Visible‑Light Photocathodic Protection of 304 Stainless Steel Using Bi₂Se₃‑Sensitized TiO₂ Nanotube Films
Abstract
By coupling titanium dioxide (TiO₂) nanotube arrays with the narrow‑gap semiconductor bismuth selenide (Bi₂Se₃), we achieved a substantial improvement in photocathodic protection for 304 stainless steel under visible light. The Bi₂Se₃/TiO₂ nanocomposites were fabricated via a two‑step, scalable approach: anodic oxidation of Ti foil to produce ordered TiO₂ nanotubes, followed by chemical‑bath deposition of Bi₂Se₃ nanoflowers. Structural and compositional analyses using SEM, EDS, XPS, and XRD confirmed a uniform, flower‑like Bi₂Se₃ coverage on the TiO₂ surface. Photocurrent measurements revealed that the Bi₂Se₃/TiO₂ films generated significantly higher visible‑light photocurrents than pristine TiO₂, owing to efficient separation of photo‑excited electron‑hole pairs. When coupled with 304 stainless steel, the Bi₂Se₃/TiO₂ photoanode with a Bi³⁺ precursor concentration of 0.5 mmol L⁻¹ delivered the most negative open‑circuit potential shift (−996 mV) and the largest photocurrent density (415 µA cm⁻²). These results demonstrate that Bi₂Se₃‑sensitized TiO₂ nanotube films are a promising, eco‑friendly strategy for enhancing visible‑light photocathodic protection of stainless steel.
Background
Stainless steels, especially 304 grade, are widely used in infrastructure and marine applications due to their inherent corrosion resistance. Nonetheless, they remain vulnerable in aggressive environments—acidic media, chloride‑rich solutions, or elevated temperatures—where conventional protection methods (coatings, sacrificial anodes, impressed‑current cathodic protection) face limitations such as environmental concerns and finite service life. Photocathodic protection, introduced in the mid‑1990s, offers a renewable, self‑sustaining alternative by harnessing solar energy to drive protective cathodic currents.
Titanium dioxide (TiO₂) is a prototypical photocatalyst known for its chemical stability and low cost. However, its wide bandgap (3.2 eV) restricts light absorption to the UV region, and rapid recombination of photogenerated carriers hampers its protective performance. Numerous strategies—hetero‑atom doping, coupling with narrow‑gap semiconductors, and surface sensitization—have been explored to extend TiO₂’s photoresponse into the visible spectrum. Bismuth selenide (Bi₂Se₃), a layered n‑type semiconductor with a narrow bandgap (0.35 eV) and high absorption coefficient, emerges as an ideal sensitizer. Its surface states and strong spin–orbit coupling further enhance charge separation, while its synthesis via cost‑effective chemical‑bath deposition enables scalable production of nanoflowers that can be uniformly deposited on TiO₂ nanotube arrays.
Methods
All reagents were analytical grade and used without further purification; deionized water was employed for all aqueous solutions.
TiO₂ Film Preparation
Commercial Ti foils (20 mm × 10 mm × 0.3 mm, > 99.9 % purity) were polished with a mixture of NH₄F, H₂O, H₂O₂ and HNO₃, then cleaned in deionized water and ethanol. TiO₂ nanotube arrays were grown by anodic oxidation at 20 V for 1 h in an ethylene glycol solution containing NH₄F and H₂O. The resulting films were rinsed, dried, and annealed at 450 °C for 2 h to crystallize anatase TiO₂.
Bi₂Se₃ Deposition
Bi₂Se₃ nanoflowers were deposited via chemical‑bath deposition. A 1.0 mmol L⁻¹ Bi³⁺ solution (prepared from Bi(NO₃)₃·5H₂O and H₃NTA) was mixed with ascorbic acid and ammonium hydroxide to adjust the pH to 8.6–8.9. Na₂SeSO₃ was then introduced. TiO₂ substrates were immersed in the bath, covered, and heated at 80 °C for 200 min. By varying the initial Bi³⁺ concentration (0.25, 0.5, 1.0 mmol L⁻¹), three composites—Bi₂Se₃/TiO₂‑0.25, ‑0.5, ‑1.0—were obtained.
Characterization
SEM (Hitachi S‑4800) revealed nanotube morphology and Bi₂Se₃ coverage. EDS (Oxford Energy 350) and XPS (Thermo ESCALAB 250Xi) provided elemental composition and oxidation states. UV‑Vis diffuse reflectance spectroscopy (Hitachi UH4150) measured optical absorption, while XRD (Bruker AXS‑D8) identified crystalline phases.
Photoelectrochemical Tests
A two‑cell setup was employed: a photolysis cell (0.1 mol L⁻¹ Na₂S/0.2 mol L⁻¹ NaOH) acted as a sacrificial agent, and an electrolytic cell (0.5 mol L⁻¹ NaCl) housed the 304 ss working electrode. A Pt counter electrode and SCE reference electrode completed the three‑electrode system. Visible light was provided by a 300 W xenon lamp. Open‑circuit potential (OCP) and photocurrent were recorded with a Gamry potentiostat before and during illumination.
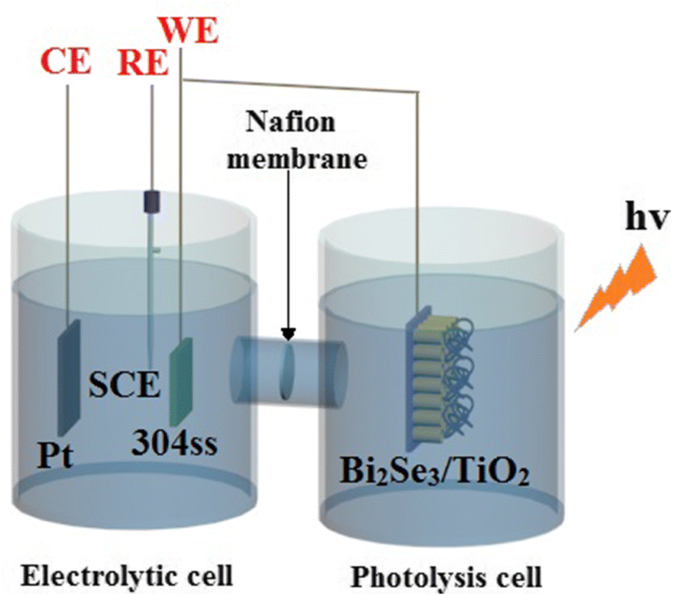
Schematic of the photoelectrochemical testing configuration.
Results and Discussion
Structural and Morphological Analysis
SEM images (Fig. 2a) confirmed highly ordered TiO₂ nanotubes (≈ 55 nm diameter, 680 nm length). Bi₂Se₃ deposition transformed the surface: at 0.25 mmol L⁻¹ Bi³⁺, isolated nanoflakes appeared (Fig. 2b); at 0.5 mmol L⁻¹, dense flower‑like assemblies (~ 800 nm) covered the tubes without occluding the pores (Fig. 2c); at 1.0 mmol L⁻¹, excessive agglomeration blocked openings (Fig. 2d). EDS of the ‑0.5 sample showed Ti, O, Bi, and Se in a 2:3 atomic ratio, confirming successful sensitization.

SEM images of (a) pristine TiO₂, (b) Bi₂Se₃/TiO₂‑0.25, (c) Bi₂Se₃/TiO₂‑0.5, (d) Bi₂Se₃/TiO₂‑1.0; (e) EDS spectrum of the ‑0.5 film.
XRD patterns (Fig. 3a) revealed anatase TiO₂ peaks (JCPDS 21‑1272) and, for the ‑1.0 film, additional Bi₂Se₃ reflections (JCPDS 33‑0214), albeit weak due to low loading. XPS confirmed Ti⁴⁺ oxidation state, lattice and adsorbed oxygen species, and the presence of Bi in both elemental and oxidized forms, as well as Se²⁻, corroborating the formation of a Bi₂Se₃/TiO₂ heterojunction.
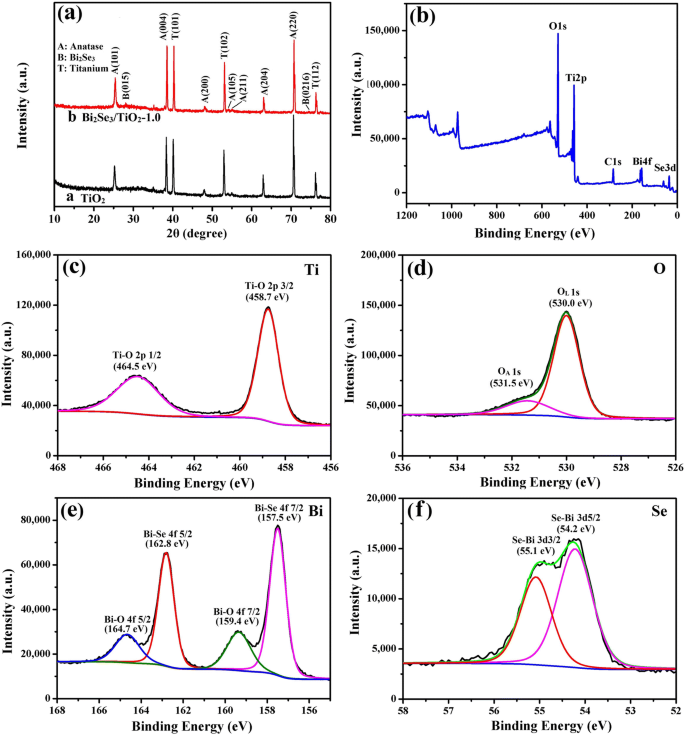
Structural characterization of the ‑1.0 composite: (a) XRD; (b) survey XPS; (c) Ti 2p; (d) O 1s; (e) Bi 4f; (f) Se 3d.
UV‑Vis diffuse reflectance (Fig. 4) showed the TiO₂ band edge at ~ 380 nm (3.2 eV). Bi₂Se₃/TiO₂ films exhibited strong absorption across 350–800 nm, confirming that the narrow‑gap Bi₂Se₃ extends photoresponse into the visible region.
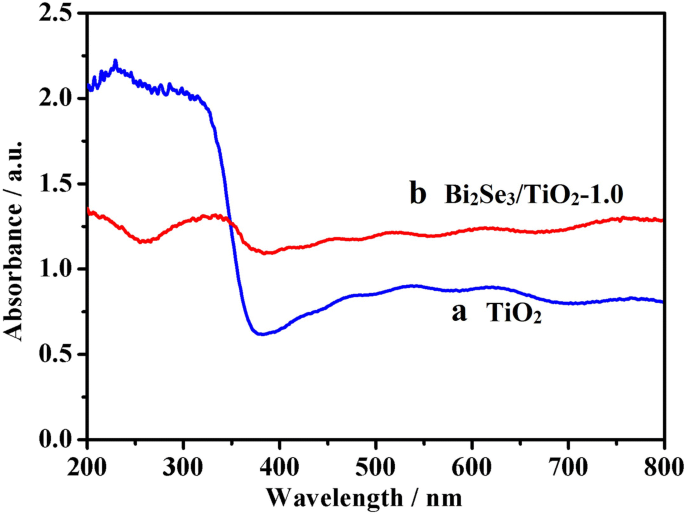
UV‑Vis spectra of TiO₂ (a) and Bi₂Se₃/TiO₂‑1.0 (b).
Photocathodic Protection Performance
Under intermittent visible light, the OCP of 304 ss coupled to Bi₂Se₃/TiO₂‑0.5 dropped to −996 mV, markedly more negative than the −905 mV observed with TiO₂ alone (Fig. 5). This shift indicates effective cathodic polarization and superior protection. Excess Bi₂Se₃ (‑1.0) slightly reduced performance, likely due to increased recombination sites, whereas too little Bi₂Se₃ (‑0.25) offered insufficient light harvesting.
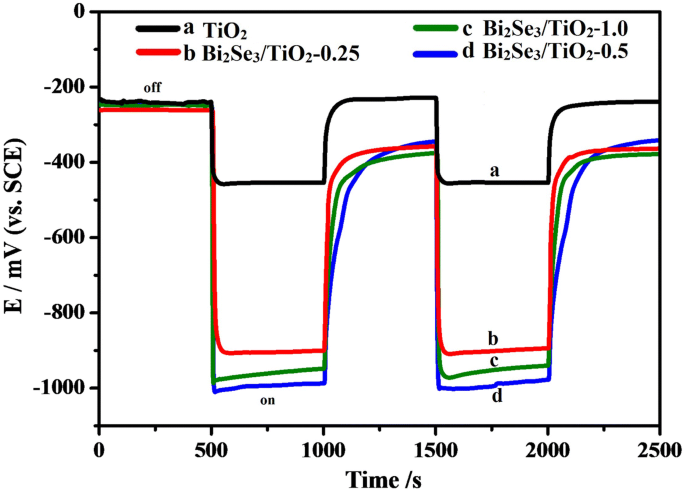
OCP response of 304 ss with TiO₂ and Bi₂Se₃/TiO₂ composites in 0.5 mol L⁻¹ NaCl.
Photocurrent measurements (Fig. 6) showed that the ‑0.5 composite achieved 415 µA cm⁻², surpassing both TiO₂ (≈ 85 µA cm⁻²) and the ‑1.0 sample (≈ 160 µA cm⁻²). The enhanced current confirms efficient separation of photo‑generated carriers, facilitated by the Bi₂Se₃/TiO₂ heterojunction and the flower‑like morphology that maximizes surface area.
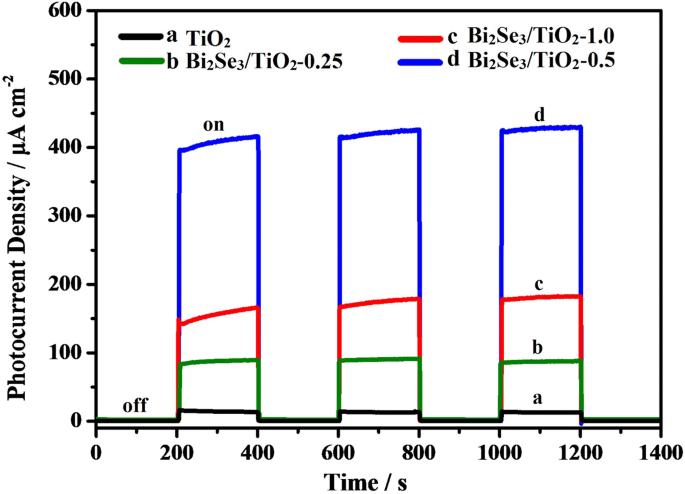
Photocurrent density vs. time for TiO₂ and Bi₂Se₃/TiO₂ composites in 0.1 mol L⁻¹ Na₂S/0.2 mol L⁻¹ NaOH.
Figure 7 illustrates the charge‑transfer pathway: visible photons excite electrons in Bi₂Se₃ (CB → TiO₂ CB), while holes transfer to Bi₂Se₃ VB and are scavenged by S²⁻, generating S. Electrons flow through the external circuit to the 304 ss electrode, reducing dissolved O₂ and shifting the potential negatively. The presence of adsorbed oxygen (O_A) on Bi₂Se₃ further promotes charge separation, enhancing photocathodic protection.
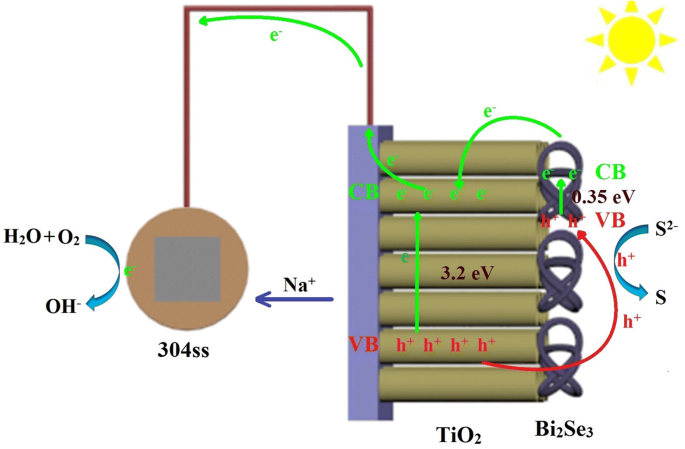
Charge‑transfer schematic for Bi₂Se₃/TiO₂ photoanode.
Conclusions
The study demonstrates that TiO₂ nanotube arrays sensitized with Bi₂Se₃ nanoflowers, fabricated via a straightforward anodization and chemical‑bath deposition route, provide a highly effective, visible‑light photocathodic protection system for 304 stainless steel. Among the tested composites, the sample prepared with 0.5 mmol L⁻¹ Bi³⁺ (Bi₂Se₃/TiO₂‑0.5) exhibited the optimal balance of light harvesting, charge separation, and surface coverage, achieving the most negative OCP shift (−996 mV) and the highest photocurrent density (415 µA cm⁻²). These findings support the potential of Bi₂Se₃/TiO₂ nanocomposites as a green, scalable solution for long‑term corrosion protection in aggressive environments.
Nanomaterials
- Choosing the Right Stainless Steel: 304 vs 316 Explained
- Facile Thermal Decomposition Loading of CeO₂ Nanoparticles onto Anodic TiO₂ Nanotube Arrays
- Enhanced Visible‑Light Photocatalysis via Cu2−xSe‑Modified Monoclinic BiVO4: 15.8‑Fold Activity Boost
- Enhanced Visible‑Light Photocatalysis via Z‑Scheme Ag3PO4/TiO2 Heterojunctions
- TiO₂/Porous Carbon Coated Celgard 2400 Separators Enhance Lithium/Sulfur Battery Performance
- Self‑assembled SnIn4S8/TiO₂ Nanotube Photoanode for Enhanced Visible‑Light Photocathodic Protection of Q235 Steel
- Choosing the Right Stainless Steel: 304 vs. 430 Explained
- Comparing 304 vs 409 Stainless Steel for Exhaust Systems: Which Is Best?
- Choosing Between 304 and 316 Stainless Steel: A Practical Guide for Your Project
- Comparing 304, 316, and 316L Stainless Steel: Performance & Applications



