Nanoporous N‑Doped Carbon Microfibers from Bamboo Waste: A High‑Performance, Platinum‑Free Electrocatalyst for Oxygen Reduction
Abstract
Metal‑free carbon electrocatalysts that rival platinum for the oxygen reduction reaction (ORR) are a research priority. Here we report a facile, activation‑assisted carbonization route that produces nitrogen‑doped nanoporous carbon microfibers (Me‑CFZ‑900) with an extraordinary BET surface area of ~929.4 m2 g−1. The synthesis uses melamine as a nitrogen source and bamboo‑derived biowaste as the carbon feedstock, with zinc chloride acting as an activator. Electrochemical evaluation in alkaline media shows that Me‑CFZ‑900 delivers a quasi‑four‑electron ORR pathway, an onset potential approaching 1.0 V vs. RHE, and superior long‑term stability compared with commercial Pt/C. Detailed XPS and Raman analyses reveal that graphitic‑N, pyridinic‑N, and pyrrolic‑N species coexist and contribute to the catalytic activity. This study demonstrates a scalable, low‑cost strategy to convert renewable biomass into high‑performance ORR catalysts.
Background
Fuel cells and metal‑air batteries rely on efficient ORR to achieve high energy conversion. Traditional Pt‑based catalysts, while effective, suffer from high cost and limited supply. Therefore, developing robust, inexpensive, Pt‑free catalysts is critical for commercial deployment. Heteroatom doping, especially with nitrogen, has emerged as a promising strategy to modify the electronic structure of carbon materials, creating active sites that enhance ORR kinetics. However, the precise nature of the active sites and the role of nitrogen configuration remain under debate, limiting rational catalyst design.
Biomass and animal waste have attracted attention as inexpensive nitrogen donors for carbon precursors. Prior work has employed soybean, silk, and hemin to fabricate nitrogen‑rich carbons with promising ORR activity. Yet, the conversion of bamboo biowaste—a widely available agricultural residue—into porous, nitrogen‑doped microfibers suitable for ORR has not been explored.
In this study, we develop a straightforward two‑step pyrolysis protocol that transforms bamboo carbon tissues into nitrogen‑doped, mesoporous microfibers. The resulting Me‑CFZ‑900 catalyst combines high surface area, rich nitrogen functionality, and a well‑defined pore network, achieving ORR performance comparable to commercial Pt/C while offering excellent durability.
Methods
Synthesis of Carbon‑Based ORR Catalysts
The process begins with shredded bamboo carbon tissues, carbonized at 350 °C for 1 h under N2 to remove volatiles, yielding CF350. This precursor is mixed with melamine (1 g) and ZnCl2 (1 g) in a 0.5 g CF350 batch, ground for 30 min, and then heated to 900 °C for 2 h (10 °C min−1) under N2, producing Me‑CFZ‑900. Control samples—CF‑900, CFZ‑900, and Me‑CFZ catalysts at 700, 800, and 1000 °C—were also prepared. All catalysts were acid‑washed (0.5 M HCl, 2 h) to remove residual salts before electrochemical testing. Each synthesis was replicated thrice with <5 % variation.

The schematic illustration for synthesis of mesoporous nitrogen‑doped carbon microfibers via the activation‑assisted carbonization of bamboo‑carbon biowastes
Physical Characterization
Morphology was examined by SEM (Hitachi UHR S4800) and TEM (FEI Tecnai‑G2 F30, 300 kV). XPS (Kratos XSAM800) quantified elemental composition. Nitrogen adsorption/desorption (Micromeritics ASAP 2010) provided BET surface area and pore size distribution. XRD (Shimadzu XRD‑6000, Cu Kα, λ = 1.5418 Å) confirmed amorphous structure. Raman spectra (Horiba HR800, 514.5 nm) assessed defect density. Electrochemical impedance spectroscopy (EIS) employed a 1 mM K3[Fe(CN)6]/K4[Fe(CN)6] redox couple in 0.1 M KCl.
Electrochemical Measurements
Electrocatalytic activity was evaluated on a CHI760E potentiostat using a GC‑RRDE (Φ = 5 mm). Working electrode preparation followed our established protocol, loading ~600 µg cm−2. All potentials were referenced to RHE. ORR was performed in 0.1 M KOH saturated with O2; the ring potential was fixed at 0.5 V vs. SCE to quantify H2O2. Electron transfer number (n) and peroxide yield (%) were calculated via standard equations (1) and (2). Koutecky‑Levich analysis (equations 3–4) provided kinetic parameters across rotation rates.
Results and Discussion
SEM and TEM images (Fig. 2) reveal that Me‑CFZ‑900 consists of irregular, nitrogen‑doped microfibers with abundant mesopores. ZnCl2 activation induces rapid dehydration and the evolution of H2O and O2 vapor, generating a porous network. High‑resolution imaging confirms the presence of edge defects that enhance ORR activity.

The SEM (a, b) and TEM (c, d) images of Me‑CFZ‑900
BET analysis (Fig. 3a) shows an exceptional surface area of 929.4 m2 g−1 and a total pore volume of 0.53 cm3 g−1, with a dominant pore size of 3.88 nm—ideal for mass transport during ORR. XRD patterns (Fig. 3b) confirm an amorphous carbon framework with slight shifts in the (002) and (101) peaks attributable to nitrogen incorporation. Raman spectra (Fig. 3c) display an ID/IG ratio of 0.92, indicating a high density of defects that favor catalytic sites. EIS data (Fig. 3d) reveal lower charge‑transfer resistance for Me‑CFZ‑900 compared to CF‑900 and CFZ‑900, reflecting enhanced electrical conductivity.
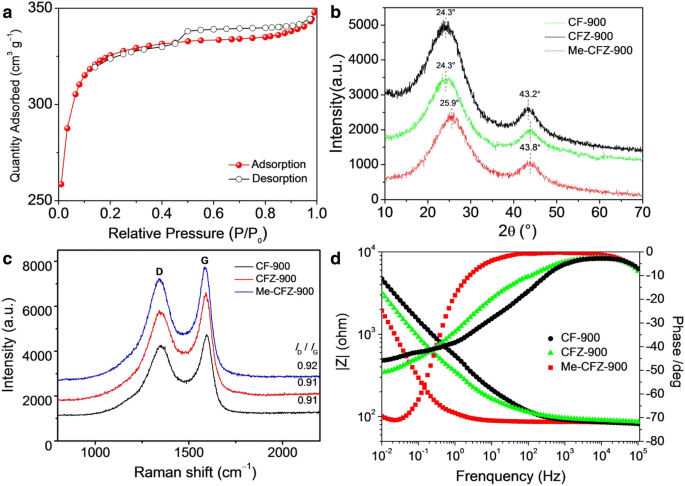
a N2-sorption isotherms of Me‑CFZ‑900. b XRD patterns of CF‑900, CFZ‑900, and Me‑CFZ‑900. c Raman spectra of CF‑900, CFZ‑900, and Me‑CFZ‑900. d Bode spectra of CF‑900, CFZ‑900, and Me‑CFZ‑900 under a sine wave of 5.0 mV amplitude in the frequency range of 100 kHz to 10 MHz
XPS survey spectra confirm the presence of C, N, and O in Me‑CFZ‑900, whereas CF‑900 and CFZ‑900 contain negligible nitrogen. High‑resolution N1s spectra (Fig. 4) show pyridinic‑N (398.3 eV), pyrrolic‑N (398.8 eV), graphitic‑N (401.2 eV), and oxidized‑N (403.4 eV) in Me‑CFZ‑900, whereas CF‑900/CFZ‑900 display only graphitic‑N (~401.5 eV). The total nitrogen content rises from 0.91 at.% in CF‑900 to 2.71 at.% in Me‑CFZ‑900, underscoring the effectiveness of melamine as a nitrogen source.
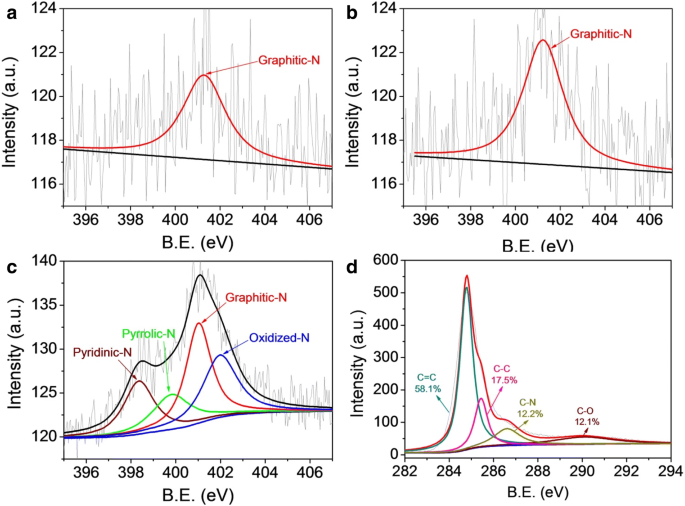
High‑resolution N1s XPS spectra of CF‑900 (a), CFZ‑900 (b), and Me‑CFZ‑900 (c). (d) High‑resolution C1s XPS spectra of Me‑CFZ‑900 (d)
Electrochemical tests (Fig. 5) demonstrate that Me‑CFZ‑900 outperforms CF‑900 and CFZ‑900, achieving a peak potential of 0.91 V vs. RHE and a half‑wave potential of 0.86 V. LSV curves at 1600 rpm confirm a quasi‑four‑electron pathway (n ≈ 3.9) and a peroxide yield below 14 %. Compared with commercial Pt/C (20 wt % Pt), Me‑CFZ‑900 delivers comparable activity and exhibits superior durability, showing only a 21 mV shift after 5000 CV cycles.
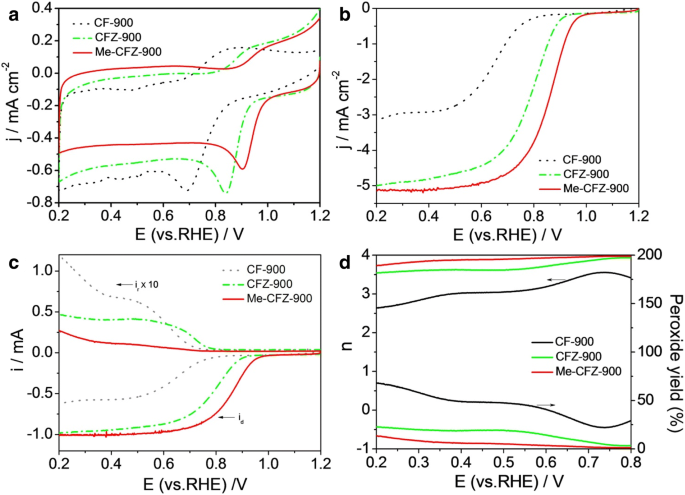
a CV curves of CF‑900, CFZ‑900, and Me‑CFZ‑900 in O2-saturated 0.1 M KOH. b LSV curves for ORR of CF‑900, CFZ‑900, Me‑CFZ‑900, and 20 wt % Pt/C at 1600 rpm. c Disk and ring currents from RRDE. d Electron transfer number and H2O2 yield.
Temperature dependence studies (Fig. 6) reveal that 900 °C is optimal; lower or higher pyrolysis temperatures reduce surface area, nitrogen content, and ORR performance. This underscores the importance of controlled activation to maximize active site density and pore accessibility.
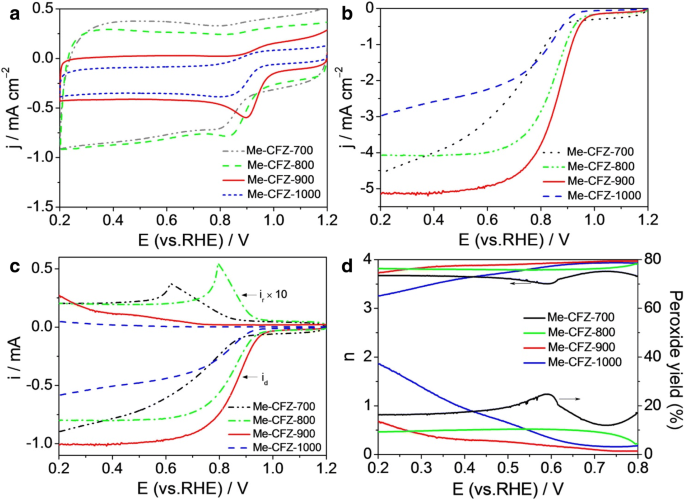
a CV and b LSV curves of Me‑CFZ‑700, 800, 900, 1000 in O2-saturated 0.1 M KOH. c Disk and ring currents from RRDE. d Corresponding electron transfer number and H2O2 yield.
Stability assessment via accelerated aging (AAT) confirms that Me‑CFZ‑900 retains > 98 % of its initial activity after 5000 CV cycles, outperforming Pt/C, which typically loses ~10 % under identical conditions.
Collectively, the synergy of high surface area, mesoporosity, and diverse nitrogen functionalities—particularly graphitic‑N and planar pyridinic/pyrrolic N—renders Me‑CFZ‑900 a compelling, cost‑effective alternative to platinum catalysts for alkaline fuel cell applications.
Conclusions
We have introduced a scalable, activation‑assisted synthesis that converts bamboo waste into nitrogen‑doped, nanoporous microfibers (Me‑CFZ‑900) with exceptional ORR performance in alkaline media. Key metrics include a half‑wave potential of 0.86 V vs. RHE, a peak potential of 0.91 V, a near‑four‑electron pathway (n ≈ 3.84), and a peroxide yield below 14 %. The catalyst also exhibits remarkable durability, shifting only 21 mV after 5000 CV cycles. The high BET surface area (929.4 m2 g−1) and mesoporous structure facilitate efficient oxygen transport, while the enriched nitrogen species provide abundant active sites. This work establishes a robust platform for converting renewable biowaste into high‑performance, platinum‑free ORR catalysts.
Abbreviations
- AAT:
Accelerated aging test
- AE:
Auxiliary electrode
- BET:
Brunauer–Emmett–Teller
- CF:
Carbon microfibers
- CV:
Cyclic voltammetry
- E1/2:
Half‑wave potential
- EORR:
Onset potential
- Ep:
Peak potential
- FE-SEM:
Field‑emission scanning electron microscopy
- GC:
Glassy carbon
- HR-TEM:
High‑resolution transmission electron microscopy
- LSV:
Linear sweep voltammetry
- Me-CFZ-900:
Nitrogen‑doped porous carbon microfibers
- ORR:
Oxygen reduction reaction
- Pt/C:
Platinum/carbon catalyst
- RDE:
Rotation disk electrode
- RE:
Reference electrode
- RHE:
Reversible hydrogen electrode
- RRDE:
Rotation ring‑disk electrode
- SCE:
Saturated calomel electrode
- WE:
Working electrode
- XPS:
X‑ray photoelectron spectroscopy
Nanomaterials
- Hierarchical Activated Carbon Fibers from Waste Cotton Gloves: High‑Performance Electrodes for Supercapacitors
- Fe‑N‑C Nanofiber Electrocatalysts with Superior ORR Performance via Electrospinning and Sealed Pyrolysis
- One‑Step Synthesis of Nitrogen‑Doped Hydrophilic Mesoporous Carbon Spheres from Chitosan for Enhanced Hydroxycamptothecin Delivery
- Nitrogen‑Doped Porous Carbon Networks Derived from Graphitic Carbon Nitride for High‑Performance Oxygen Reduction
- Hemp‑Stem‑Derived Nanoporous Carbon Anodes for High‑Capacity Lithium‑Ion Batteries
- First‑Principles Study of the Photogalvanic Response in Nitrogen‑Doped Monolayer MoS₂
- Carbon Dots Derived from Phellodendri Chinensis Cortex Carbonisata Safeguard Kidneys from Deinagkistrodon acutus Venom-Induced Acute Kidney Injury
- Co‑P‑C Co‑Doped MOF Electrocatalysts for Efficient ORR and OER
- Bright, Multicolor N‑Doped Carbon Dots from Ascorbic Acid and Phenylenediamine: Tunable Emission via Solvent Polarity and pH
- MAE Museum: A Carbon Fiber Landmark Celebrating Sustainability and Innovation



