Highly Sensitive and Selective Hg²⁺ Detection via FRET between CdSe Quantum Dots and g‑C₃N₄ Nanosheets
Abstract
We engineered a Förster resonance energy transfer (FRET) system that couples CdSe quantum dots (QDs) as donors with g‑C₃N₄ nanosheets as acceptors to achieve rapid, cost‑effective, and highly selective detection of Hg²⁺ in aqueous media. The CdSe QDs/g‑C₃N₄ nanocomposite was fabricated via a simple electrostatic self‑assembly in water, yielding randomly decorated nanosheets with an average QD diameter of ~7 nm. Fluorescence quenching of the donor was linear with Hg²⁺ concentration (0–32 nM) at neutral pH, providing a detection limit of 5.3 nM. The probe discriminated Hg²⁺ from a panel of common metal ions (Na⁺, Mg²⁺, Ca²⁺, Pb²⁺, Cr³⁺, Cd²⁺, Zn²⁺, Cu²⁺) and maintained >92 % of its initial fluorescence after 2 weeks of storage. Real‑water samples (well, lake, tap) were quantified with recoveries between 95.4 % and 101.6 % and relative standard deviations below 2 %. This FRET platform offers a robust, reusable, and environmentally friendly solution for monitoring mercury contamination.
Background
Mercury contamination of natural waters remains the leading source of human exposure to this neurotoxic element. The microbial conversion of Hg²⁺ to methyl‑mercury, a potent neurotoxin, is associated with cognitive and motor disorders worldwide. Rapid, sensitive, and selective detection methods that can operate in complex matrices are therefore essential for environmental monitoring and public health protection.
Nanomaterials with unique optical properties—such as semiconductor quantum dots, fluorescent metal nanoclusters, noble metal nanoparticles, and carbon nanodots—have been extensively explored for mercury sensing. These platforms benefit from facile synthesis, high photostability, functionalization versatility, and biocompatibility. Several FRET‑based sensors employing quantum dots or other nanostructures have been reported; however, a FRET system that couples CdSe QDs with graphitic carbon nitride (g‑C₃N₄) nanosheets for Hg²⁺ detection has not yet been described.
g‑C₃N₄ nanosheets possess a graphene‑like, few‑layered structure, strong visible absorption, and abundant surface amine groups, making them attractive as energy acceptors. By combining these nanosheets with CdSe QDs via electrostatic interactions, we created a compact, highly responsive FRET sensor capable of distinguishing Hg²⁺ from other metal ions.
The sensor’s operating principle is illustrated in Figure 1 (mechanism diagram).
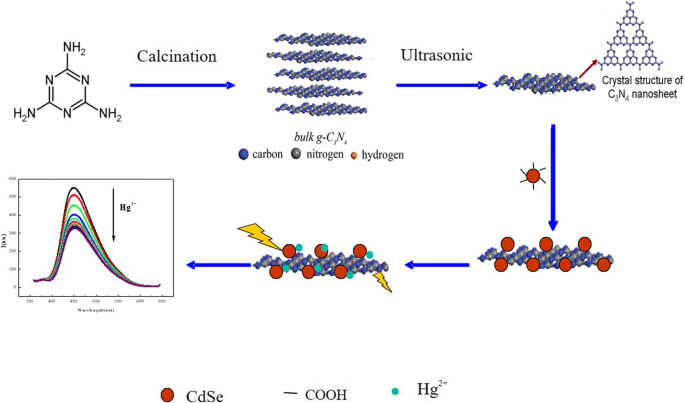
The mechanisms of FRET‑based detection for mercury ions
Methods
Materials
Mercury(II) chloride (HgCl₂) was sourced from Tong Ren Chemical Research Institute (Guizhou, China). Urea and CdSe QDs were purchased from Aladdin Reagent Company (Shanghai, China). All other reagents were of analytical grade and used without further purification. Solutions were prepared with Milli‑Q water (18.2 MΩ cm).
Characterization
X‑ray diffraction (Rigaku D/max‑2400), UV–vis spectroscopy (UV–vis 800), Fourier‑transform infrared (FTIR) spectroscopy (Nicolet‑nexus670), fluorescence spectroscopy (RF‑5301PC), X‑ray photoelectron spectroscopy (Thermo Scientific), and transmission electron microscopy (TEM) were employed to probe the structural and optical properties of the nanocomposite.
Construction of the FRET Sensor
In a typical synthesis, 125 mg of g‑C₃N₄ (pre‑synthesized) were dispersed in 250 mL of water and sonicated for 5 h. Subsequently, 1.838 g of CdSe QDs (0.0216 mol) were added and sonicated for 2 h to promote electrostatic attachment via the amine groups on g‑C₃N₄ and carboxylated QDs. The resulting CdSe QDs/g‑C₃N₄ conjugate was excited at 334 nm (near the QD absorption edge) and its emission recorded at 452 nm.
Fluorescence Detection of Hg²⁺
For each assay, 10 µL of the conjugate was mixed with 3 mL ultrapure water, followed by addition of the desired Hg²⁺ concentration. Fluorescence spectra were acquired after 2 min at room temperature.
Interference and Competition Analyses
Potential interference from Na⁺, Mg²⁺, Ca²⁺, Pb²⁺, Cr³⁺, Cd²⁺, Zn²⁺, and Cu²⁺ (32 nM each) was assessed by measuring the fluorescence at 450 nm under 334 nm excitation. Competition experiments were performed by adding 32 nM Hg²⁺ in the presence of each interferent.
Results and Discussion
Characterization
Transmission electron microscopy (Figure 2a) confirmed the few‑layered, graphene‑like morphology of g‑C₃N₄ nanosheets. X‑ray diffraction (Figure 2b) displayed the characteristic (002) peak at 27.4°, confirming graphitic stacking, while a minor peak at 13.1° indicated in‑plane periodicity. X‑ray photoelectron spectroscopy (Figure 2c) revealed C–C/N bonding at 284.8 and 288.0 eV and N 1s at 397.04 eV. FTIR (Figure 2d) showed triazine ring vibrations at 811 cm⁻¹, CN heterocycle stretches near 1000 cm⁻¹, C–NH–C at 1800 cm⁻¹, and N–H/O–H stretching between 300–3600 cm⁻¹.
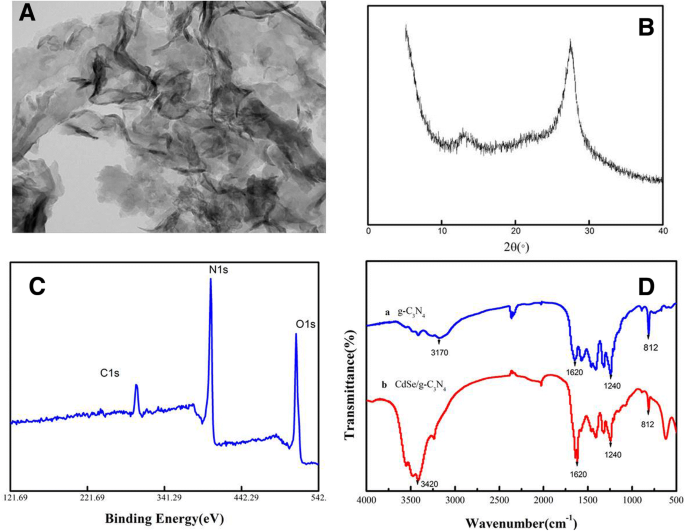
Characterization of the as‑prepared g‑C₃N₄ nanosheet. a TEM image. b XRD pattern. c XPS spectrum. d FTIR spectrum
UV‑vis and Fluorescence Properties
The CdSe QDs/g‑C₃N₄ conjugate exhibited a prominent absorption peak at ~334 nm (Figure 3a). Fluorescence emission peaked at 452 nm upon 334 nm excitation (Figure 3b), a 14–16 nm red‑shift relative to pristine g‑C₃N₄ (438 nm emission, 310 nm excitation) attributable to efficient FRET.
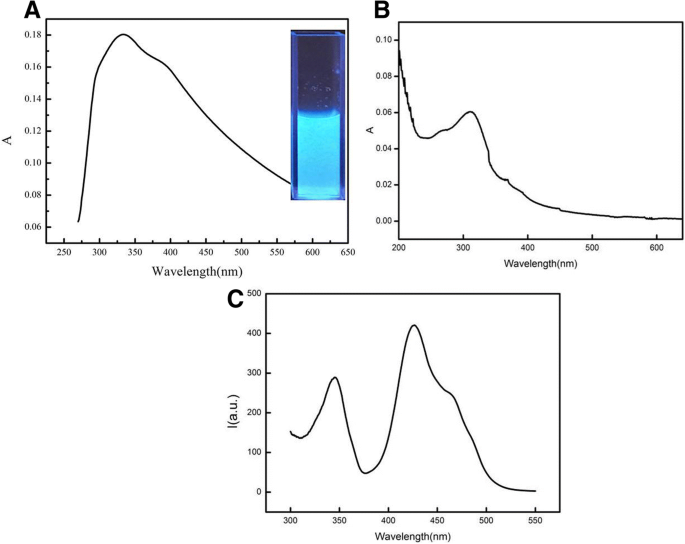
The UV‑vis absorption and fluorescence spectrum of CdSe QDs/g‑C₃N₄ conjugates
Effect of pH
Fluorescence intensity increased from pH 3 to 7 and declined from 7 to 10 (Figure 4), likely due to protonation/deprotonation of surface amine groups affecting surface charge. Neutral pH 7 was chosen for Hg²⁺ sensing, where high ionic strength (NaCl) had negligible impact on fluorescence.
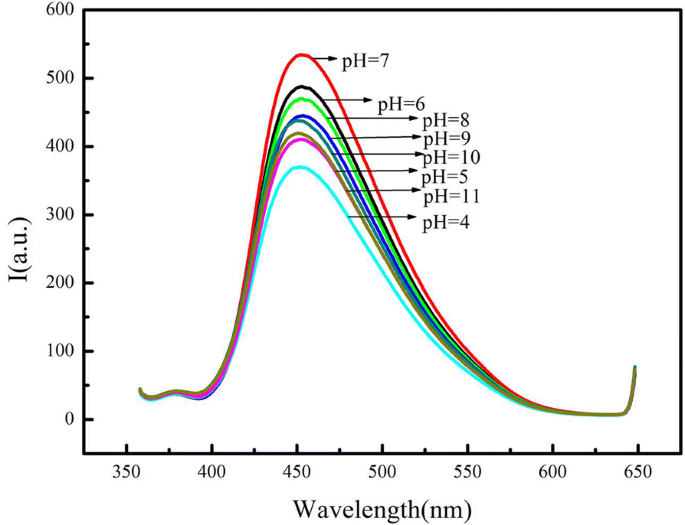
The effect of pH on the fluorescence of the CdSe QDs/g‑C₃N₄ nanosheet conjugate
Selectivity
When exposed to a panel of competing metal ions, the sensor’s fluorescence ratio (I/I₀) increased markedly only in the presence of Hg²⁺ (Figure 5a), whereas other ions produced negligible changes. This selectivity contrasts with pristine g‑C₃N₄, which responds to both Cu²⁺ and Hg²⁺, underscoring the role of CdSe QDs in conferring Hg²⁺ specificity (Figure 5b).
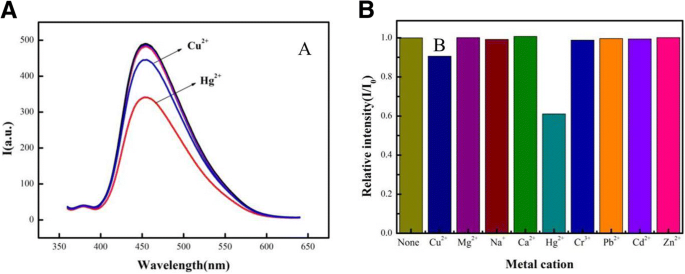
The selectivity experiments for the CdSe QDs/g‑C₃N₄ nanosheet FRET sensor
Feasibility and Sensitivity
Fluorescence quenching increased linearly with Hg²⁺ concentration from 0 to 32 nM (Figure 6a). The I/I₀ ratio followed the equation I = –9.6 × 10⁷ + 550.5 (R² = 0.9882), yielding a detection limit of 5.3 nM. The probe exhibited negligible response to other metal ions, confirming high selectivity. Stability tests showed that after 14 days at ambient conditions, the conjugate retained >92 % of its initial fluorescence.
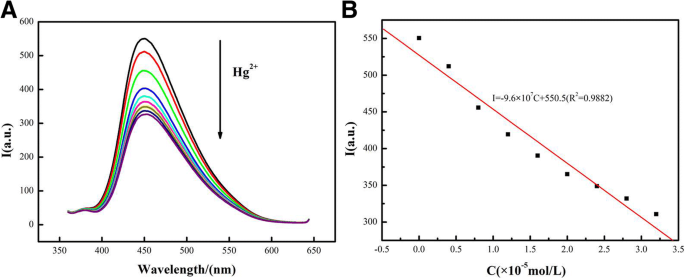
The sensing properties of the CdSe QDs/g‑C₃N₄ nanosheet conjugate and the dependence of fluorescence intensity on Hg²⁺ concentration (0–32 nM)
Co‑existing cations (Na⁺, Mg²⁺, Ca²⁺, Pb²⁺, Cr³⁺, Cd²⁺, Zn²⁺, Cu²⁺) did not interfere with Hg²⁺ detection, as shown in Table 1. Long‑term stability and reproducibility were confirmed through periodic fluorescence measurements over 14 days.
Application to Real Samples
The sensor was applied to lake, tap, and well water spiked with Hg²⁺. Recoveries ranged from 95.4 % to 101.6 % with RSDs between 0.64 % and 1.72 % (Table 3), demonstrating reliable performance in complex matrices.
Conclusions
We have developed a FRET‑based Hg²⁺ sensor that couples CdSe QDs with g‑C₃N₄ nanosheets. The probe offers a 5.3 nM detection limit, a linear range of 0–32 nM, and exceptional selectivity against common metal ions. Its simple, aqueous‑phase synthesis, high photostability, and low cost make it well‑suited for environmental monitoring and potentially for biomedical applications.
Highlights
- Construction of a FRET system between CdSe quantum dots and g‑C₃N₄ nanosheets for Hg²⁺ detection.
- Synthesis of CdSe QDs/g‑C₃N₄ nanocomposites via straightforward electrostatic self‑assembly in water.
- Demonstrated linear fluorescence quenching of Hg²⁺ (0–32 nM) at pH 7 with a detection limit of 5.3 nM.
- Proposed FRET platform provides a versatile, reusable assay for quantifying Hg²⁺ in complex water samples.
Abbreviations
- FRET:
Förster resonance energy transfer
- FTIR:
Fourier‑transform infrared
- UV–vis:
Ultraviolet–visible
- XPS:
X‑ray photoelectron spectroscopy
- XRD:
X‑ray diffractometer
Nanomaterials
- Water‑Soluble Antimony Sulfide Quantum Dots: Efficient Synthesis and Superior Photo‑Electric Performance
- Highly Sensitive Nonenzymatic Glucose Sensing with Hollow Porous Nickel Oxide
- Impact of Shell Thickness on Energy Transfer in Eu‑Doped Core–Shell ZnSe/ZnS Quantum Dots
- Blue‑Emitting Mn‑Alloyed CdTe Quantum Dots: Synthesis, Structural Insight, and Electrochemical Characterization
- High‑Performance Pr2CuO4 Nanosheet Adsorbent: Controlled Synthesis, Superior Selectivity for Malachite Green, and Mechanistic Insights
- Wireless Magnetoelastic Nanobiosensor Achieves Ultra‑Sensitive Atrazine Detection
- Enhanced Formaldehyde Detection at ppb Levels Using Reduced Graphene Oxide–Coated Silicon Nanowire Sensors
- High‑Quality In₂S₃ Quantum Dots Synthesized at Room Temperature for Ultra‑Sensitive UV Photodetectors
- Advanced PAMAM‑Quantum Dot Complexes for Rapid Clinical Immunoassays
- Ultra‑Sensitive, Selective H₂S Detection with CuO/SnO₂ Thick Films via Precipitation‑Impregnation



