Bisulfate and Sulfate Poisoning of PtCo Nanocatalysts: Effects on Oxygen Reduction Reaction Kinetics
Abstract
Pt‑based alloys are widely adopted in polymer electrolyte membrane fuel cells (PEMFCs) due to their superior oxygen reduction reaction (ORR) activity. However, anion adsorption—particularly from bisulfate (HSO4−) and sulfate (SO42−)—can markedly diminish performance. In this study, we employ rotating disk electrode (RDE) techniques to quantify the influence of bisulfate and sulfate ions on the ORR activity of a commercial 30 wt.% PtCo nanoparticle catalyst. We observe a linear decline in specific activity with the logarithm of anion concentration across a range of high potentials, indicating that adsorption does not alter the free‑energy landscape of ORR activation. Furthermore, the data support a first‑order dependence on available Pt sites, arguing against simultaneous occupation of two Pt sites by a single O2 molecule during the rate‑determining step.
Background
Polymer electrolyte membrane fuel cells rely on Pt‑based catalysts to reduce oxygen efficiently. Pt alloys, such as PtCo, have shown higher ORR activity than monometallic Pt [1–16]. Yet, the catalytic surface is prone to anion adsorption and surface‑oxide formation, both of which are potential dependent [17–19]. Even trace concentrations of sulfate, bisulfate, and halide species are inevitable in fuel‑cell systems due to ionomer degradation and trace impurities. Previous work has characterized anion adsorption on flat Pt surfaces and bulk Pt alloys [20–34], but systematic studies on carbon‑supported PtCo nanoparticles remain scarce.
Given the high sulfonate content of perfluorosulfonic ionomers, bisulfate and sulfate ions are especially problematic. Recent observations in single‑cell operation [38] revealed a linear relationship between Pt/C activity and log(anion concentration). Building on this, we investigate how bisulfate and sulfate ions quantitatively impact ORR kinetics on PtCo nanoparticles across multiple potentials, providing the first detailed assessment for practical carbon‑supported catalysts.
Methods
Materials
The catalyst employed was 30 wt.% PtCo dispersed on high‑surface‑area carbon (Tanaka Kikinzoku, Japan). A 15 mL ink comprising ultrapure water (Milli‑Q), 2‑propanol (HPLC grade), and 5.37 wt% Nafion solution (ethanol + Milli‑Q) was mixed in a 200:50:1 volume ratio and sonicated for 5 min. Ten microliters of this ink was deposited on a glassy‑carbon disk (0.196 cm2) and dried for 1 h before testing.
Electrochemical Evaluation
RDE experiments were performed in a standard three‑electrode cell using a potentiostat and a rotation controller (Pine Instrument Co). The electrolyte was 0.1 M HClO4, with bisulfate or sulfate added via H2SO4 injections to achieve desired concentrations. Potentials were referenced to the reversible hydrogen electrode (RHE) via a Ag/AgCl reference electrode separated by a salt bridge. ORR curves were recorded at 5 mV s−1 from 0.05 V to 1.0 V under O2-saturated conditions at 1600 rpm. All measurements were conducted at ambient temperature.
Results and Discussion
Transmission electron microscopy confirmed a uniform particle size distribution of 3–7 nm on the carbon support (Fig. 1). Cyclic voltammetry exhibited symmetrical hydrogen adsorption/desorption peaks at ~0.15 V and Pt oxidation/reduction features at ~0.85 V/0.79 V vs. RHE (Fig. 2a), indicating reversible surface behavior. The measured H2 adsorption charge (210 µC cm−2) corresponds to a BET surface area of 62 m2 g−1 for PtCo.
Linear sweep voltammetry (LSV) revealed that increasing bisulfate or sulfate concentration shifts the ORR polarization curves toward more negative potentials, reflecting activity loss (Fig. 2b). The half‑wave potential and limiting current both decline with anion concentration.

a TEM image of the nanocatalyst, scale bar = 50 nm. b Zoomed‑up TEM image, scale bar = 20 nm
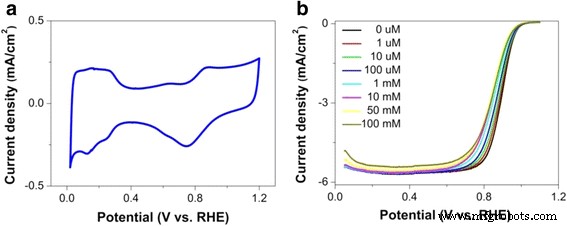
a CV curve at 20 mV s−1. b LSV curves at 0–100 mM bisulfate/sulfate
Figure 3a demonstrates a semilogarithmic linear dependence of ORR specific activity (I) on bisulfate concentration (CHSO4), described by:
Here, G represents the activity at zero anion concentration, and D quantifies the slope of the decline. Across potentials from 0.88 to 0.95 V, the relationship holds robustly. The measured ORR activity at 0.9 V and 0 mM bisulfate (521 µA cm−2 Pt) aligns with literature values [39].
Adopting a semilogarithmic adsorption isotherm for bisulfate on PtCo, the kinetic expression reduces to:
Thus, the exponential coverage term vanishes, indicating that bisulfate adsorption does not modify the activation free energy at a fixed potential. The rate law remains first order with respect to free Pt sites, supporting the conclusion that simultaneous occupation of two Pt sites by a single O2 molecule is unlikely.
Illustration of ORR mechanism
Analysis of the slopes (G and D) versus potential yields a consistent transfer coefficient of α ≈ 0.8 (Fig. 3b), indicating an asymmetric activation barrier. Tafel plots (Fig. 5) show slopes of 77–89 mV dec−1 that are invariant with bisulfate concentration, further confirming that the rate‑determining step remains unchanged by anion adsorption.
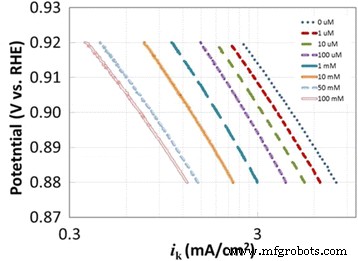
Tafel slope of 30 wt.% PtCo at various bisulfate concentrations
Conclusions
Bisulfate and sulfate ions linearly suppress ORR activity on PtCo nanoparticles, with a transfer coefficient of ~0.8. Importantly, anion adsorption does not alter the activation free energy at a given potential, and the ORR mechanism remains first order in free Pt sites. These findings suggest that surface blockage, rather than kinetic pathway alteration, underlies the observed poisoning.
Abbreviations
- CV:
Cyclic voltammetry
- LSV:
Linear sweep voltammetry
- ORR:
Oxygen reduction reaction
- PEMFC:
Polymer electrolyte membrane fuel cell
- RDE:
Rotating disk electrode
- RHE:
Reversible hydrogen electrode
- RRDE:
Rotating ring‑disk electrode
- TEM:
Transmission electron microscopy
- XAS:
X‑ray adsorption spectroscopy
Nanomaterials
- Oxygen: From Ancient Discovery to Modern Industrial Powerhouse
- Heavily Graphitic‑Nitrogen‑Doped High‑Porosity Carbon from Kidney Bean Biomass for Robust Oxygen Reduction Electrocatalysis
- Fe‑N‑C Nanofiber Electrocatalysts with Superior ORR Performance via Electrospinning and Sealed Pyrolysis
- First‑Principles Study of Small‑Molecule Adsorption on Penta‑Graphene for Gas‑Sensing Applications
- First‑Principles Insights into Transition‑Metal Adsorption on Black Phosphorene: Implications for Catalysis and Spintronics
- Surfactant‑Free Cu₂O@Au Hollow Cubes Deliver Enhanced CO₂ Electroreduction to CO
- Nitrogen‑Doped Porous Carbon Networks Derived from Graphitic Carbon Nitride for High‑Performance Oxygen Reduction
- Li₂RuO₃-Activated Lithia Nanocomposites for Advanced Oxygen Redox Cathodes
- Ultra‑High Nitrogen Doping through Nanochannel‑Mediated Synthesis of Mesoporous Fe/N/C Catalysts for Superior Oxygen Reduction
- Co/CoP Nanoparticles Encapsulated in N,P-Doped Carbon Nanotubes on Nanoporous MOF Nanosheets: High-Performance Electrocatalysts for Oxygen Reduction and Evolution Reactions



