Surfactant‑Free Cu₂O@Au Hollow Cubes Deliver Enhanced CO₂ Electroreduction to CO
Abstract
We report surfactant‑free, low‑Au‑loading Cu₂O@Au and Au hollow cubes fabricated by galvanic replacement of electrodeposited Cu₂O cubes. The Cu₂O@Au composite delivers a Faradaic efficiency (FE) of 30.1 % for CO at –1.0 V vs. RHE—approximately twice the FE obtained with either Cu₂O or Au alone—underscoring the catalytic advantage of the Cu₂O/Au metal‑oxide interface.
Background
Carbon dioxide (CO₂) is the principal greenhouse gas driving global warming, and its mitigation through capture, storage, or conversion into value‑added chemicals has become a global priority [1, 2]. Conventional CO₂ removal methods—such as underground sequestration [3, 4]—are energy intensive, whereas electrochemical reduction (CO₂RR) offers a promising route to produce fuels and chemicals under ambient conditions [8–10]. Yet, CO₂RR faces intrinsic challenges: the molecule’s chemical inertness, the relatively low thermodynamic driving force, and poor product selectivity [11]. Consequently, the design of catalysts that combine high activity, selectivity, and stability remains a critical research frontier.
Various metals have been explored for CO₂RR, including Au, Ag, Cu, Pd, and Sn [12]. Copper uniquely enables C₁–C₃ hydrocarbons and alcohols, whereas gold yields CO with high selectivity and low overpotential [11]. However, copper’s selectivity and durability can be limited, and gold’s high cost hampers industrial deployment [18, 19]. Bimetallic Cu–Au composites therefore hold promise, but most reported systems suffer from uncontrolled morphology, aggregation, and surface oxidation [20–22]. Additionally, the metal‑oxide interface has emerged as a powerful strategy to enhance CO₂RR activity [23].
In this study, we synthesize a surfactant‑free Cu₂O@Au nanocomposite that incorporates a well‑defined Cu₂O/Au interface for CO₂RR. A comparison with hollow cubic Au obtained by selectively removing Cu₂O demonstrates the pivotal role of the metal‑oxide boundary in activating CO₂ and boosting CO FE.
Methods
Materials
Copper(II) trifluoroacetate (Cu(TFA)₂, 98 %), potassium trifluoroacetate (KTFA, 98 %), and chloroauric acid (HAuCl₄, 99.9 %) were purchased from Sigma‑Aldrich and used without further purification. All solutions were prepared in Milli‑Q ultrapure water (≥ 18.2 MΩ cm). Nitrogen (N₂, 99.999 %) and CO₂ (99.999 %) gases were sourced from Foshan MS Messer Gas CO., Ltd. Carbon paper (0.3 mm thick) was obtained from Hesen, Shanghai.
Preparation of Cu₂O Nanocubes and Cu₂O@Au
Cu₂O nanocubes were synthesized following established protocols [24] and electrodeposited onto 1 cm × 1 cm carbon paper at –0.06 V (vs. SCE) for 1 h in 10 mM Cu(TFA)₂ and 0.2 M KTFA. Prior to deposition, the carbon paper was rinsed with water and ethanol. The Cu₂O@Au composite was formed by immersing the Cu₂O cubes in 2 mL 1 mM HAuCl₄ for 30 min at 277 K.
Preparation of Hollow Cubic Au
Selective removal of Cu₂O was achieved by treating the Cu₂O@Au composite in 2 M aqueous ammonia for 12 h at 277 K, leaving a hollow cubic Au framework on the carbon paper.
Characterization
Morphology and elemental distribution were examined by scanning electron microscopy (SEM, JEOL‑6701F) equipped with an energy‑dispersive X‑ray (EDX) detector. X‑ray diffraction (XRD) patterns were recorded on a Rigaku Ultima IV diffractometer (Cu Kα, λ = 1.5406 Å) to confirm phase composition.
Electrochemical Measurements of CO₂
All electrochemical experiments were performed on a CH Instruments 760D system in a three‑electrode configuration. The CO₂RR cell was a two‑compartment H‑type cell, with an Ag/AgCl reference and a platinum counter electrode. 85 % iR compensation was applied, and potentials were referenced to the reversible hydrogen electrode (RHE) using the relation E_RHE = E_Ag/AgCl + 0.197 V + 0.059 V × pH [25]. The cell was separated by a Nafion 117 proton exchange membrane.
Linear sweep voltammetry (LSV) was carried out in 0.1 M KHCO₃ under N₂ or CO₂ saturation. Prior to LSV, the electrolyte was purged with the respective gas for 20 min. CO₂RR was conducted under potentiostatic control; current density and product distribution were monitored over time. Gas products were quantified by online gas chromatography (GC Agilent 7890B) equipped with a thermal conductivity detector (TCD) and flame ionization detector (FID). Liquid products were negligible and thus omitted from analysis. CO₂ was continuously fed into the cathodic cell at 20 mL min⁻¹.
The FE for each product was calculated using equations (1) and (2) as described in the manuscript, where i_total is the measured total current density.
Results and Discussion
Morphology
SEM images (Fig. 2) reveal that the electrodeposited Cu₂O cubes exhibit a regular cubic shape with a smooth surface (Fig. 2a–b). The average edge length is ~1 µm. Subsequent galvanic replacement yields Cu₂O@Au nanostructures that retain the cubic morphology while incorporating uniformly distributed Au nanoparticles on the surface (Fig. 2c–d). EDX mapping (Fig. 2e–f) confirms the homogeneous distribution of Cu and Au, consistent with the GRR mechanism that produces an internal hollow core and surface Au precipitation [27, 28].

The SEM images of Cu₂O nanocubes (a, b), Cu₂O@Au nanoparticles (c, d), and EDX of Cu₂O@Au nanoparticles (e, f)
After ammonia treatment, the Cu₂O core is removed, leaving a hollow cubic Au skeleton that preserves the original cubic frame (Fig. 3). The resulting Au nanoparticles within the hollow framework have diameters of 20–30 nm.

The SEM images of hollow cubic Au (a–c) of different magnification
XRD Analysis
XRD patterns (Fig. 4) confirm the crystalline phases of the catalysts. Peaks at 2θ = 36.46°, 42.36°, 61.44°, and 73.55° correspond to the (111), (200), (220), and (311) planes of Cu₂O (JCPDS 78‑2076). The hollow Au exhibits weak peaks at 2θ = 38.18°, 44.39°, 64.57°, and 77.54°, assigned to the (111), (200), (220), and (311) planes of Au (JCPDS 04‑0784). In the Cu₂O@Au composite, the Cu₂O peaks diminish, indicating partial replacement by Au.
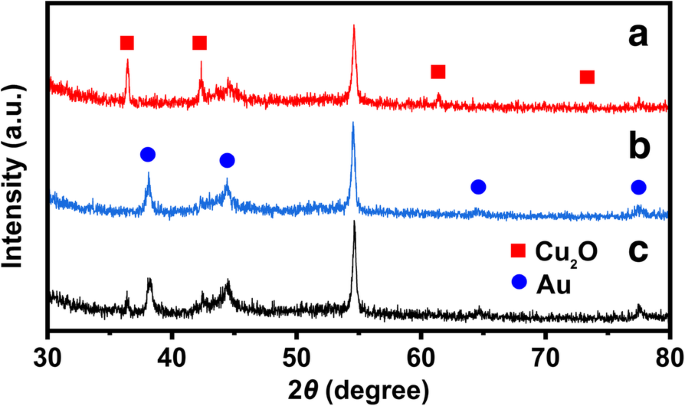
XRD patterns of (a) Cu₂O cube, (b) hollow cubic Au, and (c) Cu₂O@Au
CO₂ Electrochemical Reduction Performance
LSV curves (Fig. 5) show that under CO₂ saturation, the Cu₂O@Au electrode delivers the highest cathodic current density among the three catalysts, reflecting enhanced CO₂ adsorption and electron transfer. The Cu₂O@Au electrode also exhibits a lower HER contribution, as evidenced by the reduced hydrogen current relative to CO production.
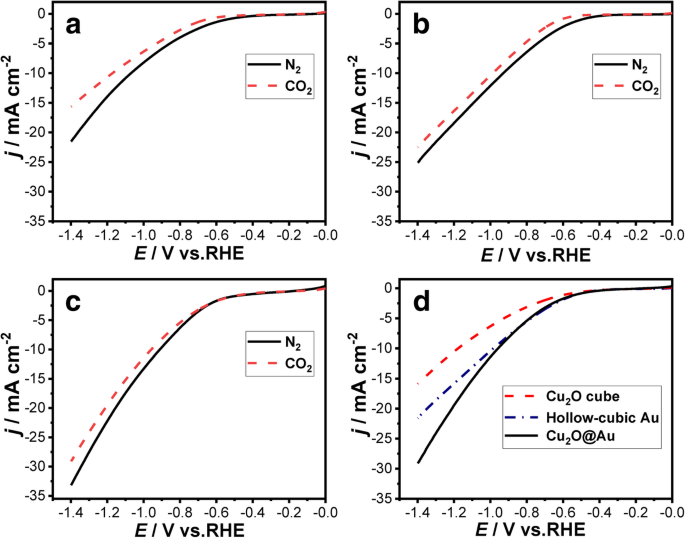
LSV curves obtained on a Cu₂O cube, b hollow cubic Au, and c Cu₂O@Au electrodes in N₂‑saturated (black solid line) and CO₂‑saturated (red dotted line) 0.1 M KHCO₃ solutions. d LSV curves of the three samples in CO₂‑purged 0.1 M KHCO₃ solutions
Time‑resolved amperometric measurements (i–t) were performed between –0.7 and –1.2 V vs. RHE. The FE of CO for Cu₂O@Au peaks at 30.1 % at –1.0 V, roughly double that of hollow Au (16.3 %) and Cu₂O alone (≈ 15 %). H₂ FE decreases as the potential becomes more negative, indicating effective HER suppression by CO coverage. The CO₂RR products of the three catalysts are summarized in Fig. 6.
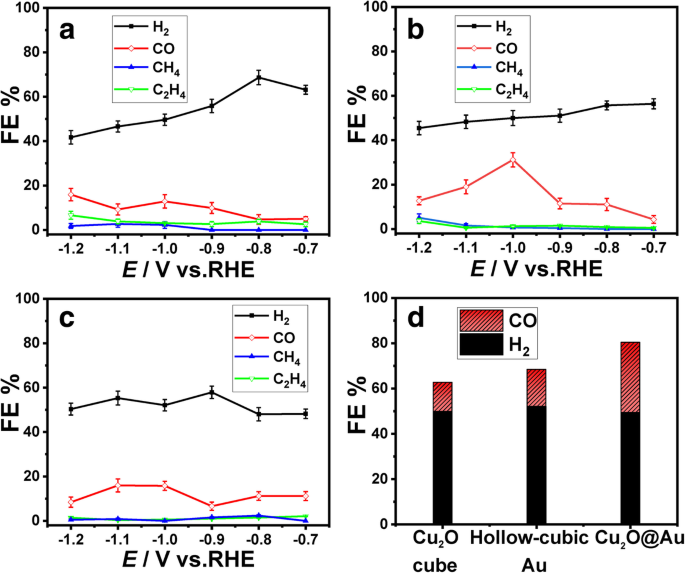
FE of a Cu₂O cube catalyst, b Cu₂O@Au catalyst, and c hollow cubic Au catalyst. d Comparison of FE for CO and H₂ at −1.0 V vs RHE on three catalysts
The superior performance of Cu₂O@Au can be rationalized by the Cu₂O/Au interface. The interface lowers the binding energy of the CO₂* intermediate (step 3 in Eq. 3), facilitates the formation of COOH*, and enhances the subsequent reduction to CO (steps 4–5). These effects collectively improve CO₂ adsorption, charge transfer, and CO FE, as reported for other metal‑oxide interfaces [23, 31, 32].
At –1.0 V vs. RHE, the H₂/CO ratio for Cu₂O@Au is 1.7, close to the syngas ratio of 2 favored in Fischer–Tropsch synthesis, highlighting its potential for syngas production.
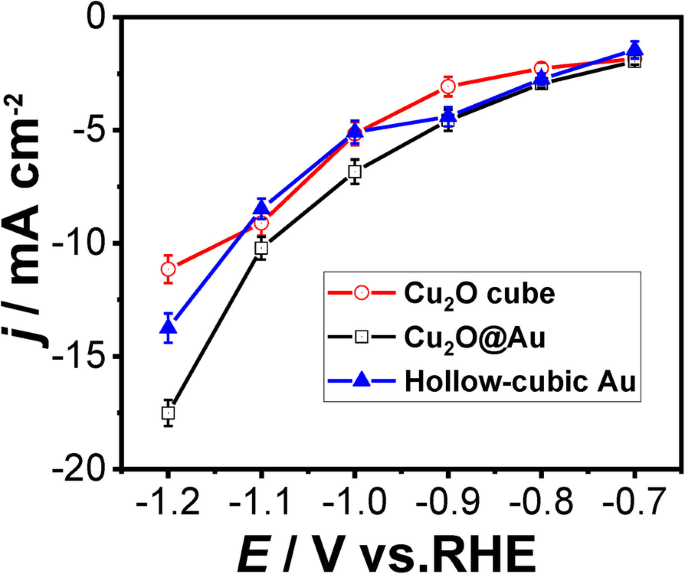
The average total current density of the three catalysts for CO₂ reduction at different potentials
Conclusions
We have demonstrated that a surfactant‑free Cu₂O@Au hollow cube catalyst, prepared via electrodeposition and galvanic replacement, outperforms both Cu₂O and hollow Au in CO₂RR. The Cu₂O/Au metal‑oxide interface activates CO₂, enabling a maximum CO FE of 30.1 % at –1.0 V vs. RHE. The resulting H₂/CO ratio of 1.7 approaches the ideal syngas composition for Fischer–Tropsch synthesis. These findings confirm that engineering metal‑oxide interfaces is an effective strategy for designing high‑performance CO₂RR catalysts.
Abbreviations
- CO:
Carbon monoxide
- CO₂
Carbon dioxide
- CO₂RR
CO₂ reduction reaction
- EDX
Energy dispersive X‑ray
- GC
Gas chromatography
- GRR
Galvanic replacement reaction
- HCOO⁻
Formate
- HER
Hydrogen evolution reaction
- LSV
Linear sweep voltammetry
- N₂
Nitrogen
- RHE
Reversible hydrogen electrode
- SEM
Scanning electron microscopy
- XRD
X‑ray diffraction
Nanomaterials
- Cubic Zirconia: Production, Quality, and Market Outlook
- Fibre Laser vs CO₂ Laser: Choosing the Right Cutting Technology for Your Business
- A Biomimetic Iron Catalyst Turns CO₂ into Methane Using Visible Light
- Enhanced Visible‑Light Photocatalysis of Methylene Blue and Phenol with N‑Doped ZnO/g‑C₃N₄ Nanocomposites
- Highly Sensitive Nonenzymatic Glucose Sensing with Hollow Porous Nickel Oxide
- Enhanced Photocatalytic Degradation of Rhodamine B Using SrTiO3/Bi5O7I Nanocomposites: Fabrication, Characterization, and Mechanistic Insights
- Efficient Synthesis of CNT–Cu₂O Nanocomposites for Rapid p‑Nitrophenol Reduction: A Low‑Cost, Reusable Catalyst
- Tin Disulfide Nanoflake-Based Electrochemical Biosensor for Rapid, Sensitive Detection of Salivary Cortisol
- MoS₂ Nanocomposite-Based Enzyme-Free Glucose Biosensors: A Cutting-Edge Approach
- Compressed CO2 vs Compressed Air: Key Differences & Practical Implications



