Fast On‑Site Detection of Rongalite in Food Using an Aptamer‑Based Sandwich Lateral Flow Strip Assay
Abstract
We present a rapid, user‑friendly lateral flow strip assay (LFSA) that employs two highly specific aptamers—A09 and B09—functionalized with gold nanoparticles (AuNPs) to detect rongalite, a hazardous reagent occasionally used as a food additive. The biotin‑labeled A09 aptamer is immobilized on a streptavidin‑coated nitrocellulose membrane as the capture probe, while the AuNP‑conjugated B09 aptamer serves as the signaling probe. The assay delivers a clear red line at the test zone for rongalite concentrations as low as 1 µg mL−1, observable by the naked eye within 15 minutes, making it ideal for on‑site screening of agricultural and food products.
Background
Rongalite (sodium hydroxymethylsulfinate) is widely employed in industrial dyeing and polymer chemistry as a reducing agent. However, its inadvertent or intentional addition to food items has caused serious health concerns, including formaldehyde release and adverse effects reported in China [5]. Reliable, rapid detection methods are therefore essential to safeguard food safety and public health.
Aptamers—single‑stranded DNA or RNA molecules—offer distinct advantages over antibodies: they are chemically synthesized with high batch consistency, exhibit low immunogenicity, and can be readily modified for diverse sensing platforms [6]. While electrochemical and enzyme‑linked aptamer assays exist for rongalite, they often require complex instrumentation and prolonged analysis times [10]. In contrast, lateral flow strip assays (LFSAs) provide a single‑step, portable, and low‑cost solution suitable for point‑of‑care testing.
To date, only a handful of aptamer‑based chromatographic strip assays have targeted rongalite [13]. Given the growing regulatory scrutiny and the prevalence of illicit additives, we developed a sandwich‑format LFSA that harnesses the specificity of two aptamers and the optical clarity of AuNPs, enabling rapid, on‑site detection of rongalite in diverse food matrices.
Methods
Materials and Reagents
Rongalite was sourced from Tokyo Chemical Industry Co., Ltd. Gold(III) chloride trihydrate (HAuCl4) and trisodium citrate were purchased from Sigma‑Aldrich (USA). Supporting reagents (NaCl, BSA, sucrose, PEG20000, Tween‑20) were obtained from Beijing Biotopped Science and Technology Co., Ltd. Nitrocellulose (NC) membranes (Pall 90, Pall 170, Millipore 135) were supplied by Pall Corporation and Millipore Corporation via Jiening Biotech Company. Food samples—including rice cake (ersi), noodles, tofu, and glucono‑δ‑lactone tofu—were collected from local markets.
Aptamer Preparation via SELEX
The systematic evolution of ligands by exponential enrichment (SELEX) protocol was followed to isolate aptamers with high affinity for rongalite. The initial 82‑mer ssDNA library (5′‑GACATATTCAGTCTGACAGCG‑N40‑GATGGACGAATATCGTCTAGC‑3′) was incubated with rongalite, unbound sequences were washed away, and bound aptamers were amplified by PCR. After 12 enrichment cycles—including counter‑selection against structurally related compounds—clonal sequencing yielded two high‑affinity aptamers: A09 and B09. Their sequences are listed below and were synthesized by Tsingke Biological Technology.
Primary aptamer A09 (biotin‑labeled): 5′‑Biotin‑GACATATTCAGTCTGACAGCGGAAGCGGGTCAGTCCAACTCACGGTCTCGCATGCACGGGAGATGGACGAATATCGTCTAGC
Secondary aptamer B09 (biotin‑labeled): 5′‑Biotin‑GCTAGACGATATTCGTCCATCTCCCGTGCATGCGAGACCGTGAGTTGGACTGACCCGCTTCCGCTGTCAGACTGAATATGTC‑HS‑3′
Production of AuNPs
Gold nanoparticles (~40 nm diameter) were synthesized by citrate reduction of HAuCl4 following a standard protocol [18]. Size and monodispersity were confirmed by UV/Vis spectroscopy (λmax ≈ 530 nm) and transmission electron microscopy (TEM) (Fig. 2). The secondary aptamer B09 was conjugated to AuNPs by incubating 1 mL AuNP solution with 4 µL of 100 µM B09, followed by salt‑induced precipitation, centrifugation, and resuspension in 1 mL PBS (pH 7.4) containing 5 % BSA, 5 % sucrose, 1 % PEG20000, and 0.05 % Tween‑20.
Lateral Flow Strip Design
The LFSA assembly (Fig. 3) involved a sample pad, gold conjugate pad, NC membrane (Pall 90), and absorbent pad on a backing card. The sample pad was pre‑treated with PBS + 3 % BSA + 0.05 % Tween‑20 and dried at 37 °C for 2 h. The gold conjugate pad received the B09‑AuNPs. The test line on the NC membrane was created by spotting 1 µL biotin‑A09 (10 µM) mixed with 10 µL streptavidin (1 mg mL−1) for 1 h at 4 °C, while the control line consisted of streptavidin alone. After drying, the strip was cut into 40 mm sections and stored overnight at 37 °C.
Specificity Test
Test strips were challenged with 80 µL of 10 µg mL−1 rongalite, formalin, or deionized water. The appearance of a distinct red test line confirmed specificity, whereas the control line remained visible under all conditions.
Dose‑Dependent Test
Rongalite concentrations (0, 0.8, 1, 5, 10 µg mL−1) were prepared in PBS and 80 µL was applied to the sample pad. A visible red line within 15 min defined the detection limit, which was observed at 1 µg mL−1.
Food Sample Test
Five food samples suspected of containing rongalite were extracted (1 g + 10 mL water). After filtration, 80 µL of each extract was tested on the LFSA. Results were cross‑validated by high‑performance liquid chromatography (HPLC).
Results and Discussion
Affinity and Specificity of Aptamers
Non‑linear regression of saturation curves (Fig. 4a) yielded dissociation constants of 61.1 ± 16.4 nM for A09 and 39.8 ± 12.7 nM for B09, confirming strong, specific binding to rongalite (Fig. 4b).
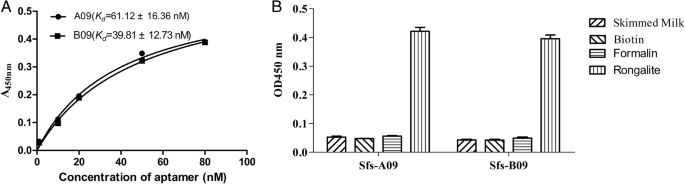
a Binding curves for A09 and B09 (Kd determination). b High‑affinity interaction between aptamers and rongalite.
Assay Performance
The sandwich LFSA produces a red test line when AuNP‑B09 binds rongalite and is captured by immobilized A09. The control line, formed by streptavidin binding excess AuNP‑B09, confirms assay integrity (Fig. 5). Specificity testing against formalin and deionized water produced no false positives, and the detection limit was established at 1 µg mL−1—well below the levels reported in contaminated food samples [5].
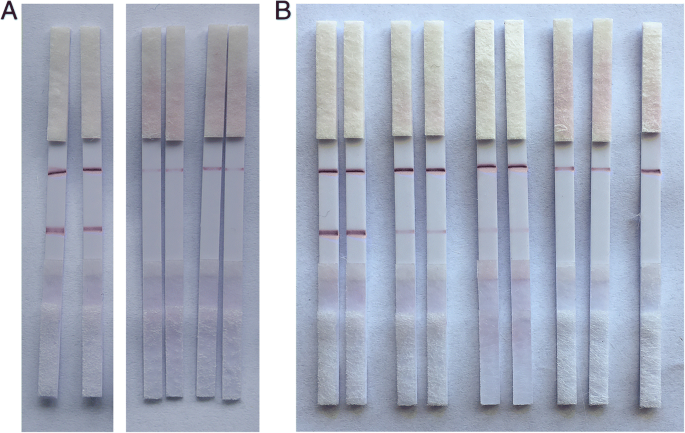
Specificity (a) and sensitivity (b) of the LFSA. a 80 µL of 10 µg mL−1 rongalite; b Standard solutions (0–10 µg mL−1) tested.
Food sample analysis confirmed the presence of rongalite in several products, corroborated by HPLC (Table 1). The assay’s robustness was attributed to the optimized buffer (PBS pH 7.4 + 5 % BSA + 5 % sucrose + 1 % PEG20000 + 0.05 % Tween‑20) and the selection of Pall 90 NC membrane, which provided optimal flow and minimal non‑specific binding.
Compared with chromatographic methods (GC, HPLC), the LFSA offers comparable sensitivity while eliminating the need for sophisticated instrumentation and extended processing times. Its low cost, single‑step operation, and visual readout make it ideal for on‑site food safety inspections and regulatory compliance.
Conclusions
We have developed a reliable, cost‑effective sandwich‑format LFSA that detects rongalite in food matrices with a limit of 1 µg mL−1. The assay harnesses the specificity of dual aptamers and the optical clarity of AuNPs, enabling immediate, on‑site screening. This platform can play a pivotal role in mitigating food‑borne rongalite contamination and safeguarding public health.
Abbreviations
- AuNPs:
Gold nanoparticles
- HPLC:
High‑performance liquid chromatography
- LFIA:
Lateral flow immunoassay
- LFSA:
Lateral flow strip assay
- NC membrane:
Nitrocellulose membrane
- POCT:
Point‑of‑care testing
- SELEX:
Systematic evolution of ligands by exponential enrichment
- TEM:
Transmission electron microscopy
Nanomaterials
- A Flexible Graphene Biosensor for Rapid, Highly Sensitive Detection of Ovarian Cancer Cells
- Rapid Thermal Annealing Enables Uniform In₂Se₃ Nanowire Growth at Low Temperatures
- Optimizing a-IGZO TFTs for Large‑Area Displays with a Clean Etch‑Stopper Nano‑Layer Process
- Ethanol‑Driven Direct CVD Growth of High‑Quality Graphene on SiO₂ via an Intermediate Carbon Nanotube Stage
- Portable Plasmonic Thermal Device for Accurate Quantification of Lateral Flow Assays
- Optimizing Imaging Flow Cytometry to Quantify HEK293T Extracellular Vesicle Uptake Kinetics and Specificity
- Accelerated, Hydrogen-Free Production of Silver Nanofibers Using Platinum Nanoparticles
- The Ultimate Stainless Steel Guide for Rapid Prototyping: Selecting the Right Grade
- Why VIA-in-Pads Enhance PCB Performance: Key Benefits
- Accelerate Production: Rapid Prototyping with Urethane Molding



