A Flexible Graphene Biosensor for Rapid, Highly Sensitive Detection of Ovarian Cancer Cells
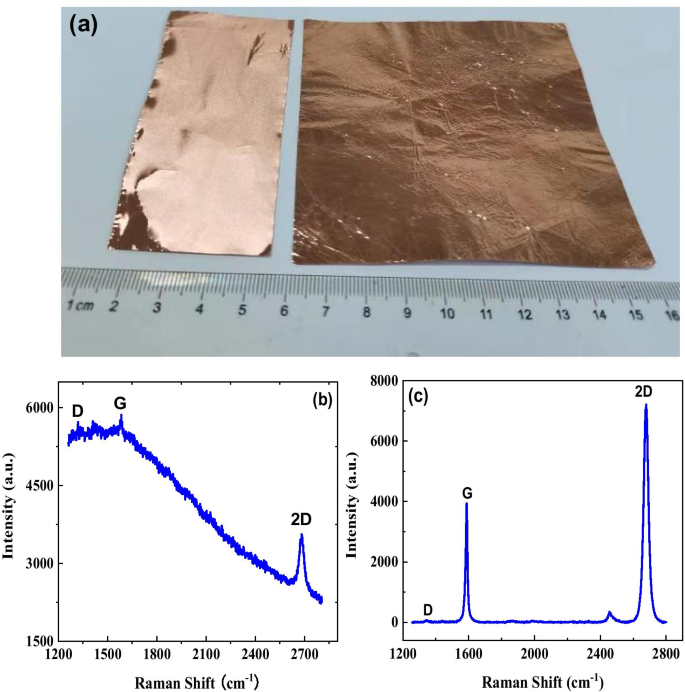
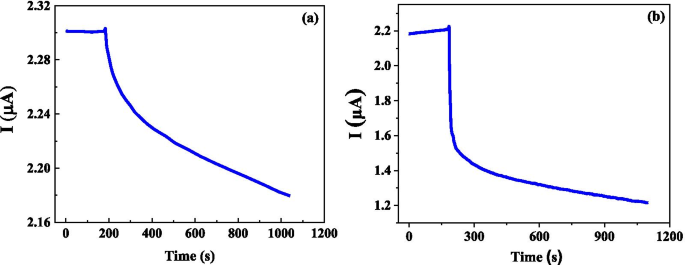
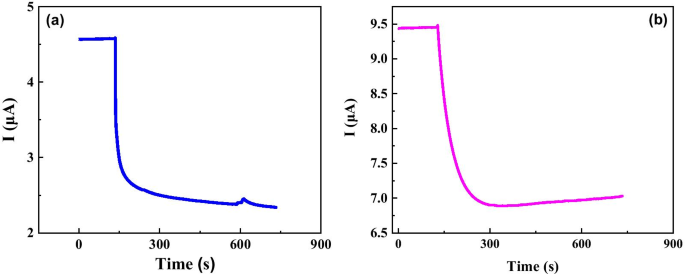
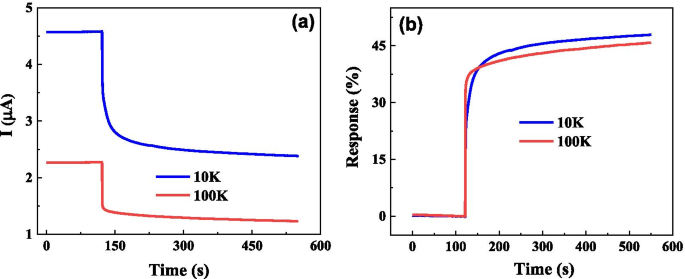
Early detection of ovarian cancer remains a critical challenge, largely due to nonspecific symptoms and the absence of reliable screening tools. We present a cost‑effective, flexible graphene biosensor fabricated on polyethylene terephthalate (PET) that detects circulating tumor cells (CTCs) with high sensitivity and speed. The sensor distinguishes between cell culture medium and ovarian cancer cell suspensions, responds distinctly to different cell types, and maintains a detection limit of only a few dozen cells per milliliter. Remarkably, the biosensor delivers a measurable electrical response within 5 seconds, offering a promising platform for rapid, point‑of‑care diagnostics. Ovarian cancer is the second most prevalent gynecological malignancy and the leading cause of death among women with gynecologic cancers. Current screening strategies—imaging combined with CA125 antigen testing—offer limited sensitivity, particularly in early-stage disease, where survival rates drop sharply. Early-stage tumors release CTCs into the bloodstream, providing a noninvasive biomarker that, if detected with sufficient sensitivity, could transform patient outcomes. Although several immunomagnetic and microfluidic techniques have been developed, none yet match the high specificity and rapid turnaround needed for clinical adoption. Graphene, a two‑dimensional lattice of sp^2‑bonded carbon, has emerged as a versatile platform for biosensing due to its exceptional electrical conductivity and large surface area. Prior graphene‑based devices have successfully quantified protein biomarkers such as CEA and PSA, yet flexible, low‑cost graphene biosensors for ovarian cancer CTCs remain unexplored. This study introduces a hand‑fabricated graphene/PET sensor that addresses these gaps by combining mechanical flexibility, rapid response, and high sensitivity. Monolayer graphene was synthesized on copper foil via chemical vapor deposition (CVD). The foil underwent 5‑minute hydrochloric acid cleaning, followed by de‑ionized water rinses and nitrogen drying. In the CVD chamber, the temperature was ramped to 1000 °C under a hydrogen atmosphere (50 sccm) for 20 minutes, then methane (50 sccm) was introduced for an additional 20 minutes to grow the graphene layer. After cooling, the graphene/Cu substrate was cut, coated with PMMA, and the copper was etched in 1 M FeCl_3. The PMMA/graphene stack was rinsed, transferred onto PET, and the PMMA was removed with acetone to yield a graphene/PET film. The graphene film was cut to 1 cm×2 cm pieces and transferred onto PET. Two silver‑paste electrodes were positioned centrally. A silicone gel well, measuring several millimeters in length and width and ~1 mm in height, was cast around the electrodes to create a cell pool. The completed sensor was verified with an Agilent 4155B semiconductor analyzer. SKOV3 cells (obtained from the public laboratory of the Second Affiliated Hospital of West China) were grown in RPMI‑1640 medium supplemented with 10% calf serum, maintained at 37 °C and 5% CO_2. Cell suspensions were diluted to desired concentrations in culture medium. A 50 µL aliquot was placed in the silicone well, and the resulting current change was recorded using the Agilent 4155B analyzer. The 10 × 10 cm graphene film (Figure 1a) displayed a subtle darkening relative to bare Cu, confirming graphene coverage. Raman analysis (Figure 1b) revealed G and 2D peaks at 1580 cm^−1 and 2680 cm^−1, while the I_G/I_2D ratio (<0.5) and negligible D peak (Figure 1c) confirmed monolayer quality and low defect density. a Photograph of bare Cu foil (left) and graphene‑coated Cu foil (right). b Raman spectrum of graphene/Cu. c Raman spectrum of graphene/SiO_2/Si. Flexible sensors on PET (Figure 2) produced clear electrical signals when a 50 µL sample was added to the cell pool. The baseline current remained steady before sample addition; upon contact, the current dropped and stabilized at a new value. The relative response, defined as η = (I_0 – I)/I_0 × 100%, was 2.96% for cell‑free medium and 37.04% for a CTC solution containing 30 cells/mL, indicating a pronounced sensitivity to cancer cells. Photograph of the graphene/PET biosensor. Time‑resolved measurements (Figure 3) showed that a 30 cells/mL CTC solution reached a 5% response in just 2.1 s, while the same concentration of medium only produced a 0.15% change. The rapid, sub‑10‑second response confirms the sensor’s suitability for point‑of‑care use. a Electrical response for cell‑free medium. b Response for a 30 cells/mL cancer solution. We further tested two distinct CTC lines (SUDHL8 and OCILYS) at 10,000 cells/mL. Although both exhibited measurable responses, their current–time profiles differed significantly, suggesting that the sensor can discriminate between cell types (Figure 5). Time‑dependent response for (a) SUDHL8 and (b) OCILYS cells. Finally, we evaluated the sensor’s concentration‑dependent behavior using SUDHL8 cells at 10 K and 100 K cells/mL. Higher concentrations produced a slightly smaller current change, reflecting the insulating nature of densely packed cells (Figure 6). Nonetheless, the sensor maintained a distinct response, underscoring its quantitative potential. a Current vs. time for 10 K vs. 100 K cells/mL. b Corresponding response curves. Collectively, the data demonstrate that a hand‑crafted, flexible graphene biosensor can sensitively and rapidly detect ovarian cancer CTCs, discriminate between cell types, and quantify cell concentration—all within seconds. We have fabricated a simple, flexible graphene biosensor on PET that detects ovarian cancer CTCs with remarkable speed and sensitivity. By comparing pre‑ and post‑sample electrical signals, the device distinguishes cancerous from non‑cancerous media, identifies different cell lines, and quantifies cell concentration. This low‑cost platform holds promise for rapid, point‑of‑care diagnostics that could improve early detection and monitoring of ovarian cancer. All relevant data are included in the article and its supplementary files. Carbohydrate antigen Circulating tumor cell Polyethylene terephthalate Food and Drug Administration Chemical vapor depositionAbstract
Graphical Abstract
Introduction
Materials and Methods
Growth and Transfer of Graphene Film
Fabrication of Graphene-Based Biosensors
Culture of SKOV3 Ovarian Cancer Cells
Preparation of Cell Solution and Electrical Measurement
Results and Discussion

Conclusion
Availability of Data and Materials
Abbreviations
Nanomaterials
- Nanoparticle-Based Cancer Therapy: Advances, Mechanisms, and Clinical Translation
- Cell‑Based Drug Delivery Systems for Advanced Cancer Therapy
- Wireless Magnetoelastic Nanobiosensor Achieves Ultra‑Sensitive Atrazine Detection
- Enhanced Formaldehyde Detection at ppb Levels Using Reduced Graphene Oxide–Coated Silicon Nanowire Sensors
- Magnetic Graphene Field‑Effect Transistor Biosensor for Ultra‑Sensitive Single‑Strand DNA Detection
- Laminarin-Based Nanoparticles Deliver Protoporphyrin IX for Enhanced Photodynamic Therapy: Cellular Uptake, ROS Generation, and In Vivo Efficacy
- Nanograting‑Enhanced Flexible Waveguide for Advanced Tactile Sensing
- Eco-Friendly Microplasma Synthesis of Yellow-Emitting Carbon Quantum Dots for Cancer Cell Imaging and Photocatalytic Inactivation
- Quantitative Study of PLGA Nanoparticle Uptake in Laryngeal Cancer and Immune Cells to Improve Drug Delivery
- All‑Metal Terahertz Metamaterial Biosensor Achieves Ultra‑High Sensitivity for Protein Detection



