiV₀.₉₉₅Mo₀.₀₀₅O₄, BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄, and BiV₀.₉₉₀Mo₀.₀₁O₄, respectively.
Preparation of Ag/BiVO₄ and Ag/BiV₁₋ₓMoₓO₄ Samples
0.50 g of the BiVO₄ powder and 0.05 g of AgNO₃ were dispersed in 100 mL of an ammonium carbonate solution (0.8 g L⁻¹). The mixture was sonicated until a homogeneous suspension formed, then irradiated with a 300 W xenon lamp for 30 min under magnetic stirring. The color change from yellow to gray‑green confirmed the in‑situ reduction of Ag⁺ to metallic Ag. The resulting composites were filtered, washed with deionized water, and dried at 60 °C for 12 h.
Photocatalytic Activity
Photocatalytic performance was evaluated by monitoring RhB degradation under visible light (λ > 420 nm). A 150‑mL aqueous solution containing 10 mg L⁻¹ RhB was stirred with 0.1 g of the photocatalyst in a glass reactor equipped with a cooling water circulation system to maintain room temperature. After 30 min of dark adsorption–desorption equilibrium, the suspension was illuminated for 2 h. Aliquots (5 mL) were withdrawn every 30 min, centrifuged, and the supernatant’s absorbance at 552 nm was recorded.
Characterization Techniques
Morphology was examined by field‑emission scanning electron microscopy (FE‑SEM, S4800) and transmission electron microscopy (TEM, JEM‑2100F). X‑ray photoelectron spectroscopy (XPS, VGESCA‑LAB MKII) quantified elemental composition. Crystalline phases were identified via X‑ray diffraction (XRD, Bruker D8) using Cu Kα radiation. Inductively coupled plasma (ICP) measured elemental content. UV–vis diffuse reflectance spectra (Shimadzu UV‑2450) were collected over 360–800 nm to estimate bandgaps using the Kubelka–Munk method.
Results and Discussion
XRD patterns (Fig. 1a) confirm that the as‑prepared BiVO₄ adopts a monoclinic scheelite structure (JCPDS 14‑0688). Mo doping does not alter the phase but induces peak splitting at 18.7° and 30.5°, corresponding to (110) and (040) facets. The emergence of a weak (111) Ag reflection at 38.1° in Ag‑decorated samples verifies successful photoreduction of Ag⁺ to metallic Ag nanoparticles, though the low Ag content limits XRD intensity.
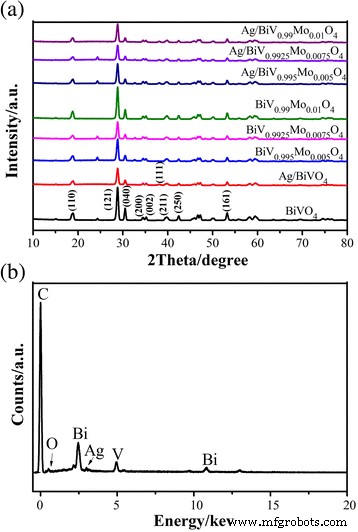
a XRD patterns of pure BiVO₄, Ag/BiVO₄, BiV₁₋ₓMoₓO₄, and Ag/BiV₁₋ₓMoₓO₄. b Corresponding EDX mapping of Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄.
EDX analysis (Fig. 2) confirms uniform distribution of Bi, V, O, Mo, and Ag across the composite. ICP quantification reveals an actual Mo content of 0.16 % in Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄, lower than the nominal 0.75 % due to precursor loss during hydrothermal processing, a phenomenon reported in prior studies [35, 36].
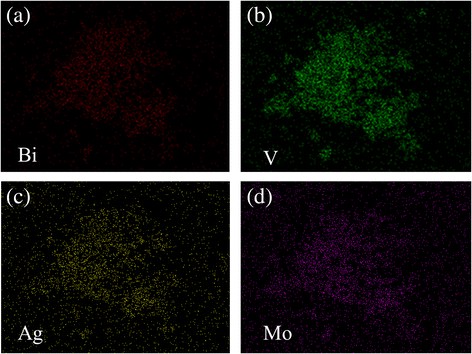
a–d Elemental mapping of Bi, V, Ag, and Mo in Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄.
SEM micrographs (Fig. 3) reveal that pristine BiVO₄ exhibits a layered morphology with clustered platelets. Ag/BiVO₄ shows Ag nanoparticles densely distributed on the (040) facet, corroborating XRD findings. The Mo‑doped composite displays uniformly sized Ag nanoparticles on the (040) surface, which is known for its high charge‑carrier mobility [37], thereby favoring photocatalytic activity.

SEM images: a, b low‑ and high‑magnification of pure BiVO₄; c, d low‑ and high‑magnification of Ag/BiVO₄; e, f low‑ and high‑magnification of Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄.

TEM images: a pure BiVO₄, c Ag/BiVO₄, e Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄; b, d, f high‑magnification of the respective samples.
XPS spectra (Fig. 5) confirm the presence of Bi³⁺ (Bi 4f = 158.94/164.27 eV), V⁵⁺ (V 2p = 516.5/524.1 eV), Mo⁶⁺ (Mo 3d = 231.7/234.9 eV), O 1s (529.71 eV), and metallic Ag (Ag 3d = 367.98/374.0 eV). The Ag content, 6.6 % by XPS, aligns with ICP data.
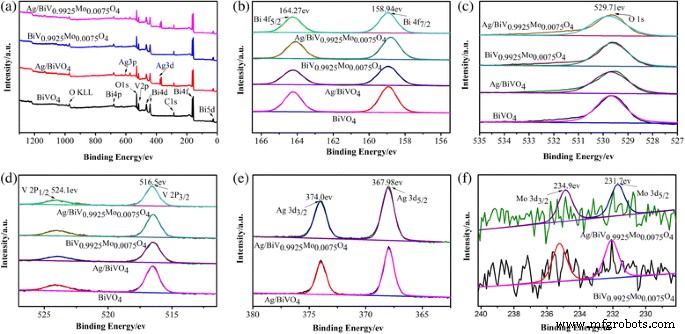
XPS spectra: a survey; b Bi 4f; c O 1s; d V 2p; e Ag 3d; f Mo 3d.
Diffuse‑reflectance UV–vis spectra (Fig. 6a) show that Mo doping narrows the bandgap relative to pure BiVO₄ (≈2.3 eV). All Ag‑decorated samples exhibit enhanced visible‑light absorption, with Ag/BiVO₄ displaying the strongest response. Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄ shows intermediate absorption, suggesting that Mo slightly moderates the Ag‑induced plasmonic effect.
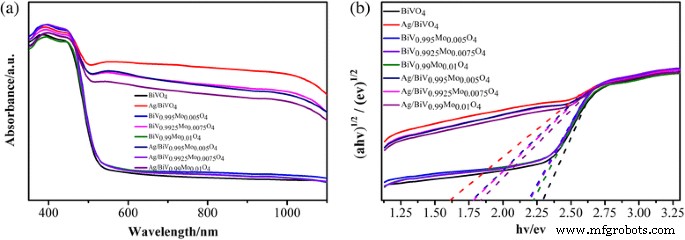
UV–vis diffuse reflectance spectra: a for all samples; b derived bandgap values.
Photoluminescence (PL) measurements (Fig. 7) at 310 nm excitation reveal decreasing emission intensity from BiVO₄ to Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄, indicating progressively more efficient electron–hole separation. The lowest PL intensity for Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄ correlates with its superior photocatalytic activity.
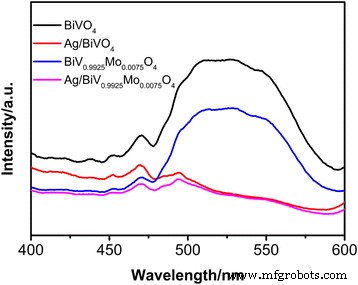
PL spectra of pristine BiVO₄, Ag/BiVO₄, BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄, and Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄ composites.
Visible‑light photocatalytic tests (Fig. 8) demonstrate that Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄ achieves complete RhB decolorization within 2 h, whereas BiVO₄, Ag/BiVO₄, and BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄ alone show only 7 %, 8 %, and 10 % degradation, respectively. This underscores the synergistic effect of Mo doping and Ag decoration in suppressing recombination and enhancing charge‑carrier lifetimes.
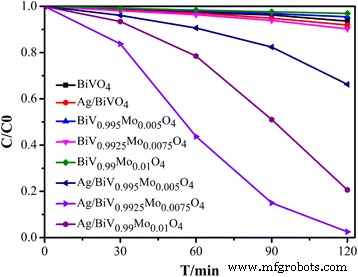
Photocatalytic degradation of RhB by BiVO₄, Ag/BiVO₄, BiV₁₋ₓMoₓO₄, and Ag/BiV₁₋ₓMoₓO₄ composites.
Recyclability tests (Fig. 9) show that Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄ retains 99 % RhB degradation after five consecutive cycles, indicating excellent structural stability.
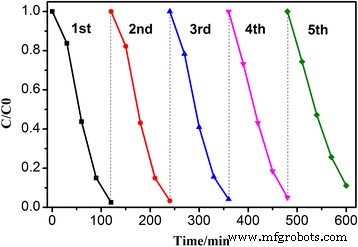
Five‑cycle performance of Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄ for RhB photodegradation.
Time‑resolved photoluminescence (Fig. 10) yields carrier lifetimes of 1.23 ns (BiVO₄), 1.82 ns (Ag/BiVO₄), and 2.09 ns (Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄). The extended lifetimes confirm that Ag and Mo synergistically facilitate charge separation.
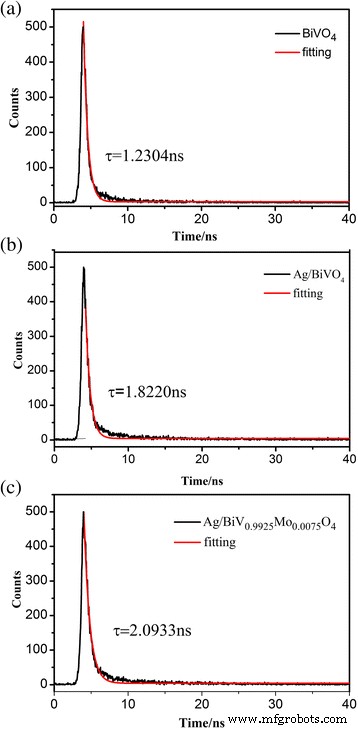
ns‑level decay curves of BiVO₄, Ag/BiVO₄, and Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄ composites.
Quenching experiments (Fig. 11) reveal that adding tert‑butanol reduces activity from 97.5 % to 78.1 %, indicating •OH as a primary oxidant. In contrast, 1.4‑benzoquinone actually accelerates degradation, suggesting that the surface plasmon resonance (SPR) of Ag enhances electron transfer to O₂, forming •O₂⁻. The pronounced decline upon ammonium oxalate addition confirms that photogenerated holes (h⁺) are also essential.
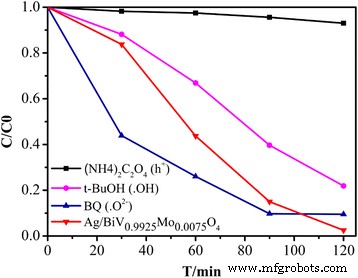
Effect of radical scavengers on RhB degradation by Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄.
Electron spin resonance (ESR) under visible light (Fig. 12) confirms the generation of •O₂⁻ and •OH radicals, reinforcing the proposed mechanism involving h⁺, •O₂⁻, and •OH.
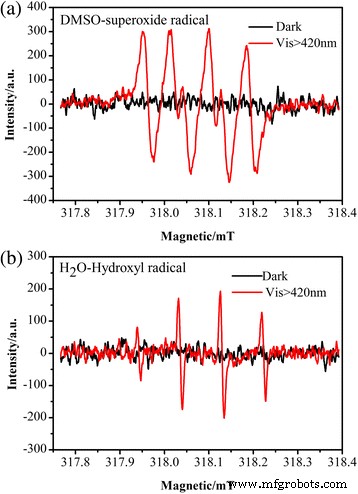
ESR spectra of Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄ in (a) DMSO and (b) water.
Figure 13 illustrates the proposed photocatalytic cycle. Under visible light, electrons are excited from the valence band of BiVO₄ to its conduction band, then migrate to Ag nanoparticles acting as electron sinks, which suppress recombination and facilitate transfer to surface O₂, generating •O₂⁻. Concurrently, holes oxidize adsorbed H₂O to •OH, while both h⁺ and •OH directly attack RhB molecules, leading to complete mineralization.
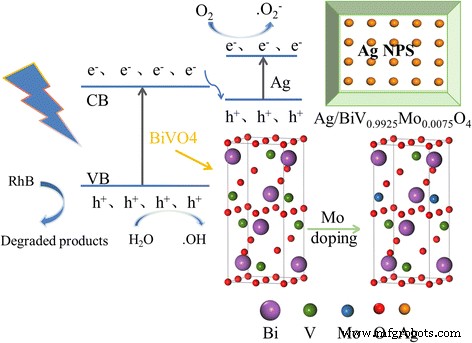
Schematic of charge‑transfer pathways in Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄ under visible light.
Conclusions
We have demonstrated a straightforward hydrothermal synthesis of Mo‑doped BiVO₄ followed by photoreduction of Ag⁺, yielding Ag/BiV₀.₉₉₂₅Mo₀.₀₀₇₅O₄ composites with Ag nanoparticles selectively anchored on the (040) facet. Structural analyses confirm that the (040) surface is enlarged during synthesis, enhancing electron‑hole separation. The resulting material exhibits exceptional photocatalytic degradation of RhB under visible light, achieving complete removal in two hours and maintaining 99 % activity over five cycles. This work provides a practical strategy for tailoring BiVO₄ photocatalysts with high activity, offering promising avenues for energy conversion and environmental remediation.
Nanomaterials
- Optimizing Sb-Loaded Palygorskite Nanoparticles for Superior Catalytic Hydrogenation of p-Nitrophenol
- Impact of Magneto‑Dipole Interactions on the Specific Absorption Rate of Iron Oxide Nanoparticle Assemblies
- Enhanced Photocatalytic Water Splitting with Au‑Plasmonic Nanoparticles on TiO₂/MoO₃ Nanotube Photoelectrodes
- Enhanced Visible‑Light Photocatalytic Degradation of Rhodamine B Using Bi<sub>4</sub>Ti<sub>3</sub>O<sub>12</sub>/Ag<sub>3</sub>PO<sub>4</sub> Heterojunction Nanocomposites
- Magnesium‑Doped Zinc Oxide Nanoparticles: Optimizing Photocatalytic Degradation and Antibacterial Efficacy
- Enhanced Visible‑Light Photocatalysis via In‑Situ Z‑Scheme BiOBr0.3I0.7/Ag/AgI Nanocomposites
- Partially BiVO4-Modified ZnO Porous Nanosheets: Solar‑Driven Photocatalysis with Superior Charge Separation
- Enhanced Visible-Light Photocatalysis by Anchoring Plasmonic Ag@AgCl Nanocrystals on ZnCo₂O₄ Microspheres
- ZnTiO3/Bi4NbO8Cl Heterojunctions: A Novel Approach to Boost Photocatalytic Efficiency
- Anion‑Controlled Fabrication of ZnO 1D Necklace‑Like Nanostructures for Superior Photocatalytic Performance



