APTES‑GLU Surface Functionalization Boosts ELISA Sensitivity for HIV‑p24 Detection to 1 nM
Abstract
Enzyme‑linked immunosorbent assay (ELISA) remains a gold standard for disease screening, yet its sensitivity hinges on how well antigens are immobilized on the polystyrene (PS) plate. We developed a simple, reproducible surface chemistry using 3‑(aminopropyl) triethoxysilane (APTES) followed by glutaraldehyde (GLU) cross‑linking, which provides a high‑density, oriented protein layer. After treating PS with 1% KOH, a 1:1 ratio of 1% APTES and 1% GLU gave the strongest signal, and overnight GLU incubation maximized binding. Using this platform, we detected HIV‑p24, an early infection marker, at 1 nM—30‑fold lower than conventional ELISA. The modified surface also maintained excellent specificity against human serum and HIV‑TAT. This chemistry is readily transferable to other biomarker assays requiring enhanced capture efficiency.
Background
Accurate detection of disease biomarkers, including viral antigens, is critical for early diagnosis and patient management. ELISA’s popularity stems from its high sensitivity and ease of use, yet its performance can be limited by suboptimal antigen attachment to the PS surface. Conventional adsorption relies on electrostatic interactions between surface carboxyls and protein amines, which can yield uneven coverage and reduced orientation. Numerous strategies—photochemistry, polymer coatings such as PVLA, and PEGylation—have been explored to improve immobilization, but many still struggle with small proteins or peptides.
Polystyrene’s hydrophobic backbone offers limited sites for covalent attachment. APTES introduces primary amines, enabling further functionalization with bifunctional cross‑linkers like GLU that possess two aldehyde groups. This approach covalently anchors proteins through Schiff base formation, promoting a denser, more uniform layer. Prior work has shown that APTES‑GLU chemistry can enhance antibody capture on gold or silica surfaces, but its application to ELISA plates for small antigen detection had not been fully demonstrated.
HIV‑p24 is a core capsid protein released early in infection, making it an ideal target for sensitive assays. Here, we employed APTES‑GLU surface chemistry to achieve ultra‑sensitive p24 detection, setting a new benchmark for ELISA‑based diagnostics.
Materials and Methods
Reagents and Biomolecules
Recombinant HIV‑p24 and Tat proteins, along with anti‑p24 antibody, were sourced from Abcam (Malaysia). APTES, GLU, and human serum were purchased from Sigma‑Aldrich (USA). Anti‑mouse‑IgG‑HRP was obtained from Thermo Scientific (USA). ELISA plates came from Becton Dickinson (France), and ELISA 5× coating buffer was from Biolegend (UK). BSA and TMB substrate were from Promega (USA). An analytical ELISA reader was supplied by Fisher Scientific (Malaysia).
Optimization of APTES and GLU Ratios
After activating PS with 1% KOH for 10 min, we tested APTES at 0.5%, 1%, and 2% (v/v) overnight at RT. Subsequent GLU treatments at 1% and 2% (v/v) were applied. A fixed 250 nM HIV‑p24 was then incubated on each surface, followed by blocking with 2% BSA. After adding a 1:1000 dilution of anti‑p24 antibody and anti‑mouse‑IgG‑HRP, the reaction was visualized with TMB and measured at 405 nm. Controls omitted the target antigen.
GLU Incubation Time
To assess the effect of GLU exposure, surfaces were incubated with GLU for either 3 h or overnight before adding 250 nM HIV‑p24. Remaining steps mirrored the previous protocol.
Comparative Detection Strategies
We compared two GLU‑mediated approaches: (1) “amine‑GLU‑p24” where GLU‑treated PS was directly exposed to 100 nM p24, and (2) “amine‑GLU premix” where GLU (2%) was mixed with 100 nM p24 for 30 min at RT before adsorption onto the APTES‑modified plate. Detection proceeded with the same antibody incubation sequence.
Limit of Detection Assessment
Serial dilutions of p24 (0.5–500 nM) were prepared. For each dilution, we performed the conventional ELISA and the two APTES‑GLU methods (1 and 2). Optical densities at 405 nm were recorded after standard washing steps.
Specificity Testing
To verify assay specificity, human serum and HIV‑TAT protein (mixed with 1% GLU) were tested under identical conditions, replacing p24. No significant signal was observed compared to the p24 positive control.
Results and Discussion
Surface modification via KOH activation, APTES amination, and GLU cross‑linking creates a chemically robust platform for protein capture. Figure 1a illustrates the sequence: KOH activation → APTES → GLU → antigen immobilization → antibody detection.
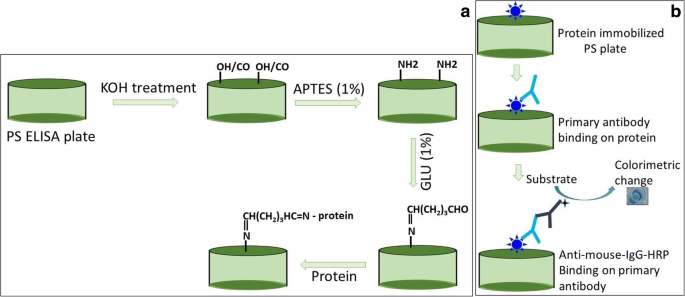
Schematic representation of chemically modified ELISA (a). The ELISA surface was modified chemically using APTES and GLU after potassium hydroxide treatment. The antigen was immobilized on the GLU-modified surface. b The immobilized antigen was detected by its partner antibody.
In optimizing APTES/GLU concentrations, 1% each yielded the highest specific signal (OD ≈ 0.22) with minimal background (OD ≈ 0.08). Higher concentrations (2%) increased non‑specific binding, likely due to excess aldehyde groups engaging other surface sites. Overnight GLU incubation produced roughly double the OD compared to 3‑h exposure, indicating more complete surface coverage (Figure 4a).
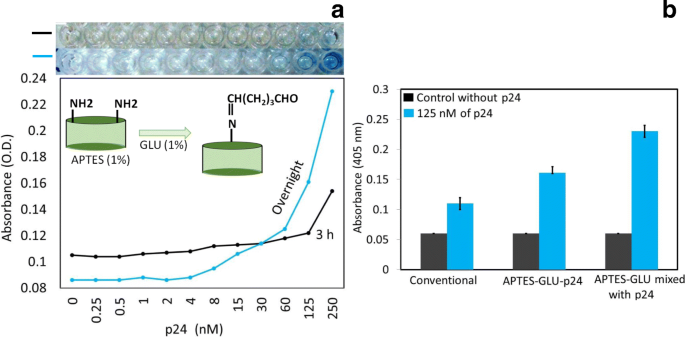
Optimization of the incubation period for GLU. a Optimized to have an incubation period of 3 h and an overnight incubation using 1% GLU on a 1% APTES‑modified surface. b Detection of 125 nM p24 by three different approaches, conventional, APTES‑GLU‑p24 (method 1), and APTES‑GLU‑premixed p24 (method 2).
When comparing the two GLU strategies, the premix approach (method 2) consistently produced higher OD values across all concentrations, underscoring the benefit of pre‑binding GLU to the antigen before plate adsorption.
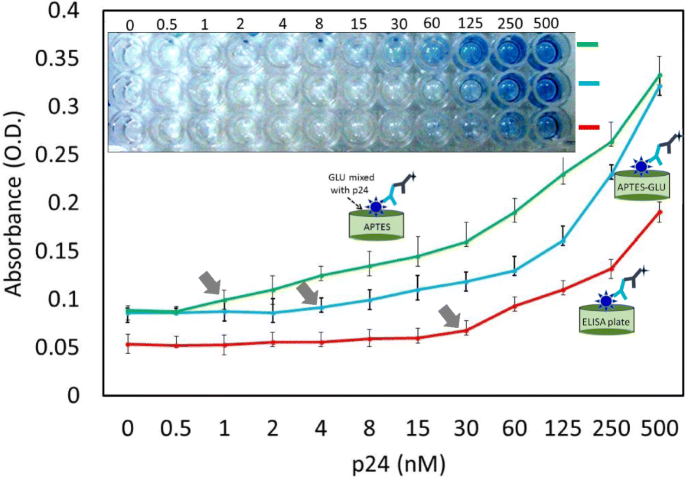
Limit of detection for the HIV‑p24 antigen. The p24 was titrated from 0.5 to 500 nM. Three different approaches were followed, conventional and methods 1 and 2.
The conventional ELISA reached a LOD of 30 nM. Method 1 reduced the LOD to 4 nM—a 7.5‑fold improvement—while method 2 achieved an impressive 1 nM LOD, a 30‑fold enhancement. This dramatic sensitivity gain is attributed to the higher protein density and optimal orientation afforded by the APTES‑GLU chemistry.
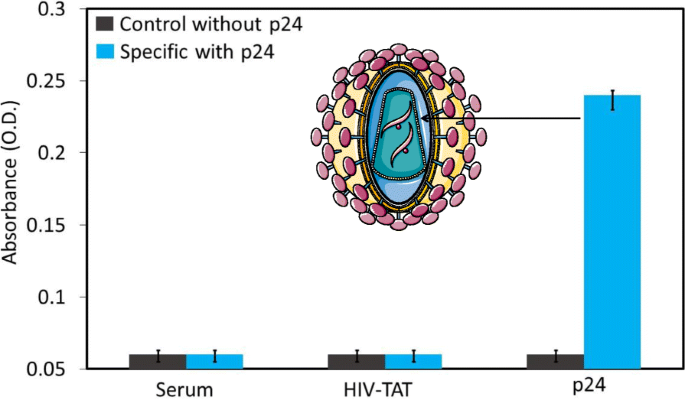
Specific detection of the HIV‑p24 antigen. Instead of HIV‑p24, the human serum and HIV‑TAT protein were used to check the specificity.
Specificity assays confirmed that neither human serum nor HIV‑TAT produced significant signals, reinforcing the assay’s selectivity for p24.
Conclusions
Optimized covalent immobilization of antigens on PS ELISA plates via APTES and GLU markedly improves detection limits. By enabling a dense, oriented protein layer, this surface chemistry achieves a 30‑fold lower LOD for HIV‑p24, illustrating its potential for other low‑abundance biomarkers. The method is simple, scalable, and compatible with existing ELISA workflows, paving the way for more sensitive, clinically relevant diagnostics.
Abbreviations
- APTES:
3-(Aminopropyl) triethoxysilane
- BSA:
Bovine serum albumin
- COOH:
Carboxyl
- ELISA:
Enzyme‑linked immunosorbent assay
- Fc:
Fragment crystallizable
- GLU:
Glutaraldehyde
- HIV:
Human immunodeficiency virus
- HRP:
Horseradish peroxidase
- IgG:
Immunoglobulin
- OD:
One optical density
- PEG:
Polyethylene glycol
- PS:
Polystyrene
- PVLA:
Polyvinyl benzyl lactonoylamide
- TMB:
3,3′,5,5′-Tetramethylbenzidine
Nanomaterials
- Harnessing Atomic Layer Deposition for Next‑Generation Micro‑LEDs and VCSELs
- Electrospun Polymer Nanofibers Enhanced with Noble Metal Nanoparticles for Advanced Chemical Sensing
- Ultra‑Sensitive Magnetoelastic Immunosensor for Carcinoembryonic Antigen Detection
- Ultra‑Smooth Copper Substrates Enhance Graphene Quality: Annealing + Electro‑Polishing
- Smartphone-Enabled Plasmonic ELISA for Ultra‑Sensitive Myoglobin Detection at Point of Care
- Boosting Planar Sb₂S₃ Solar Cell Efficiency with Cs₂CO₃‑Modified TiO₂ Electron Transport Layer
- Gold‑Nanoparticle‑Enhanced ELISA and Interdigitated Electrodes for Ultra‑Sensitive Detection of Human Factor IX Deficiency
- Improved Chemical‑Potential Model for Fast Deformation in Silicon Nanoparticle Electrodes of Li‑Ion Batteries
- Industrial Etching & Chemical Milling: Precision Surface Finishing for CNC Metal Parts
- From Factories to Labs: The Versatile Power of Industrial Robots



