Enhanced Formaldehyde Detection at ppb Levels Using Reduced Graphene Oxide–Coated Silicon Nanowire Sensors
Abstract
Despite recent advances in low‑concentration formaldehyde monitoring, most gas‑sensor platforms still struggle to achieve reliable ppb‑level sensitivity. In this study, <100>-oriented silicon nanowires (SiNWs) were synthesized via metal‑assisted chemical etching (MACE), yielding high specific surface area structures. These SiNWs were uniformly coated with graphene oxide (GO) and subsequently reduced in an H2/Ar atmosphere at 800 °C to produce reduced graphene oxide (RGO). The resulting RGO@SiNWs (RGO@n‑SiNWs) exhibit a pronounced enhancement in formaldehyde response, attributable to increased surface area, RGO’s sensitization effect, and the formation of a p‑n junction between SiNWs and RGO. Specifically, the sensor delivers a response of 6.4 to 10 ppm HCHO at 300 °C, roughly 2.6 times higher than pristine SiNWs (~2.5). Moreover, RGO@n‑SiNWs achieve a response of 2.4 at 0.1 ppm, the highest concentration relevant to indoor air, with a detection limit of 35 ppb (determined by non‑linear fitting) and rapid response/recovery times of 30 s/10 s. The device also demonstrates superior selectivity against common interferents such as ethanol, acetone, ammonia, methanol, xylene, and toluene, and maintains stability over a 6‑day measurement period. These findings establish RGO‑coated SiNWs as a highly sensitive, selective, and durable platform for indoor formaldehyde monitoring.
Introduction
Formaldehyde (HCHO) is a pervasive volatile organic compound (VOC) in newly constructed indoor environments, posing significant health risks, including sick building syndrome (SBS) and carcinogenicity, as recognized by the International Agency for Research on Cancer (IAIC) [2]. To mitigate these risks, regulatory bodies such as the National Institute for Occupational Safety and Health (NIOSH) and the World Health Organization (WHO) have set stringent indoor air limits of 0.1 ppm (living rooms) and 0.08 ppm (30‑min average) respectively [2,15]. Consequently, the development of portable, real‑time sensors capable of detecting HCHO at the ppb scale is essential for safeguarding indoor air quality.
Conventional analytical methods—liquid chromatography, spectroscopy—are unsuitable for on‑site monitoring due to their bulk and complexity. Semiconductor nanostructure‑based gas sensors (In2O3, SnO2, etc.) offer rapid, low‑cost, and miniaturizable alternatives but typically exhibit limited sensitivity at the ppb level [2,10,19,24–32]. Enhancing sensor performance to meet the practical requirement of an air‑to‑gas resistance ratio (Ra/Rg) of ≥2 remains a critical challenge. Silicon nanowires (SiNWs) are attractive for chemical sensing due to their high surface‑to‑volume ratio, yet their field‑effect‑based devices demand costly, complex fabrication.
Integrating graphene or reduced graphene oxide (RGO) with semiconductor nanostructures has emerged as a promising strategy to boost gas‑sensing performance, leveraging graphene’s high surface area and exceptional electron mobility. Unlike noble‑metal sensitizers, RGO offers low cost, abundant surface sites, and efficient charge transfer. Prior studies have shown RGO‑SnO2 and RGO‑Cu2O composites to markedly improve sensitivity. However, most implementations involve depositing semiconductor particles onto a graphene sheet, limiting the interfacial contact area. Core‑shell architectures—where RGO encapsulates semiconductor nanowires—can maximize the interface, potentially unlocking superior sensitivity.
In this work, we present a core‑shell RGO@SiNW sensor that achieves a response of 6.4 to 10 ppm HCHO at 300 °C, a 2.6‑fold improvement over pristine SiNWs, and a detection limit of 35 ppb. The sensor demonstrates fast dynamics, high selectivity, and excellent stability, thereby advancing the state of the art in low‑concentration indoor formaldehyde detection.
Materials and Methods
Fabrication of SiNWs Arrays
Commercial n‑type (100) and p‑type (100) Si wafers (3.0 cm × 3.0 cm, resistivity 0.005–0.02 Ω·cm and 0.001–0.005 Ω·cm, respectively) were sequentially cleaned in acetone, ethanol, and deionized (DI) water (10 min each). The wafers were then immersed in an oxidant solution (H2SO4 97 % + H2O2 35 %) at a 3:1 volume ratio for 30 min to remove organic residues, followed by 5 % HF dip for 8 min to strip native oxide and produce H‑terminated surfaces. Next, a 0.005 M AgNO3 solution containing 4.8 M HF was used to deposit Ag nanoparticles onto the wafer surfaces for 1 min under gentle stirring. The Ag‑coated wafers were rinsed with DI water to eliminate excess Ag+ ions and then etched in a solution of 0.4 M H2O2 and 4.8 M HF at room temperature for 30 min in the dark. After etching, the Ag catalyst was removed by immersion in 70 % HNO3 and subsequent DI water rinses. The resulting SiNWs were gently scraped from the wafer surface with a sharp blade for further processing.
Functionalization with RGO
Graphene oxide (GO) was prepared via a modified Hummers’ method [40] and dispersed in 60 mL DI water (30 mg) by ultrasonication for 3 h. SiNWs (0.2 g) were dispersed in a mixture of 10 mL DI water and 30 mL ethanol, followed by the addition of 400 µL ethylenediamine. After 20 min of sonication, 20 mL of the GO dispersion was introduced and stirred vigorously. The composite was collected by centrifugation, washed with ethanol, and dried at 60 °C to yield GO@SiNWs. Finally, GO@SiNWs were reduced by annealing in an H2/Ar atmosphere at 800 °C (ramp rate 2 °C min−1) to produce RGO@SiNWs.
Characterization
Scanning electron microscopy (SEM, JSM‑7001F+INCA X‑MAX) and transmission electron microscopy (TEM, JEM‑2100F) were used to examine morphology. X‑ray diffraction (XRD, X’Pert PRO MPD) provided crystallographic information. Nitrogen adsorption–desorption isotherms (SSA‑7300, BUILDER) yielded specific surface area (BET) and pore size (BJH) data. Raman spectroscopy (Thermo Scientific DXR2) confirmed RGO formation, and X‑ray photoelectron spectroscopy (XPS, ESCALAB 250) determined elemental composition and chemical states.
Device Fabrication and Measurement
RGO@SiNWs (≈5 mg) were dispersed in 100 µL ethanol and ultrasonicated to achieve uniform dispersion. The suspension was coated onto a ceramic substrate equipped with Pt heating and measurement electrodes, then aged at 5 V for 3 days in air. Sensor performance was evaluated using a Winsen WS‑30A gas‑sensor analyzer. Formaldehyde was introduced by heating a 40 wt % aqueous solution; ethanol, acetone, ammonia, methanol, xylene, and toluene were generated from pure liquids. The sensor response is defined as Ra/Rg, where Ra and Rg are resistances in air and formaldehyde, respectively. Response/recovery times correspond to the intervals required to reach 90 % of the total response change.
Results and Discussions
SEM and TEM imaging (Figure 1) confirmed that the as‑etched SiNWs are uniform, densely packed, and oriented <100> with diameters ranging from 100 to 300 nm and lengths near 24 µm. The RGO coating (Figure 1g–h) is compact and evenly distributed, establishing a robust p‑n junction between RGO and SiNWs.

a Top view, b zoomed‑in top view, c cross‑section SEM of n‑SiNWs, d cross‑section SEM of p‑SiNWs, e TEM of n‑SiNWs, f HRTEM of n‑SiNWs, g SEM of RGO@n‑SiNWs, h zoomed SEM of RGO@n‑SiNWs.
XRD patterns (Figure 2a) show characteristic Si peaks (111, 200, 400, 331, 422) with no impurities. The RGO@SiNWs pattern displays a reduced peak intensity due to the amorphous RGO layer, and a distinct peak at ~22° confirms successful GO reduction. Raman spectra (Figure 6b) reveal Si–Si peaks at 500 and 912 cm−1 and G/D bands at 1390 and 1590 cm−1, with an ID/IG ratio of 0.72, indicating a well‑reduced graphene structure.
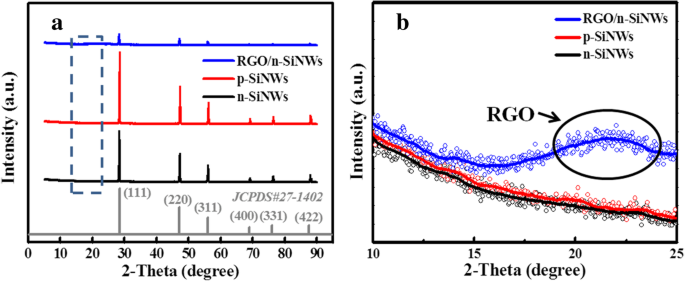
a XRD patterns of n‑/p‑SiNWs and RGO@n‑SiNWs; b Zoomed‑in XRD (10–25°) highlighting the RGO reduction peak.
Electrical testing (Figure 3) demonstrates that pristine n‑SiNWs achieve a response of 2.5 to 10 ppm HCHO at 300 °C, whereas RGO@n‑SiNWs reach 6.4 under the same conditions—a 2.6‑fold enhancement. Dynamic measurements (Figure 3c) show that RGO@n‑SiNWs deliver a response of 2.4 even at 0.1 ppm. Non‑linear fitting (Figure 3d) yields a detection limit of 35 ppb (Ra/Rg = 2), comfortably below indoor safety thresholds.
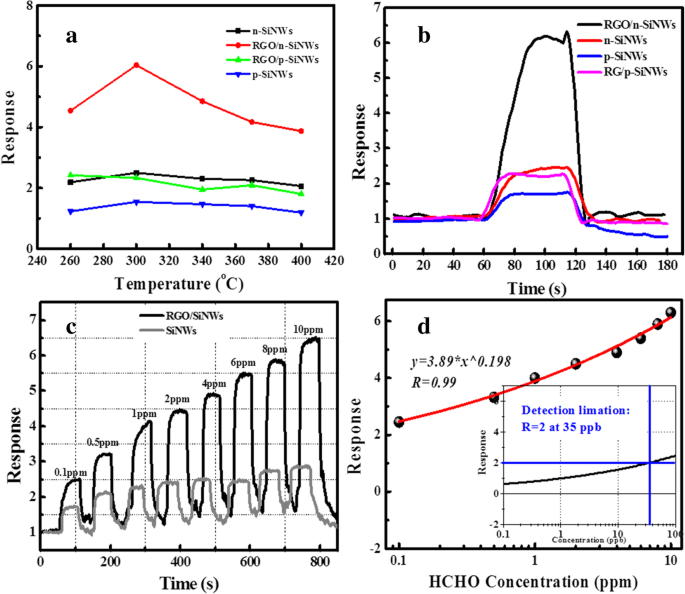
a Responses of n‑/p‑SiNWs, RGO/n‑ and RGO@p‑SiNWs to 10 ppm HCHO at 300 °C; b Response of n‑SiNWs and RGO@n‑SiNWs across temperatures; c Dynamic response from 0.1–10 ppm; d Non‑linear fit for detection limit.
Response dynamics (Figure 4a) indicate rapid detection: 11 s for n‑SiNWs and 13 s for RGO@n‑SiNWs at 0.1 ppm HCHO. Selectivity tests (Figure 4b) confirm minimal interference from ethanol, acetone, ammonia, methanol, xylene, and toluene, attributable to the preferential oxidation of HCHO on the RGO surface. Stability assessments (Figure 5) show <5 % drift over 6 days at 300 °C, evidencing robust long‑term performance.
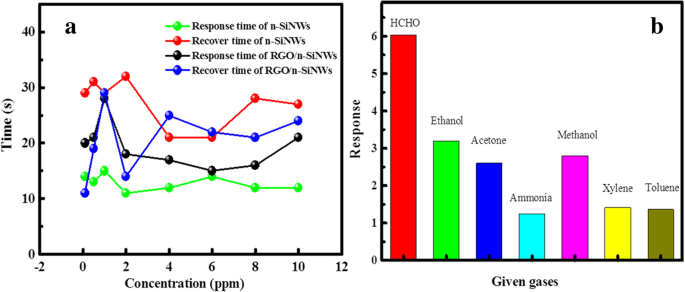
a Response and recovery times for n‑SiNWs and RGO@n‑SiNWs; b Responses to seven common VOCs at 10 ppm.
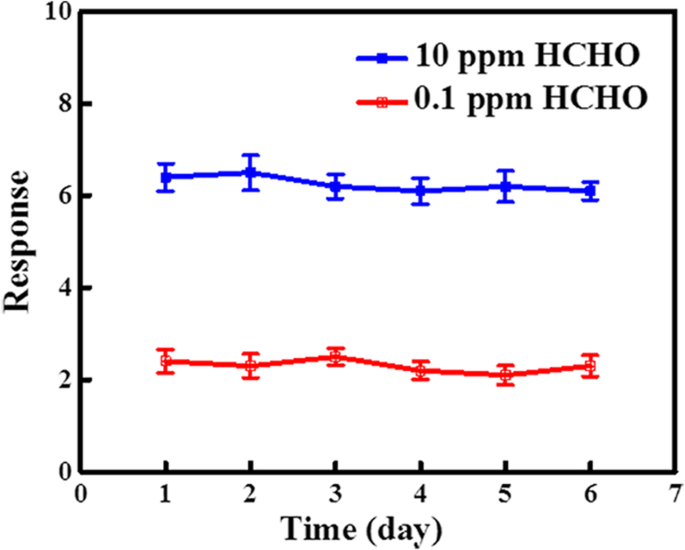
Stability test of n‑SiNWs and RGO@n‑SiNWs for 0.1 ppm and 10 ppm.
BET analysis (Figure 6a) reveals that RGO coating doubles the specific surface area from 37.3 to 74.5 m2 g−1, facilitating greater gas adsorption. XPS (Figure 7) confirms successful RGO coverage: the Si 2p signal diminishes while the C 1s intensity increases, and a binding‑energy shift toward higher energy indicates electron transfer from Si to RGO, consistent with p‑n junction formation.
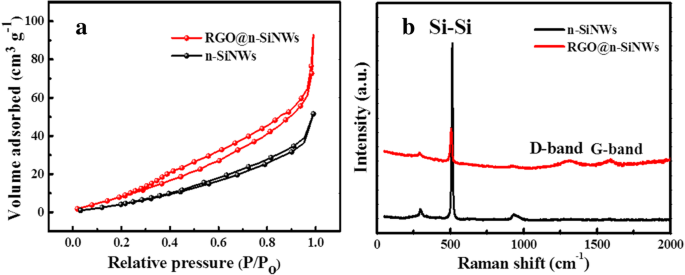
a Nitrogen adsorption isotherms; b Raman shift of n‑SiNWs and RGO@n‑SiNWs.
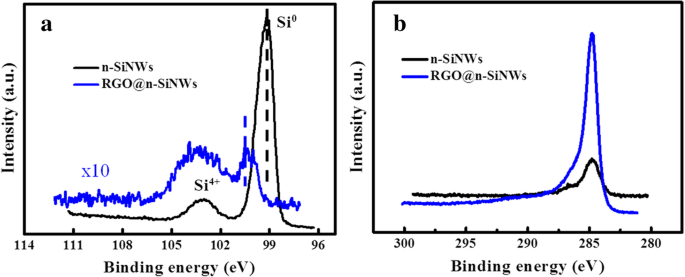
a XPS Si 2p spectra; b XPS C 1s spectra.
The sensing mechanism (Figure 8a) follows the classic chemisorption model: in air, adsorbed O2 captures electrons, forming O− and O2− species and creating a depletion layer. Exposure to HCHO triggers oxidation (HCHO + O− → CO2 + H2O + e−), releasing electrons into RGO and reducing resistance. The p‑n junction between RGO and n‑SiNWs further enhances electron transfer and amplifies the signal, as illustrated in the band‑diagram (Figure 8b).
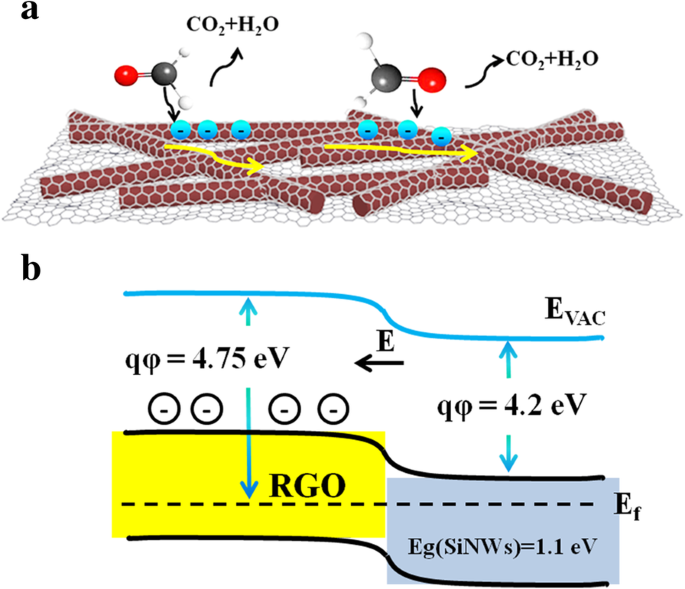
a Schematic of HCHO detection mechanism; b Band structure of the RGO/n‑SiNW interface.
Conclusions
We have demonstrated that metal‑assisted chemical etching produces high‑surface‑area SiNWs, which, when encapsulated with reduced graphene oxide, form a robust p‑n junction that markedly enhances formaldehyde detection. The RGO@n‑SiNW sensor achieves a 2‑fold increase in response to 10 ppm HCHO, a 35 ppb detection limit, and rapid, selective, and stable operation at 300 °C. These attributes make the RGO@SiNW platform a compelling solution for precise indoor formaldehyde monitoring.
Abbreviations
- GO:
Graphene oxide
- HCHO:
Formaldehyde
- HRTEM:
High‑resolution transmission electron microscopy
- IAIC:
International Agency for Research on Cancer
- MACE:
Metal‑assisted chemical etching
- NIOSH:
National Institute for Occupational Safety and Health
- RGO:
Reduced graphene oxide
- RGO@n‑SiNWs:
Reduced graphene oxide‑coated n‑type silicon nanowires
- RGO@SiNWs:
Reduced graphene oxide‑coated silicon nanowires
- SBS:
Sick building syndrome
- SEM:
Scanning electron microscopy
- SiNWs:
Silicon nanowires
- TEM:
Transmission electron microscopy
- VOCs:
Volatile organic compounds
- WHO:
World Health Organization
- XPS:
X‑ray photoelectron spectroscopy
- XRD:
X‑ray diffraction
Nanomaterials
- A Flexible Graphene Biosensor for Rapid, Highly Sensitive Detection of Ovarian Cancer Cells
- Hybrid Graphene/WO₃ and Graphene/CeOx Electrodes for High‑Performance Supercapacitors
- Highly Compressible Graphene/Polyaniline Aerogel: Superelasticity Meets 713 F g⁻¹ Capacitance for All‑Solid‑State Supercapacitors
- Highly Sensitive and Selective Hg²⁺ Detection via FRET between CdSe Quantum Dots and g‑C₃N₄ Nanosheets
- Wireless Magnetoelastic Nanobiosensor Achieves Ultra‑Sensitive Atrazine Detection
- Ultra-Stable Pt–Ni Jagged Nanowire Sensor for Highly Sensitive Caffeic Acid Detection
- Highly Selective Phenanthroline‑Based Fluorescent Probe for Detecting Extreme Alkalinity (pH > 14) in Water
- Ultra‑Sensitive Room‑Temperature CNTs/SnO₂/CuO Sensor for Rapid H₂S Detection
- Ultra‑Sensitive, Selective H₂S Detection with CuO/SnO₂ Thick Films via Precipitation‑Impregnation
- Enhanced Dye Degradation & Antibacterial Performance of Graphene Oxide‑Doped MgO Nanostructures



