Highly Sensitive Nonenzymatic Glucose Sensing with Hollow Porous Nickel Oxide
Abstract
Transition‑metal oxides (TMOs) are attractive electrocatalysts due to their low cost and chemical robustness, yet their intrinsic activity often falls short of demanding applications. Introducing a hollow‑porous architecture has emerged as an effective way to enhance electrocatalytic performance by increasing the specific surface area, providing ordered diffusion pathways, and shortening electron‑transfer distances. In this study, we fabricated a cubic hollow‑porous NiO (NiO HPA) through a coordinating‑etching‑and‑precipitating (CEP) strategy followed by calcination. When applied as a glucose sensor, the NiO HPA electrode achieved a remarkable sensitivity of 1323 µA mM⁻¹ cm⁻² and an exceptionally low detection limit of 0.32 µM. These outstanding metrics stem from the material’s large surface area, well‑defined voids, and rapid electron transfer. The results demonstrate that NiO HPA is a viable platform for nonenzymatic glucose monitoring, and the hollow‑porous design offers a general nano‑engineering route for high‑performance electrocatalysts.
Background
Accurate, rapid, and affordable glucose determination is critical across clinical biochemistry, pharmaceutical analysis, food safety, and environmental monitoring. Among the available techniques, electrochemical detection stands out for its high sensitivity, low cost, and low detection limits. Conventional glucose oxidase‑based sensors, however, are limited by enzyme instability. Earth‑abundant TMOs have been proposed as alternatives, offering cost effectiveness, structural stability, and electrochemical activity. Nonetheless, conventional TMO electrodes still lack the catalytic efficiency required for practical deployment. Therefore, rational design of TMO electrocatalysts remains a pressing challenge.
The catalytic kinetics of an electrode are tightly linked to its micro‑architecture. Enhancing surface area, tailoring pore structures, and engineering hierarchical architectures can dramatically improve activity by providing abundant active sites, efficient mass transport, and short electron‑transfer pathways. Hollow structures, in particular, combine a functional shell with an internal void, increasing electrolyte contact and mitigating volume expansion during repeated measurements. By integrating these features, a hollow‑porous architecture can transform a conventional TMO into a highly active electrocatalyst.
Nickel oxide is a prototypical TMO that can catalyze glucose oxidation via the Ni²⁺/Ni³⁺ redox couple in alkaline media, making it an attractive candidate for glucose sensing. Here, we report a cubic NiO HPA fabricated by a Cu₂O‑templated CEP method followed by calcination. Compared to a broken‑hollow counterpart (NiO BHPA), the NiO HPA exhibits superior sensitivity and lower detection limits, underscoring the value of the hollow‑porous design. This facile synthesis strategy provides a practical route to high‑performance electrochemical sensors.
Experimental
Materials
All reagents were analytical grade: CuCl₂·2H₂O, NiCl₂·6H₂O, Na₂S₂O₃·5H₂O, polyvinylpyrrolidone (PVP, M = 40 000), and NaOH from Chengdu Kelong. Glucose, lactose, sucrose, fructose, L‑ascorbic acid, uric acid, and a 5 wt % Nafion solution (alcohol/water mixture) were purchased from Sigma‑Aldrich without further purification.
Synthesis of Cu₂O Template
Cu₂O cubes were prepared following our prior protocol. Briefly, 20 mL of 2 M NaOH was added dropwise to 200 mL of 10 mM CuCl₂·2H₂O at 55 °C under stirring. After 30 min, 4 mL of 0.6 M ascorbic acid was introduced dropwise. The mixture was aged for 3 h, then washed repeatedly by centrifugation. XRD, SEM, and TEM analyses are provided in Supplementary Figure S1.
Synthesis of NiO HPA
NiO HPA was synthesized via CEP. 10 mg of Cu₂O and 3 mg of NiCl₂·6H₂O were dispersed in 10 mL of ethanol‑water (1:1) and sonicated for 7 min. 0.33 g PVP was added under vigorous stirring for 30 min. Then, 4 mL of 1 M Na₂S₂O₃ was added; the reaction proceeded at room temperature for 3 h, turning from red to light green, indicating Ni(OH)₂ formation. The precipitate was washed with warm ethanol‑water and dried. Calcination at 400 °C (1 °C min⁻¹) for 2 h in air yielded NiO HPA. NiO BHPA was produced by subjecting NiO HPA to 2 h of intense sonication.
Material Characterizations
XRD (Rigaku D/Max‑2400) confirmed phase purity. XPS (ESCALAB250Xi) used C 1s at 284.8 eV as reference. Morphologies were examined by FESEM (FEI Quanta 250, Zeiss Gemini 500) and HR‑TEM (FEI F20). BET surface area and pore structure were measured with a Belsort‑max analyzer.
Electrochemical Measurements
All tests were conducted in 0.1 M NaOH using a three‑electrode setup: NiO HPA (or BHPA) modified glassy carbon electrode (GCE, Ø 3 mm) as working, Ag/AgCl (sat. KCl) as reference, and Pt disk (Ø 2 mm) as counter. The GCE was polished sequentially with 3 µm, 0.5 µm, and 0.05 µm alumina slurries. A 10 mg NiO HPA suspension in 0.1 mL Nafion + 0.9 mL water was drop‑cast onto the GCE (70.77 µg cm⁻²) and dried. Cyclic voltammetry (CV), chronoamperometry (CA), and electrochemical impedance spectroscopy (EIS) were performed on an Autolab potentiostat. EIS was recorded from 0.01–100 kHz with 5 mV perturbation.
Results and Discussion
Characterizations
XRD patterns (Fig. 1a) display peaks at 37.21°, 43.27°, 62.87°, and 75.42° corresponding to the (111), (200), (220), and (311) planes of face‑centred cubic NiO (JCPDS #47‑1049), confirming phase purity. XPS survey spectra (Fig. 1b) reveal O 1s and Ni 2p signals at 531.5 eV and 855.7 eV, respectively. The Ni 2p spectrum (Fig. 1c) shows characteristic doublet peaks at 855.8 eV (Ni 2p₃/₂) and 873.5 eV (Ni 2p₁/₂) with a 17.7 eV separation, indicative of NiO. Satellite peaks appear at 861.5 eV and 880.0 eV. O 1s deconvolution (Fig. 1d) identifies Ni–O (529.8 eV), chemisorbed oxygen/hydroxyls (831.3 eV), and adsorbed water (532.7 eV). These analyses confirm the successful synthesis of NiO HPA.
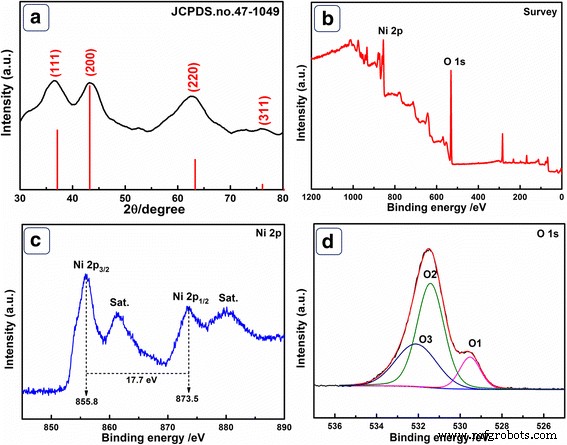
a XRD pattern of NiO HPA. b XPS survey, c Ni 2p, and d O 1s spectra.
SEM and TEM images (Fig. 2) reveal uniform cubic NiO HPA with an edge length of ~600 nm and a shell thickness of ~40 nm. The hollow morphology is confirmed by the translucent interior and the line‑scan EDX profile (Fig. 2h) showing a pronounced drop in elemental concentration at the core. HRTEM (Fig. 2f) resolves lattice fringes of 0.21 nm (200) and 0.24 nm (111), matching XRD results. SAED patterns confirm the cubic crystal structure. Elemental mapping (Fig. 2g) shows homogeneous distribution of Ni and O.

a–c SEM images, d–e TEM images, f HRTEM, g STEM‑EDX mapping, and h EDX line scan of NiO HPA.
Time‑resolved TEM (Fig. 3) and optical imaging (Fig. 3b) illustrate the CEP mechanism: Cu₂O corners are preferentially etched by S₂O₃²⁻, generating Cu²⁺–S₂O₃ complexes and liberating OH⁻. The released OH⁻ precipitates Ni²⁺ as Ni(OH)₂, forming a shell that encapsulates the shrinking Cu₂O core. Controlled synchronization of etching and precipitation yields a well‑defined hollow Ni(OH)₂ precursor, which converts to NiO HPA upon calcination.
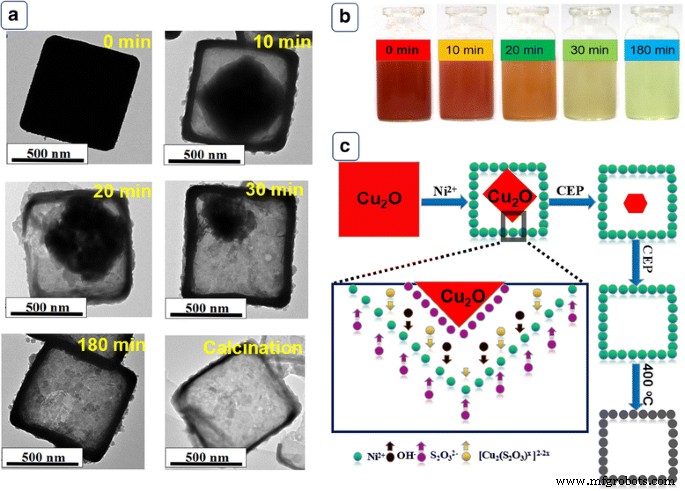
a TEM at various times, b optical photos, c CEP schematic.
BET analysis (Fig. 4) shows NiO HPA possesses a specific surface area of 27.08 m² g⁻¹ and a pore volume of 0.087 cm³ g⁻¹, with a pronounced pore size distribution centered at ~7 nm—indicative of ordered inter‑particle channels. In contrast, NiO BHPA has a drastically reduced SSA (5.24 m² g⁻¹) and negligible pore distribution due to structural collapse from sonication. These differences directly influence electrocatalytic performance.
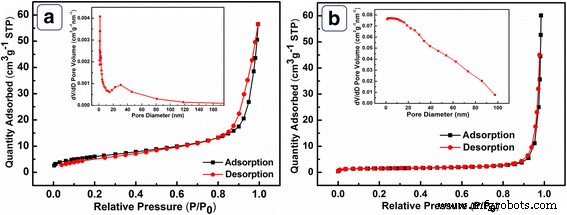
a N₂ adsorption–desorption for NiO HPA, b for NiO BHPA. Inset: pore size distribution.
Electrochemical Performance
CVs (Fig. 5a) reveal distinct Ni²⁺/Ni³⁺ redox peaks at 0.48 V and 0.38 V for the NiO HPA electrode. The peak current is markedly higher than that of NiO BHPA, reflecting the higher SSA and intact hollow architecture. Glucose addition produces a clear catalytic response on both electrodes, but NiO HPA shows a higher current and a lower onset potential (0.43 V vs. 0.46 V). The glucose oxidation mechanism involves the Ni²⁺/Ni³⁺ couple: NiO → Ni²⁺ + O²⁻, Ni²⁺ + OH⁻ → Ni³⁺ + e⁻, Ni³⁺ + glucose → Ni²⁺ + gluconic acid.
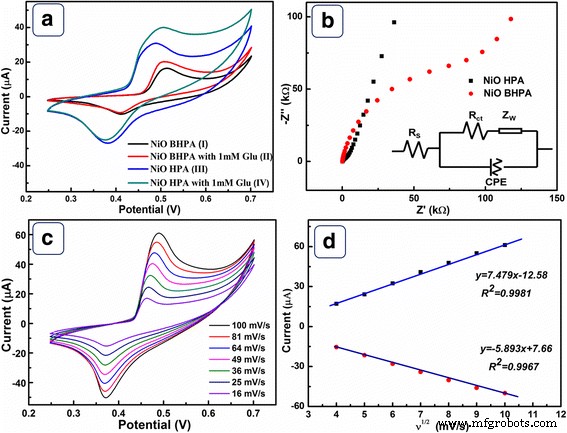
a CVs of NiO BHPA (I, II) and NiO HPA (III, IV) in 0.1 M NaOH with/without 1 mM glucose. b EIS Nyquist plots and equivalent circuit. c CVs at varying scan rates; d peak current vs. √scan rate.
EIS analysis (Fig. 5b) shows NiO HPA has a lower solution resistance (R_s) and charge‑transfer resistance (R_ct) than NiO BHPA, confirming faster electron kinetics. The Warburg region indicates superior diffusion in NiO HPA, attributable to its ordered pores. Kinetic studies (Fig. 5d) confirm a diffusion‑controlled process, with no significant peak shift, further evidence of efficient mass transport.
Selectivity, Reproducibility, and Stability
Potential optimization (Fig. 6a) identified 0.6 V as the optimal operating voltage, maximizing glucose response while minimizing ascorbic acid interference. CA curves (Fig. 6c) demonstrate a linear response from 0.32 to 1100 µM with a sensitivity of 1323 µA mM⁻¹ cm⁻² and an LOD of 0.32 µM. NiO BHPA shows 753 µA mM⁻¹ cm⁻² sensitivity and 14.2 µM LOD. Compared with literature (Table 1), NiO HPA offers superior performance.
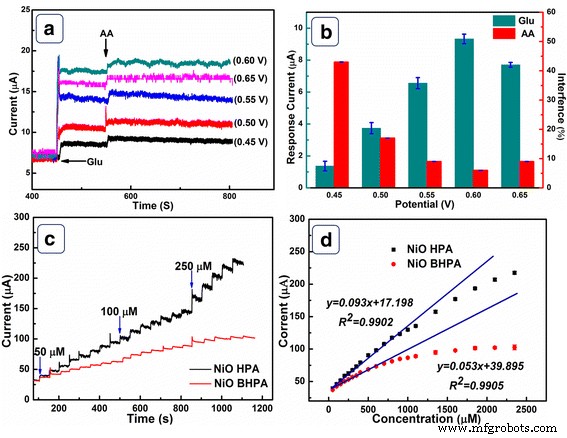
a Amperometric response at different potentials. b Current vs. potential for glucose and ascorbic acid. c CA at 0.6 V for successive glucose additions. d Current vs. glucose concentration.
Interference studies (Fig. 7a) show negligible responses to lactose, sucrose, fructose, and uric acid; ascorbic acid produces only 8.7 % interference. The negative surface charge of NiO HPA in alkaline media repels negatively charged interferents, enhancing selectivity. Long‑term stability tests (Fig. 7b) reveal 83 % retention after 30 days and 9.8 % loss after 2000 s of continuous operation. Five independently fabricated electrodes show an RSD of 3.12 % for 0.1 mM glucose, and repeated measurements on a single electrode yield an RSD of 2.36 %, confirming excellent reproducibility.
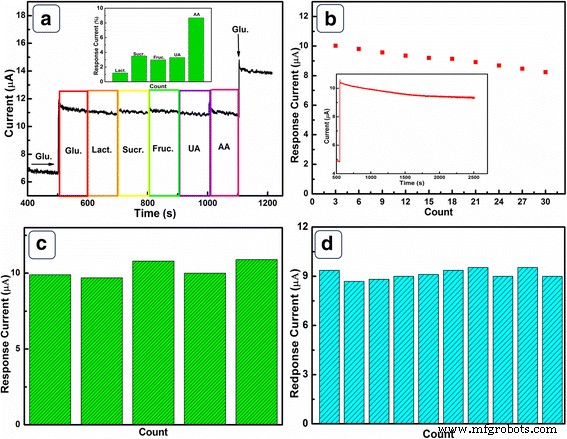
a Sequential addition of 50 µM glucose and 5 µM interferents at 0.6 V. b Long‑term stability and runtime stability. c Five‑electrode reproducibility. d Ten‑measurement reproducibility.
Detection in Human Serum
NiO HPA was applied to quantify glucose in diluted human serum. The sensor achieved a relative standard deviation of 2.85 % and recovery rates between 92 % and 102 %, demonstrating practical applicability and alignment with clinical standards.
Conclusions
We have successfully fabricated a hollow‑porous NiO electrocatalyst via a CEP route that delivers exceptional glucose sensing performance: a sensitivity of 1323 µA mM⁻¹ cm⁻², an LOD of 0.32 µM, < 8.7 % interference, and > 83 % stability over 30 days. The hollow‑porous architecture provides abundant active sites, ordered diffusion pathways, and short electron‑transfer distances, collectively boosting electrocatalytic kinetics. These findings underscore the hollow‑porous design as a general strategy for developing low‑cost, high‑performance nonenzymatic glucose sensors.
Nanomaterials
- Highly Sensitive Non‑Enzymatic Glucose Sensor Based on Mesoporous NiO Nanopetals Grown on FTO
- Highly Sensitive and Selective Hg²⁺ Detection via FRET between CdSe Quantum Dots and g‑C₃N₄ Nanosheets
- Ni(OH)₂ Hollow‑Porous Architecture Enables Ultra‑Sensitive, Enzyme‑Free Glucose Detection
- Wireless Magnetoelastic Nanobiosensor Achieves Ultra‑Sensitive Atrazine Detection
- Ultra-Stable Pt–Ni Jagged Nanowire Sensor for Highly Sensitive Caffeic Acid Detection
- Surfactant‑Free Cu₂O@Au Hollow Cubes Deliver Enhanced CO₂ Electroreduction to CO
- Tin Disulfide Nanoflake-Based Electrochemical Biosensor for Rapid, Sensitive Detection of Salivary Cortisol
- Ultra‑Sensitive NiO/β‑Ga₂O₃ Heterojunction UV Photodetector for Advanced Applications
- Dual‑Shell CuS Nanocages: Enhanced Electrocatalysis for Ultra‑Sensitive Ascorbic Acid Detection
- MoS₂ Nanocomposite-Based Enzyme-Free Glucose Biosensors: A Cutting-Edge Approach



