Fe‑N‑C Nanofiber Electrocatalysts with Superior ORR Performance via Electrospinning and Sealed Pyrolysis
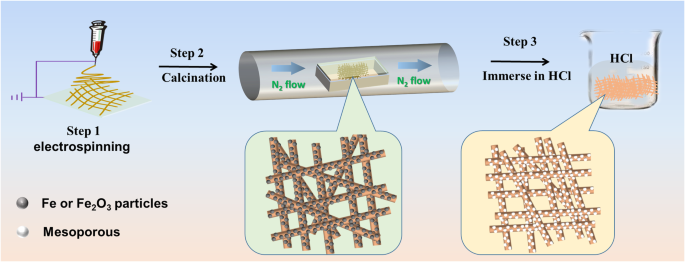
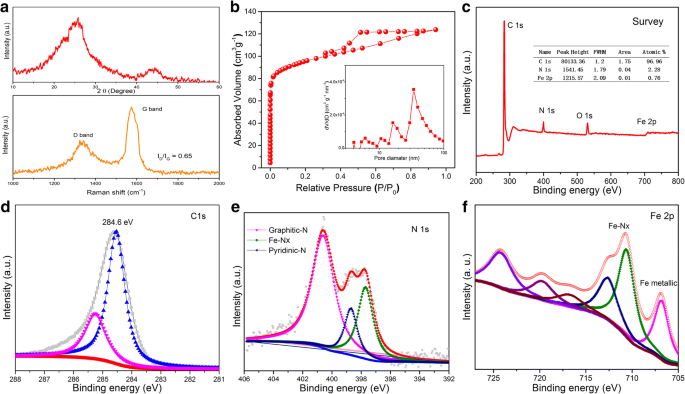
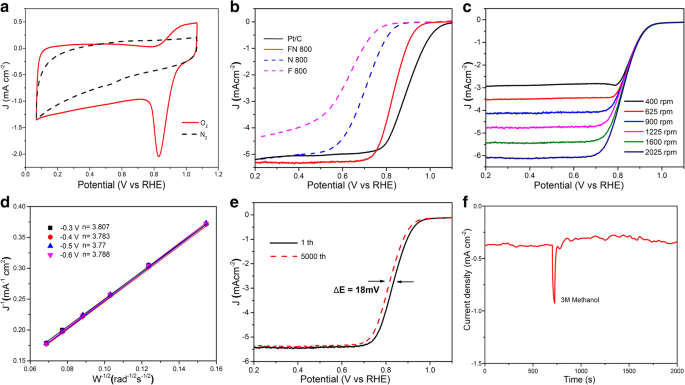
High‑performance electrocatalysts for the oxygen reduction reaction (ORR) are essential for electrochemical energy storage and conversion. Fe‑N‑C materials have emerged as the most promising non‑precious alternative to platinum, yet conventional synthesis often requires high‑temperature thermal treatment of nitrogen‑rich polymers or metal‑organic frameworks (MOFs). Here we report Fe‑N‑C mesoporous nanofibers synthesized from inexpensive urea and FeCl3 through electrospinning, in‑situ pyrolysis under a sealed environment, and acid leaching. The sealing step preserves nitrogen species during high‑temperature calcination, resulting in abundant Fe–Nx active sites and a large BET surface area of 354 m² g⁻¹. The catalyst delivers an ORR onset potential of 0.93 V and a half‑wave potential of 0.82 V in 0.1 M KOH, comparable to commercial Pt/C, while exhibiting excellent stability and methanol tolerance. This work offers a scalable route to high‑performance ORR electrocatalysts. Fuel cells are a cornerstone of clean energy conversion, but the oxygen reduction reaction (ORR) remains the primary bottleneck. Platinum‑based catalysts, although highly active, suffer from high cost, limited durability, CO poisoning, and scarce natural reserves. Consequently, non‑precious metal catalysts with competitive ORR activity are urgently needed. Among the various candidates, transition‑metal‑nitrogen‑co‑doped carbons (M–N/C, M = Fe, Co, Ni) have shown the most promise due to their excellent activity and stability. Density functional theory (DFT) studies indicate that the electronic structure of Fe–Nx sites, embedded in graphitic carbon, governs the binding energies of O₂, H₂O, and intermediate species (OOH*, OH*). Thus, a high density of Fe–Nx sites is essential for optimal ORR performance. Conventional approaches rely on pyrolysis of Fe–N₄ complexes or MOFs, which involve complex reaction pathways and limited control over active site exposure and conductivity. In this study, we introduce a streamlined synthesis of Fe‑N‑C mesoporous nanofibers that directly yields abundant Fe–Nx sites and a high surface area. All reagents were used as received. In a typical synthesis, 0.8 g polyacrylonitrile (PAN, Mw = 150,000), 0.1 g FeCl3, and 0.5 g urea were dissolved in 10 mL N‑N‑dimethylformamide (DMF) under vigorous stirring for 6 h to obtain a homogeneous solution. Electrospinning was performed using a 0.9 mm diameter spinneret, a 15 cm tip‑collector distance, and a 18 kV applied voltage. The collected fibers were carbonized in a tube furnace at 800 °C for 2 h. To suppress nitrogen loss at high temperature, a lid was placed on the porcelain boat during calcination. After pyrolysis, the product was immersed in HCl for 5 days to remove excess iron. The final Fe‑N‑C mesoporous nanofibers were denoted FN‑800. The material was characterized by X‑ray powder diffraction (XRD; Philips X’Pert Pro, λ = 1.54178 Å), transmission electron microscopy (TEM; Tecnai G20), field emission scanning electron microscopy (FE‑SEM; Hitachi SU 8010), energy‑dispersive X‑ray spectroscopy (EDS; JEOL JEM‑ARF200F), nitrogen adsorption–desorption (Micromeritics ASAP 2000), X‑ray photoelectron spectroscopy (XPS; ESCALAB MK II), and Raman spectroscopy (HR 800, Jobin Yvon, Horiba) with a 532‑nm laser. Electrochemical testing employed a three‑electrode setup on a CHI660B workstation. A catalyst ink was prepared by dispersing 5 mg of catalyst and 150 µL of 5 wt % Nafion in 350 µL of ethanol with 30 min sonication. Five microliters of the ink were deposited on a 3 mm glassy carbon electrode. Linear sweep voltammetry (LSV) was performed in 0.1 M KOH saturated with O₂ (scan rate 5 mV s⁻¹) using an Ag/AgCl (3 M KCl) reference and a platinum wire counter. Potentials were converted to the reversible hydrogen electrode (RHE) scale by E_RHE = E_Ag/AgCl + 0.059 pH + E°_Ag/AgCl (E° = 0.21 V at 20 °C). The electron transfer number was calculated from Koutecky–Levich plots using the standard equations (1)–(3) referenced in the original manuscript. The fabrication of Fe‑N‑C mesoporous nanofibers involved electrospinning of a PAN/FeCl3/urea solution, high‑temperature carbonization under a sealed atmosphere, and acid leaching to remove excess iron. Figure 1 illustrates the complete synthesis workflow. Illustration of the Fe‑N‑C mesoporous nanofiber preparation steps SEM images (Figure 2) reveal morphological evolution across the synthesis stages. The precursor nanofibers are several tens of micrometers long with a diameter of ~500 nm. Calcination reduces the diameter to ~200 nm and embeds iron particles, as confirmed by TEM. Acid treatment removes surface iron and yields a porous structure. High‑resolution TEM shows 5 nm particles with a lattice spacing of 0.197 nm, corresponding to tetragonal Fe (JCPDS 34‑0529). EDS mapping confirms uniform distribution of Fe, N, C, and O (atomic ratios: Fe 0.78 %, N 0.53 %, C 95.21 %, O 3.48 %). SEM and TEM images of FN‑800 before and after acid treatment, and elemental mapping. XRD patterns (Figure 3a) display peaks at 2θ = 26° and 44.5°, characteristic of graphitic (002) and (100) planes. No distinct Fe peaks appear, consistent with the low Fe content (0.78 %) and its uniform dispersion. Raman spectra show a G band stronger than the D band, with an I_D/I_G ratio of 0.65, indicating a highly graphitized carbon framework. The N‑800 sample (without FeCl3) shows a higher I_D/I_G of 1.06, confirming that FeCl3 promotes graphitic ordering during pyrolysis. XRD and Raman spectra of FN‑800 (a); N₂ sorption isotherms and pore size distribution (b); XPS survey and high‑resolution spectra (c‑f). BET analysis (Figure 3b) yields a surface area of 354 m² g⁻¹ and an average pore diameter of 35.9 nm, confirming a mesoporous architecture. Acid‑treated FN‑800 shows a surface area >2.5× higher than the non‑treated counterpart (140 m² g⁻¹), underscoring the role of acid leaching in creating porosity. XPS data (Figure 3c‑f) reveal a high‑carbon content (96.96 %) with significant N (2.28 %) and Fe (0.76 %). The C 1s spectrum shows peaks at 284.6 eV (graphitic C) and 285.4 eV (C–N/Fe–C). The N 1s spectrum deconvolutes into pyridinic N (398.7 eV), graphitic N (400.6 eV), and Fe–Nx (397.7 eV), confirming the presence of active sites. The Fe 2p spectrum displays a metallic Fe peak at 707.2 eV and oxidized Fe peaks (712.9, 717.4, 724.5 eV), with a Fe–Nx feature at 711.2 eV. To assess the influence of the sealed atmosphere during pyrolysis, a control sample without a lid was prepared. XPS shows a 62 % reduction in nitrogen content and loss of Fe–Nx peaks, highlighting the necessity of the sealed environment to retain nitrogen species and form Fe–Nx sites. Electrochemical performance (Figure 4) demonstrates that FN‑800 exhibits a prominent ORR peak in O₂‑saturated solution but not in N₂, confirming ORR activity. LSV curves (4b) reveal an onset potential of 0.93 V and a half‑wave potential of 0.82 V, rivaling Pt/C. Rotating disk electrode (RDE) analysis (4c–d) indicates a 4‑electron transfer number (3.77–3.81) across −0.30 to −0.60 V, confirming efficient ORR kinetics. Stability tests (4e) show only an 18 mV loss in E_1/2 after 5,000 cycles, and methanol tolerance (4f) demonstrates negligible current drop in the presence of 3 M methanol. a CV curves of FN‑800; b LSV curves of FN‑800, F‑800, N‑800, and Pt/C; c RDE curves; d Koutecky–Levich plots; e LSV after 5,000 cycles; f methanol tolerance. Fe‑N‑C mesoporous nanofibers with abundant Fe–Nx sites and a high BET surface area were fabricated via electrospinning, sealed in‑situ pyrolysis, and acid leaching. The sealed calcination preserves nitrogen species, enabling high‑density Fe–Nx active sites. The resulting catalyst delivers an ORR onset potential of 0.93 V and a half‑wave potential of 0.82 V in alkaline electrolyte, coupled with excellent durability and methanol tolerance. This approach offers a scalable route to high‑performance non‑precious ORR electrocatalysts. N‑N‑Dimethylformamide Energy‑dispersive X‑ray spectroscopy Metal‑organic frameworks Oxygen reduction reaction Polyacrylonitrile Scanning electron microscopy Transmission electron microscopy X‑ray photoelectron spectroscopy X‑ray diffractionAbstract
Background
Methods
Synthesis of the Fe‑N‑C Mesoporous Nanofibers
Instruments
Electrochemical Measurements
Result and Discussion

Conclusions
Abbreviations
Nanomaterials
- Heavily Graphitic‑Nitrogen‑Doped High‑Porosity Carbon from Kidney Bean Biomass for Robust Oxygen Reduction Electrocatalysis
- Mesoporous Silicon Microspheres via In Situ Magnesiothermic Reduction: A High‑Performance Anode for Sodium‑Ion Batteries
- Nanoporous N‑Doped Carbon Microfibers from Bamboo Waste: A High‑Performance, Platinum‑Free Electrocatalyst for Oxygen Reduction
- Co‑Sputtering and ALD Fabricated Ag Nanoparticle SERS Substrate: High Sensitivity and 30‑Day Stability for Glycerol Detection
- Nitrogen‑Doped Porous Carbon Networks Derived from Graphitic Carbon Nitride for High‑Performance Oxygen Reduction
- Hemp‑Stem‑Derived Nanoporous Carbon Anodes for High‑Capacity Lithium‑Ion Batteries
- Ultra‑High Nitrogen Doping through Nanochannel‑Mediated Synthesis of Mesoporous Fe/N/C Catalysts for Superior Oxygen Reduction
- Co/CoP Nanoparticles Encapsulated in N,P-Doped Carbon Nanotubes on Nanoporous MOF Nanosheets: High-Performance Electrocatalysts for Oxygen Reduction and Evolution Reactions
- Bright, Multicolor N‑Doped Carbon Dots from Ascorbic Acid and Phenylenediamine: Tunable Emission via Solvent Polarity and pH
- Stable, Reversible Photoluminescence of GaN Nanowires in Aqueous Solutions Tuned by Ionic Concentration



