Wireless Magnetoelastic Nanobiosensor Achieves Ultra‑Sensitive Atrazine Detection
Abstract
This study introduces the first wireless magnetoelastic (ME) nanobiosensor that employs ME materials and gold nanoparticles (AuNPs) to detect atrazine with unprecedented sensitivity via a competitive immunoassay. When exposed to a time‑varying magnetic field, the ME substrate vibrates at its resonance frequency, which shifts proportionally to the mass loading on its surface. Coating the ME ribbon with AuNPs enhances biocompatibility, stability, and signal sensitivity, while protein A‑mediated orientation of the atrazine antibody on the AuNPs layer maximizes antigen‑binding efficiency. Atomic force microscopy confirmed successful antibody immobilization. Introducing an atrazine–albumin conjugate (Atr–BSA) amplified the competitive binding, yielding a linear, logarithmic response over a 1 ng mL⁻¹–100 µg mL⁻¹ range, with a sensitivity of 3.43 Hz µg⁻¹ mL and a detection limit of 1 ng mL⁻¹—well below the US EPA threshold. The sensor demonstrated high specificity, stability, and suitability for rapid, selective environmental monitoring.
Introduction
The surge in industrial and agricultural activities has introduced numerous environmental contaminants into ecosystems, raising global concern about their ecological and health impacts. Herbicides, in particular, persist in water and soil for years, posing significant risks. Atrazine (2‑chloro‑4‑ethylamino‑6‑isopropylamino‑1,3,5‑triazine) is the most widely used herbicide for controlling broad‑leaf weeds and grasses worldwide.
While atrazine effectively suppresses weeds, it is a potent endocrine disruptor and has been linked to cancer, birth defects, and organ damage in humans and animals. The US Environmental Protection Agency (EPA) limits atrazine in drinking water to 3 µg L⁻¹ (Lifetime Health Advisory Level). Accurate, low‑concentration detection is therefore essential.
Conventional analytical methods such as LC–MS, HPLC, and GC–MS offer high accuracy but suffer from high cost, large instrumentation, and lengthy processing times. The magnetoelastic (ME) sensor platform, based on amorphous ferromagnetic alloys (e.g., Metglas 2826 MB), offers a low‑cost, highly sensitive, and miniaturized alternative. The ME sensor vibrates at a resonance frequency determined by material dimensions and mechanical properties, and any added mass causes a measurable frequency shift, enabling mass‑sensing for physical, chemical, and biological analytes. To date, no ME sensor has been reported for atrazine detection.
Here, we present a wireless ME nanobiosensor that integrates a ME substrate with AuNPs and a protein A‑oriented atrazine antibody to achieve ppb‑level detection of atrazine. The system employs a direct competitive immunoassay, where Atr–BSA competes with free atrazine for antibody binding, amplifying the mass load and the resulting frequency shift. The optimized sensor offers a linear response across a broad concentration range and meets regulatory requirements.
Materials and Methods
Materials
Atrazine antibody, Atr–BSA, atrazine, and protein A were sourced from EastCoast Bio (Maine, USA). Simazine, prometryn, and DDT were obtained from Chengdu Huaxia Chemical Reagent Co., Ltd. Cysteamine, EDC, NHS, BSA, and PBS (pH 7.4) were purchased from Sigma‑Aldrich.
ME Nanobiosensor Fabrication
Preparation of the ME Nanosensor Platform
ME ribbons (Metglas 2826 MB) were cut to 5 mm × 1 mm × 0.028 mm and cleaned ultrasonically in acetone and ethanol, then dried with nitrogen (Fig. 1a). A 100‑nm chromium seed layer was sputtered to promote adhesion, followed by AuNP coating on both sides. Scanning electron microscopy confirmed a spherical AuNP distribution. The ribbons were annealed at 200 °C for 2 h to relieve stress and enhance AuNP adhesion, yielding a ready platform (Fig. 1b).
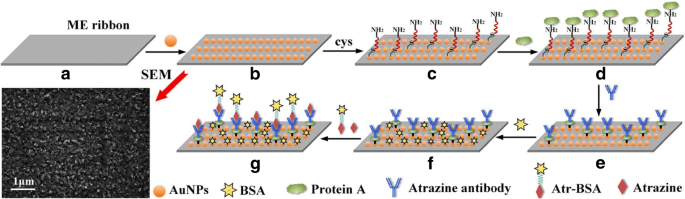
The functionalization workflow: (a) bare ME ribbon; (b) AuNP coating; (c) SAM layer; (d) protein A immobilization; (e) antibody modification; (f) BSA blocking; (g) competitive binding; SEM image of the AuNP‑coated surface
Atrazine Antibody Immobilization
The AuNP‑coated ribbons were rinsed with acetone, isopropanol, deionized water, and ethanol, then dried. They were immersed in 10 mM cysteamine for 12 h to form a self‑assembled monolayer (SAM) (Fig. 1c). Protein A (1 mg mL⁻¹) was activated with EDC/NHS (4 mg mL⁻¹ each) for 30 min, then incubated on the SAM for 30 min at 37 °C and rinsed with PBS (Fig. 1d). Atrazine antibody was added for 50 min, washed, and then blocked with 0.5 % BSA for 30 min to reduce non‑specific adsorption (Fig. 1e‑f). The completed ME nanobiosensors were ready for analysis (Fig. 1g).
Signal Measurement
The resonance frequency was measured using a pickup coil surrounding a vial, driven by a vector network analyzer (AV3620A). A static magnetic field from a bar magnet enhanced resonance. Sensors were placed wirelessly into 30 µL test samples in PBS (pH 7.4) at 25 ± 2 °C. The S11 parameter was recorded every 5 min to track the frequency shift.
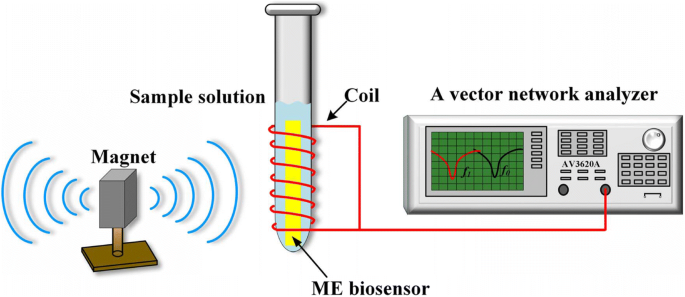
Schematic of the wireless ME nanobiosensor measurement system
Results and Discussion
Characterization of the Nanobiosensor Surface Morphology
AFM imaging confirmed successful antibody immobilization. The AuNP‑coated surface exhibited a height variation of 13.4 nm, which increased to 28.4 nm after antibody attachment—consistent with the ~15 nm diameter of IgG molecules (Fig. 3a‑b).
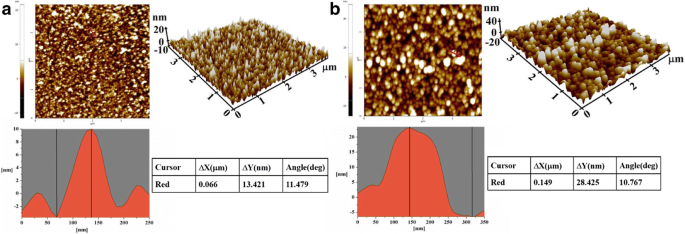
AFM images: (a) AuNP‑coated surface; (b) antibody‑modified surface
Optimization for Concentration of Atrazine Antibody
Sensor response peaked at 50 µg mL⁻¹ antibody loading. Higher concentrations (75 µg mL⁻¹) caused steric hindrance, reducing sensitivity (Fig. 4). Thus, 50 µg mL⁻¹ was selected as optimal.
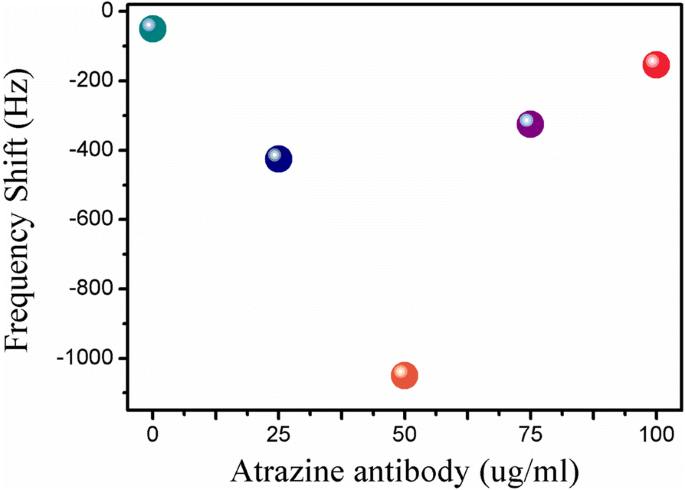
Frequency response versus antibody concentration (0–100 µg mL⁻¹)
Optimization for Concentration of Atr–BSA
Competitive binding was maximized at 40 µg mL⁻¹ Atr–BSA (Fig. 5). This concentration provided the greatest frequency shift and was used in subsequent assays.
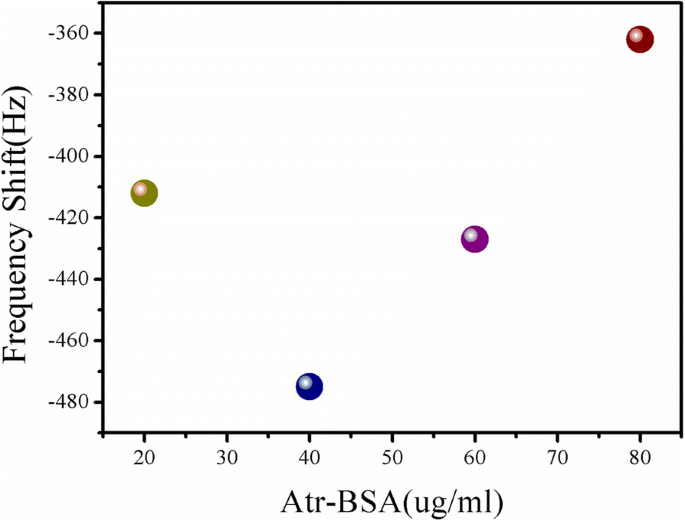
Frequency response to Atr–BSA (20–80 µg mL⁻¹)
Atrazine Detection
Real‑time monitoring showed a clear, inversely proportional shift in resonance frequency with increasing atrazine concentrations (0–100 µg mL⁻¹). The signal plateaued after ~50 min (Fig. 6). Non‑specific adsorption was negligible, as demonstrated by the blank control (<48 Hz shift).
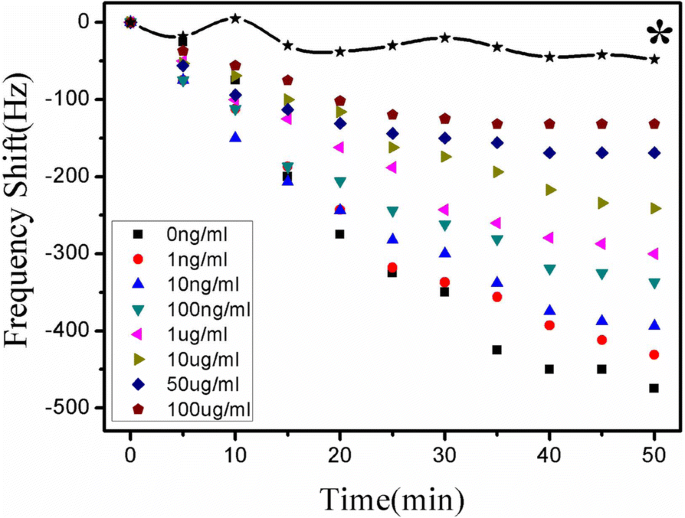
Frequency responses across atrazine concentrations (0–100 µg mL⁻¹). * Blank control
The calibration curve (Fig. 7) is linear with the logarithm of concentration (Δf = 54.717 log CAtrazine – 442.45, R² = 0.971). Sensitivity is 3.43 Hz µg⁻¹ mL, and the LOD is 1 ng mL⁻¹, well below the EPA limit. The direct competitive immunoassay format, combined with Atr–BSA amplification, yields rapid, disposable, and wireless detection.
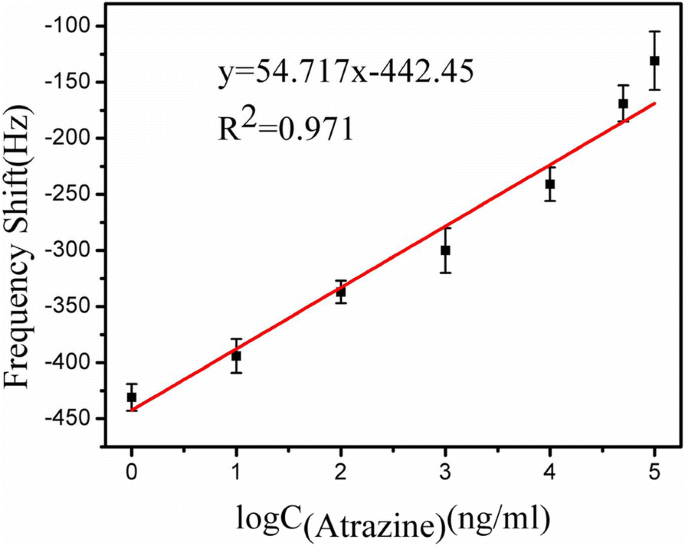
Calibration curve: 50 min frequency shift vs. atrazine concentration
ME Nanobiosensor Specificity
Testing with 100 µg mL⁻¹ of prometryn, simazine, and DDT revealed minimal cross‑reactivity; the sensor discriminated atrazine from structurally similar triazines and organochlorine insecticides (Fig. 8). This confirms high specificity.
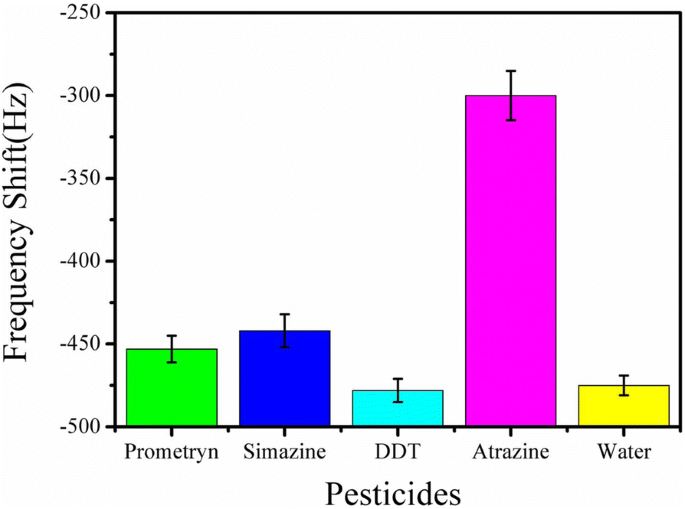
Response to interferents at 100 µg mL⁻¹
ME Nanobiosensor Stability
Six identical sensors stored at 4 °C were tested with 10 ng mL⁻¹ atrazine every other day for 6 days. The relative standard deviation was 1.8 %, indicating excellent long‑term stability (Fig. 9).
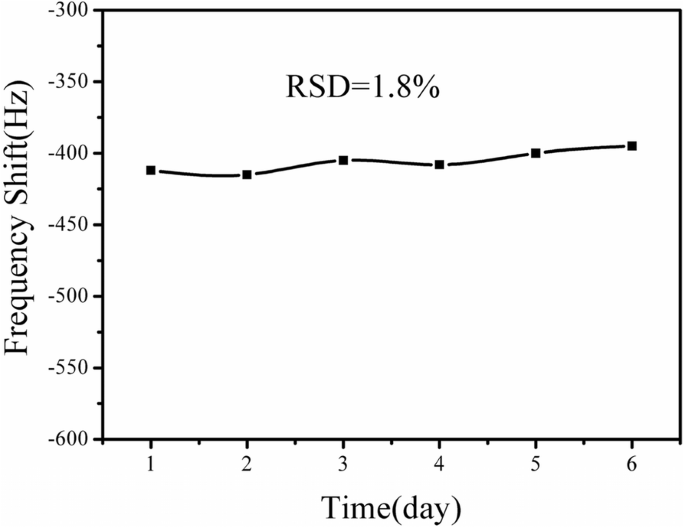
Stability over 6 days at 10 ng mL⁻¹ atrazine
Conclusions
We have engineered a wireless ME nanobiosensor that delivers real‑time, highly sensitive atrazine detection through a protein A‑oriented antibody layout and Atr–BSA‑mediated signal amplification. With 50 µg mL⁻¹ antibody and 40 µg mL⁻¹ Atr–BSA, the sensor achieves a linear range of 1 ng mL⁻¹–100 µg mL⁻¹, a sensitivity of 3.43 Hz µg⁻¹ mL, and an LOD of 1 ng mL⁻¹—well below regulatory limits. AFM validation confirms oriented antibody immobilization, and the platform shows robust specificity and stability. The low cost, disposability, and wireless nature of the sensor make it a compelling tool for monitoring atrazine and other environmental contaminants in water.
Nanomaterials
- A Flexible Graphene Biosensor for Rapid, Highly Sensitive Detection of Ovarian Cancer Cells
- Ultra‑Sensitive Electrochemical DNA Biosensor Using Acrylic‑Gold Nanocomposite for Rapid Arowana Fish Gender Determination
- Ultra‑Sensitive Magnetoelastic Immunosensor for Carcinoembryonic Antigen Detection
- Smartphone-Enabled Plasmonic ELISA for Ultra‑Sensitive Myoglobin Detection at Point of Care
- High‑Sensitivity Fe³⁺ Detection Using Ag‑Functionalized TiO₂ Nanotube Arrays via Anodic Stripping Voltammetry
- Innovative Magnetic Nanoparticle Contrast Agent for Detecting Cholesterol Deposits in Alzheimer’s Disease
- Enhanced Formaldehyde Detection at ppb Levels Using Reduced Graphene Oxide–Coated Silicon Nanowire Sensors
- Ultra‑Sensitive Biosensing Using 2‑D Hole‑Array Grating‑Coupled Bloch Surface Waves
- Highly Selective Phenanthroline‑Based Fluorescent Probe for Detecting Extreme Alkalinity (pH > 14) in Water
- High-Precision Flexible Temperature Sensors for Human Body Monitoring: A Comprehensive Review



