Enhanced Photocatalytic Water Splitting with Au‑Plasmonic Nanoparticles on TiO₂/MoO₃ Nanotube Photoelectrodes
Abstract
While TiO₂ has long been a staple in photocatalysis, its wide band gap (3.2 eV) limits absorption to ultraviolet light, which constitutes only about 4% of solar energy. Recent advances focus on tailoring TiO₂’s optical properties, often by coupling it with a secondary metal‑oxide semiconductor. When TiO₂ contacts MoO₃, the photogenerated holes from MoO₃’s valence band can transfer to TiO₂, reducing electron–hole recombination and enhancing charge separation. Additionally, incorporating plasmonic Ag or Au nanoparticles boosts photocurrent via near‑field amplification and light scattering. In this study, we fabricated a three‑dimensional TiO₂ nanotube core coated with a MoO₃ shell (TNTs@MoO₃) using a two‑step anodic oxidation followed by a hydrothermal growth of MoS₂ and subsequent annealing. The resulting core–shell structure, characterized by FE‑SEM and XPS, was further decorated with Au nanoparticles to harness surface plasmon resonance (SPR) for superior photocatalytic performance.
Background
The global energy demand continues to rise, driving research into clean, renewable sources such as solar, geothermal, tidal, and biomass energy. Photocatalytic water splitting stands out as a direct route to convert sunlight into chemical fuel. Strategies to improve efficiency include expanding reaction surfaces, depositing catalytic metals, and combining semiconductors. TiO₂ nanotube arrays, prized for their high surface area and directional charge transport, are nonetheless limited by their UV‑only absorption and rapid charge recombination. Researchers have therefore explored methods to shift TiO₂’s absorption into the visible spectrum—nitrogen doping, noble‑metal surface modification, and coupling with narrow‑bandgap semiconductors. MoO₃, a p‑type oxide with a 2.8 eV band gap and excellent hole conductivity, can form a heterojunction with TiO₂ that promotes visible‑light absorption and charge separation. Plasmonic photocatalysis further amplifies efficiency by generating intense local electric fields when the incident light resonates with metal nanoparticles. In this work, we synthesized a MoS₂‑coated TiO₂ nanotube, oxidized it to MoO₃, and then introduced Au nanoparticles to exploit SPR, aiming to boost water‑splitting activity under visible illumination.
Methods
Fabrication of the TiO₂ Nanotubes
The TiO₂ nanotubes (TNTs) were produced via a two‑step anodic oxidation. Titanium foil (cut to size) was sequentially sonicated in acetone, ethanol, and deionized water for 5 min. Anodization employed a conventional two‑electrode setup (Ti anode, carbon cathode) with 0.3 wt% NH₄F in ethylene glycol (5 vol% H₂O) at room temperature. The first anodization lasted 30 min at 60 V; the resulting tubes were removed in 1 M HCl with ultrasonic agitation. A second anodization under identical conditions yielded a uniform nanotube array, which was then washed with ethanol and DI water and annealed at 450 °C (2 °C/min) for 4 h to form anatase TNTs.
Synthesis of TNTs@MoO₃ Core–Shell Structure
MoS₂ nanosheets were first grown hydrothermally: 0.12 g Na₂MoO₄·2H₂O and 0.24 g thioacetamide were dissolved in 80 mL DI water and stirred for 15 min. TNTs were then placed in a 100‑mL Teflon‑lined autoclave with the solution, heated to 200 °C (3 °C/min) for 24 h, and cooled. The resulting TNTs@MoS₂ were washed and annealed in air at 450 °C (2 °C/min, 4 h) to oxidize MoS₂ into MoO₃, yielding the core–shell TNTs@MoO₃.
Deposition of Au Nanoparticles
Au/TNTs@MoO₃ electrodes were fabricated by first treating TNTs@MoO₃ with a hydrothermal step to promote surface adhesion, followed by sputtering of Au nanoparticles to achieve a uniform, island‑like distribution.
Characteristic Analysis and Photocurrent Measurements
Microstructure and elemental composition were examined by FE‑SEM and EDS. XPS quantified the bonding states of Ti, O, Mo, and S. Photocatalytic performance was assessed in 1 M NaOH using a three‑electrode setup under 532‑nm laser irradiation (1‑mm spot) at room temperature.
Results and Discussion
Figure 1 displays SEM images and EDS maps of the synthesized materials. TNTs (Fig. 1a) exhibit uniform pores (100–120 nm). The MoS₂‑coated tubes (Fig. 1b) retain this porosity, ensuring ample active sites. After annealing, the TNTs@MoO₃ core–shell structure (Fig. 1c) shows a smooth shell without pore blockage. Au/TNTs@MoO₃ (Fig. 1d) reveals evenly dispersed Au islands, confirming the desired SPR‑active surface.

SEM images of a TNTs, b TNTs@MoS₂, c TNTs@MoO₃, and d Au/TNTs@MoO₃ (left), with EDS mapping (right)
XPS spectra (Fig. 2) confirm the chemical states. Peaks at 464.6 eV (Ti2p₁), 458.9 eV (Ti2p₃), and 530.4 eV (O1s) are characteristic of TiO₂. MoS₂ shows Mo3d₃/₅ peaks at 231.6/228.9 eV and a S2s signal near 226 eV. After annealing, Mo3d peaks shift to 235.6/232.6 eV, indicating oxidation to Mo⁶⁺ in MoO₃. These results validate the successful conversion from MoS₂ to MoO₃.
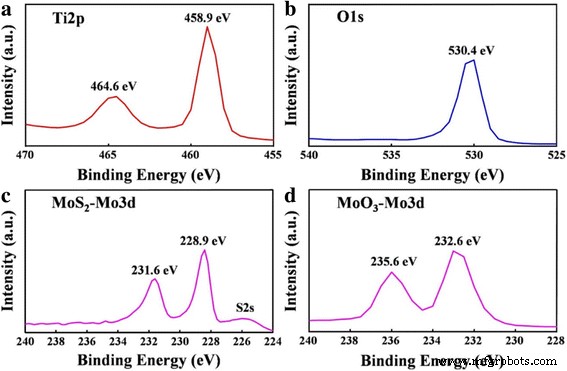
XPS analysis of a Ti2p, b O1s, c Mo3d of MoS₂, and d Mo3d of MoO₃
Photocurrent measurements (Fig. 3) demonstrate that TNTs@MoO₃ outperforms bare TNTs due to enhanced charge separation at the heterojunction. Incorporating Au nanoparticles further boosts the photocurrent by ~1.5× at –1 V bias, attributed to SPR‑induced local field enhancement. Even without bias (0 V), Au/TNTs@MoO₃ maintains superior photocurrent, confirming the intrinsic catalytic benefit of Au. Stability tests under continuous 532‑nm irradiation (Fig. 3d) show that Au/TNTs@MoO₃ retains 57% of its initial current after 1.5 h at 0.8 V. Electrochemical impedance spectroscopy (Nyquist plots, Fig. 3e) reveals a smaller semicircle for Au/TNTs@MoO₃, indicating reduced charge transfer resistance and faster carrier mobility.
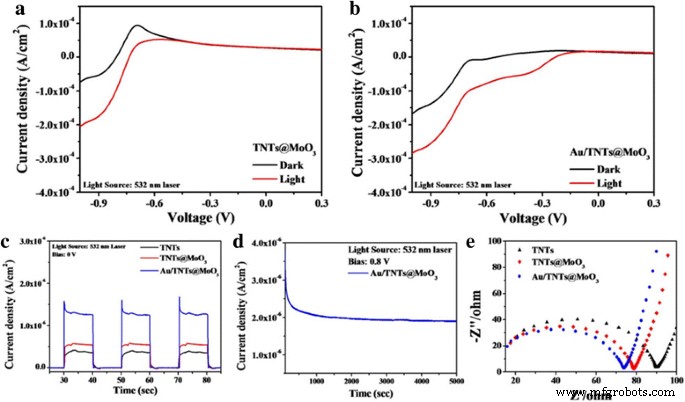
Linear sweep curves of photoelectrodes a without and b with light irradiation, and photocurrent responses at c 0 V (532‑nm laser). d Prolonged photocurrent measurements under 532‑nm laser irradiation. e Nyquist plots of various photoelectrodes
Conclusions
Supporting information
The supporting information (Additional file 1) includes Raman spectra of the MoS₂ layer, SEM‑derived pore size distribution, and a detailed discussion of the enhancement mechanism.
This work demonstrates that a MoO₃ coating on TiO₂ nanotubes effectively extends photon absorption into the visible range. Adding plasmonic Au nanoparticles yields a substantial increase in photocurrent for water‑splitting under visible light. The engineered TiO₂/MoO₃ heterojunction promotes efficient charge transfer and suppresses recombination, underscoring the importance of band‑gap engineering and plasmonic integration in next‑generation photocatalysts.
Nanomaterials
- Enhanced Photocatalytic Performance of ZnO/In₂O₃ Hybrid Nanostructures via Hydrothermal Synthesis
- Enhanced Visible‑Light Photocatalytic Degradation of Rhodamine B Using Bi<sub>4</sub>Ti<sub>3</sub>O<sub>12</sub>/Ag<sub>3</sub>PO<sub>4</sub> Heterojunction Nanocomposites
- Ag Nanoparticle-Enhanced BiV1‑xMoxO4 Composites: Synergistic Photocatalytic Performance under Visible Light
- Influence of Peptizing Acid on TiO₂ Phase Composition and Photocatalytic Efficiency: A Comparative Study of Sulfuric, Nitric, and Acetic Acids
- Facile Thermal Decomposition Loading of CeO₂ Nanoparticles onto Anodic TiO₂ Nanotube Arrays
- Boosting Perovskite Solar Cell Efficiency Using Er3+-Yb3+-Li+ Tri‑Doped TiO₂ Up‑Conversion Layer
- Enhanced Photocatalytic Degradation of Methyl Orange Using CdTe Quantum Dot/BiOI‑Coated TiO₂ Hollow Microspheres under Simulated Sunlight
- High‑Sensitivity Fe³⁺ Detection Using Ag‑Functionalized TiO₂ Nanotube Arrays via Anodic Stripping Voltammetry
- Ultra‑Thin TiO₂ Nanomembranes via Atomic Layer Deposition Deliver Record‑High Capacitance for Pseudocapacitor Electrodes
- Enhanced Photoelectrochemical Water Splitting with TiO₂ Nanosheet Arrays, Layered SnS₂, and CoOx Nanoparticles



