Electrospun Polymer Nanofibers Enhanced with Noble Metal Nanoparticles for Advanced Chemical Sensing
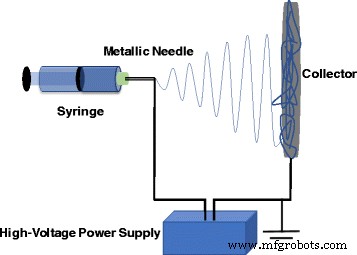
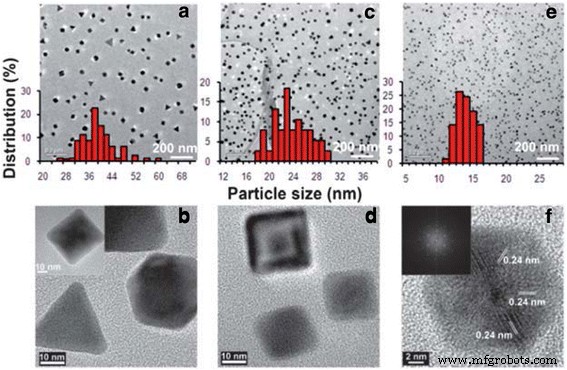
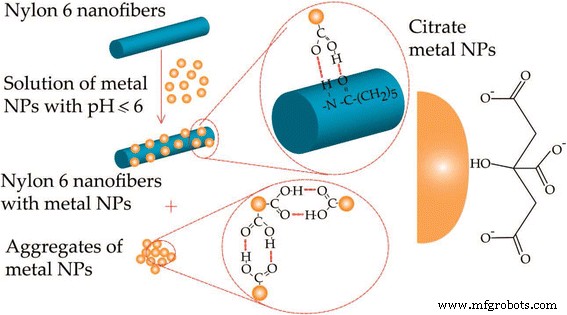
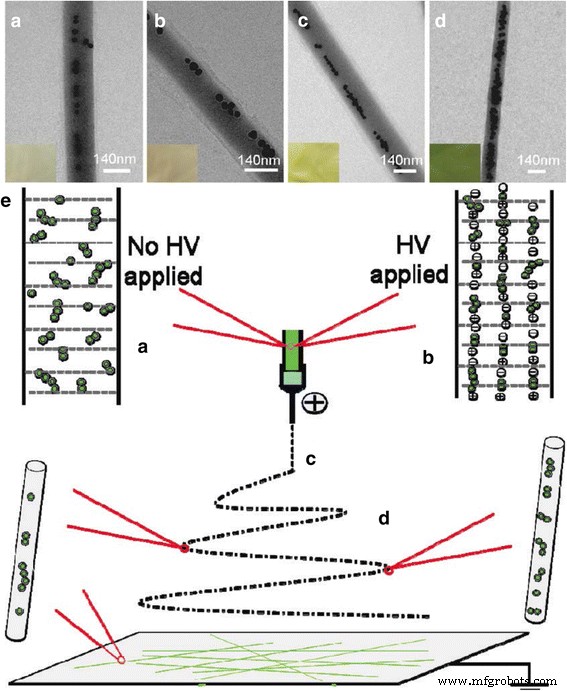
The synergy between noble metal nanoparticles—known for their plasmonic and electrocatalytic prowess—and electrospun polymer nanofibers—renowned for their exceptional mechanical and thermal attributes—has spawned hybrid systems with unprecedented functionality. This review surveys recent strategies for integrating noble metal NPs into electrospun fibers and demonstrates how such composites unlock superior sensitivity, durability, flexibility, and selectivity in chemical sensors. Continued progress in this area promises broad impact across multiple scientific domains. The rapid evolution of nanoscience has unleashed a plethora of practical applications, from air filtration and wound dressings to drug delivery, detection, energy production, and food packaging [1,2,3,4,5,6,7,8,9,10]. Because nanomaterials often exhibit physical and chemical properties that diverge dramatically from their bulk counterparts, a wide array of synthesis strategies have been devised, including template synthesis [16], self‑assembly [17], and electrospinning [18,19]. Among these, electrospinning stands out as the most versatile and straightforward method for producing one‑dimensional (1D) nanofibers such as PAN, PVdF, and PVA [20,21,18,22]. The simplicity and tunability of the technique have positioned electrospun polymer nanofibers at the forefront of chemical‑sensor research. Plasmonics, which explores light‑matter interactions at the nanoscale, has surged in popularity, driving advances in surface‑enhanced Raman scattering (SERS) [23,24,25], surface‑enhanced infrared absorption spectroscopy [26,27], surface‑enhanced fluorescence spectroscopy [28,29,30], surface plasmon resonance spectroscopy [31,32,33,34], and plasmonic colorimetry [35]. The optical behavior of plasmonic nanostructures is governed by collective oscillations of conduction electrons—surface plasmons—in noble metals such as Au, Ag, and Pt. However, assembling individual noble metal nanostructures into ordered architectures on a substrate remains challenging. Templated synthesis and assembly strategies have emerged as promising solutions, and electrospun polymer nanofibers provide an excellent scaffold for packing noble metal nanostructures with precision. By precisely controlling the integration of noble metal nanoparticles into electrospun fibers, researchers can create sensors that combine high sensitivity, stability, flexibility, compatibility, and selectivity. This review focuses on recent progress in integrating electrospun polymer nanofibers with noble metal nanoparticles and their application in chemical sensing. It covers (1) the fundamentals of electrospinning, (2) synthesis strategies for Au and Ag nanostructures, (3) fabrication of electrospun polymer nanofibers decorated with Au or Ag nanoparticles, and (4) illustrative sensing applications. The electrospinning apparatus typically consists of a high‑voltage power supply, a syringe filled with polymer solution, a metallic needle with a blunt tip, and a grounded conductive collector (Fig. 1). As the polymer solution is extruded, the high voltage generates surface charges that form a Taylor cone. When electrostatic repulsion overcomes surface tension, the jet elongates, solvent evaporates, and slender nanofibers are deposited on the collector. The basic laboratory setup for electrospinning Electrospinning offers remarkable control over fiber morphology by tuning parameters such as polymer concentration, solution viscosity, conductivity, flow rate, applied voltage, tip‑collector distance, and humidity [12]. For instance, lower polymer concentrations can lead to bead formation due to dominant surface tension, whereas higher molecular weight polymers yield more entangled chains that support uniform jet formation even at reduced concentrations [40]. Applied voltage and flow rate also influence fiber diameter, with higher voltages generally producing thinner fibers, though the effect can be complex and material‑dependent. In the past two decades, researchers have developed diverse methods to synthesize Ag and Au nanostructures with controlled sizes and shapes. These morphologies directly influence optical, electronic, magnetic, and chemical properties, making them suitable for a range of applications. Chemical reduction remains the most common approach, involving mixing a metal precursor with a reducing agent and/or stabilizer. AgNO3 and HAuCl4 are the standard precursors for silver and gold, respectively, and reducing agents such as sodium borohydride, alcohols, sodium citrate, and PVP enable precise size control. For example, Xia and colleagues synthesized Ag nanocubes (60–100 nm) and demonstrated that larger, sharper‑cornered particles exhibited superior SERS performance due to better overlap between the laser source and the plasmon resonance band [39]. Turkevich’s classic citrate reduction method (1951) remains a cornerstone for producing gold nanoparticles. By adjusting citrate concentration, researchers can tune particle size, as citrate acts as both a nucleation and growth agent [41]. Subsequent studies revealed that pH also plays a critical role, influencing nucleation kinetics and final morphology [42]. Brust and Schiffrin’s two‑phase synthesis (1994) transferred AuCl4− from aqueous to organic phase via tetraoctylammonium bromide, enabling the growth of 1–3 nm gold clusters with thiol ligands through sodium borohydride reduction [41]. Parameters such as gold/thiol ratio, temperature, and reduction rate allow fine‑tuning of particle size. Seed‑mediated growth, pioneered by Jana et al., further enhances size control by adding high‑quality seeds to a growth solution, though the process demands meticulous seed preparation [44,45,46,47,48]. TEM pictures of the thiol‑derivatized gold nanoparticles at a low and b high magnification [147]. Reprinted with permission from [147]. Copyright {2010} Royal Society of Chemistry Polymers such as PVP, PEG, PVA, PVME, chitosan, and PEI have been employed as stabilizers for gold nanoparticles [49,50,51,52,53,54,55,56,57]. For instance, PVP can act simultaneously as a reducing agent and steric stabilizer; varying the PVP/metal ion ratio yields nanoparticles of distinct shapes and sizes (Fig. 3) [56,58]. TEM images and histograms of AuNPs from AuNP–PVP nanocomposite films with weight ratios of HAuCl4 to PVP, [HAuCl4/PVP] = 1:1.5 (a, b), 1:2 (c, d), and 1:4 (e, f) [88]. Reprinted with permission from ref. [67]. Copyright {2010} Royal Society of Chemistry Physical methods—including photochemistry, sonochemistry, radiolysis, thermolysis, and microwave irradiation—have also been harnessed to refine gold nanoparticle quality [58,59,60,61,62,63,64,65]. Microwave synthesis, for instance, can modulate growth rate and morphology through the addition of oleic acid, yielding a spectrum of shapes (Fig. 4) [65]. TEM images of the gold nanoparticles prepared in i 60, ii 70, iii 80, and iv 90% oleic acid [65]. Reprinted with permission from ref. [65]. Copyright {2010} American Chemical Society Citrate‑mediated synthesis of silver colloids in aqueous solutions has long been a staple, where sodium citrate serves as both reducing agent and stabilizer. Adjusting citrate concentration or solution pH tailors particle size and morphology [66]. The polyol process, employing ethylene glycol, propylene glycol, or pentanediol as both solvent and reducing agent, yields silver nanostructures across a broad size and shape spectrum [67,68,69,70,71,72]. While the exact mechanism remains complex, glycolaldehyde is a key intermediate that strongly reduces Ag+ to Ag, explaining the temperature dependence of the reaction [72]. Seed‑mediated growth offers precise morphological control, with the final shape dictated by initial seed characteristics and facet‑specific growth rates. Capping agents such as citrate preferentially bind to {111} facets, promoting platelet formation, whereas PVP binds more strongly to {100} facets, steering growth along other crystallographic directions [13,67]. Light‑mediated synthesis, wherein laser irradiation induces silver deposition on existing seeds, enables tunable shape and size by adjusting wavelength and power. The process hinges on Ag seeds absorbing light, reducing oxygen, and releasing Ag+ that, together with citrate, generate electrons that drive further silver deposition [13,78,80]. Au and Ag nanostructures are versatile platforms for plasmonics, biomedical research, sensing, and catalysis [81,82,83,84,85,86]. By embedding these nanoparticles into electrospun polymer nanofibers, researchers can enhance reusability, broaden applicability, and stabilize the metal surface against sulfuration or oxidation. For example, incorporating Ag nanoparticles into a polymer matrix can effectively prevent surface sulfuration, while adding Au nanoparticles to PEO fibers increased fiber diameter by ~50 nm [87]. Two main strategies exist for fabricating Ag‑decorated nanofibers. The first involves pre‑synthesizing Ag nanostructures (or reducing Ag+ to Ag inside a separate solution) before mixing them with the polymer precursor. This approach eliminates the need for the polymer to act as a reducing agent and permits the use of a broad range of polymers. The second strategy dissolves the Ag precursor directly into the polymer solution or onto the fiber surface, followed by an in‑situ reduction step. Reducing polymers such as chitosan or PVP, UV irradiation, microwave heating, or hydrogen reduction can serve as the reducing agent in this approach [88,89,90,91,92,93,94,95,96,97]. For instance, Leonard et al. decorated polyurethane nanofibers with silver nitrite, and after 4 h of irradiation, silver nanoparticles formed a wire‑like structure on the composite surface [98]. Au‑decorated fibers can be produced by first synthesizing Au nanostructures (e.g., via citrate reduction or seed‑mediated growth) and then dispersing them into the polymer solution. For applications requiring surface attachment, functional groups on the polymer can attract Au nanoparticles; for example, adjusting the pH converts surface‑bound citrate to COOH, enabling hydrogen bonding with amide groups on nylon 6 fibers, thereby anchoring Au or Ag NPs to the fiber surface [103]. Postulated mechanism of pH‑induced assembly of metal nanoparticles on the surface of nylon 6 nanofibers [103]. Reprinted with permission from ref. [103]. Copyright {2008} American Chemical Society Polymers containing thiol or amine functionalities (e.g., via MPTES modification) can form strong bonds with Au nanoparticles, enhancing loading density. In addition, certain polymers act as electron donors during photo‑reduction; for example, PVA under UV irradiation can generate aldehyde groups that reduce Au3+ to Au0 [52,104]. Table 1 summarizes recent sensing studies that harness electrospun polymer nanofibers loaded with Au or Ag nanoparticles. These composites consistently demonstrate high sensitivity, selectivity, and low detection limits, owing to the high surface area and abundant hot‑spot formation between closely spaced metal particles. Surface‑enhanced Raman scattering (SERS) has become a powerful analytical tool for probing single molecules, biomolecules, and cells [105,106,107,108,109,110,111]. Au and Ag nanostructures amplify Raman signals through localized electromagnetic field enhancement, with the enhancement factor strongly dependent on nanoparticle size and shape [113]. Traditional SERS substrates require rigid supports and complex fabrication, whereas electrospun nanofibers provide flexible, high‑density, three‑dimensional platforms that can wrap around irregular surfaces and protect the metal cores from environmental degradation, especially for Ag [116]. Polymers such as PVA, cellulose, PMMA, chitosan, PAA/PVA blends, and silica have been blended with Au or Ag nanostructures to produce flexible SERS substrates. For example, nearly monodispersed Ag nanoparticles were first synthesized via a microwave‑assisted method and then incorporated into a 7 % aqueous PVA solution for electrospinning [116]. Within the fibers, Ag nanoparticles self‑assemble into linear chains along the fiber axis, driven by electrostatic attraction under the high‑voltage field, yielding exceptionally reproducible Raman signals with relative standard deviations below 0.07 [116]. a–d Typical TEM image of Ag/PVA nanofibers with the molar ratio of PVA/Ag 530:1 (a), 530:2 (b), 530:3 (c), and 530:4 (d). The increase of the molar ratio of Ag/PVA in the Ag/PVA solution led to stronger aggregation state and a larger distribution in the sizes of the aggregated Ag NPs. e Schematic representation of the formation of chain‑like arrays of Ag NP aggregates within PVA nanofibers [116]. Reprinted with permission from ref. [116]. Copyright {2009} American Chemical Society Ag and Au nanostructures can also be co‑assembled within PVA fibers, enabling tunable SERS responses by adjusting the Au/Ag ratio and excitation wavelength, as different metals resonate at distinct wavelengths [124]. Overall, both Au/PVA and Ag/PVA composites exhibit outstanding SERS performance. Metal nanoparticles—especially Au, Ag, Cu, and Ni—are widely employed in electrochemical sensing due to their high surface area, electronic conductivity, and chemical stability [125,126]. Au nanoparticles lower overpotentials and preserve redox reversibility, making them suitable for detecting analytes such as glucose [128,129], dopamine [130,131,132,133], bisphenol A [134], mercury [135,136,137,138], antimony [139], and hydrogen peroxide [140]. Their biocompatibility and ease of functionalization further enable biosensor development [141,142,143,144]. Coupling these nanoparticles with high‑surface‑area polymer nanofibers amplifies sensitivity and loading capacity [145]. For instance, Sapountzi et al. fabricated PVA/PEI/glucose oxidase nanofibers decorated with Au nanoparticles to enhance conductivity and used them as electrochemical biosensors [146]. To mitigate water solubility, the fibers were cross‑linked with glutaraldehyde vapors, preserving morphology after 15 days in water [99]. Subsequent surface modification with MPTES enabled robust attachment of Au nanoparticles via thiol–gold chemistry, yielding highly sensitive detection of H2O2 with fast response, wide linear range, and low detection limit [99]. Extensive research has underscored the unique advantages of combining Au or Ag nanostructures with electrospun polymer nanofibers. The resulting composites inherit the mechanical robustness, large surface area, and facile functionalization of polymer fibers, while leveraging the plasmonic and catalytic properties of noble metals. Such hybrids hold promise for optical sensors, antibacterial coatings, photovoltaics, and chemical/biological detection. Although the field remains in its early stages, rational integration of noble metal nanoparticles into nanofiber matrices is poised to unlock unprecedented sensing strategies. Future work should focus on precise morphology control, mechanistic understanding of nanoparticle assembly, and tailoring composites for specific analytical challenges. Glutaraldehyde 3-Mercaptopropyltrimethoxysilane Nanowires Poly (acrylic acid) Polyacrylonitrile Poly(ethylene glycol) Polyethyleneimine Poly (ethylene oxide) Polyvinyl alcohol Polyvinylidene fluoride Poly(vinyl methyl ether) Polyvinylpyrrolidone Surface-enhanced Raman scattering Transmission electron microscopy X-ray photoelectron spectroscopyAbstract
Review
Background
Electrospinning: Basic Set-up and Process Parameters
Synthesis and Assembly of Au Or Ag Nanostructures
Synthesis of Au Nanostructures


Synthesis of Ag Nanostructures
Electrospun Polymer Nanofibers Decorated with Noble Metal Nanoparticles
Preparation of Electrospun Polymer Nanofibers Decorated with Ag Nanoparticles
Preparation of Electrospun Polymer Nanofibers Decorated with Au Nanoparticles
Sensing Applications of Electrospun Polymer Nanofibers Decorated with Noble Metal Nanoparticles
Electrospun Polymer Nanofibers Decorated with Noble Metal Nanoparticles for Chemical Sensing Based on SERS
Electrospun Polymer Nanofibers Decorated with Noble Metal Nanoparticles for Chemical Sensing Based on Electrochemical Techniques
Conclusions
Abbreviations
Nanomaterials
- Graphene Oxide–Silver Nanoparticle Nanocomposites: A Potent Antibacterial and Antifungal Agent
- Magnetic MnFe₂O₄/Reduced Graphene Oxide Nanocomposite: Efficient and Recyclable Adsorption of Tetracycline from Water
- Magnetic Core‑Shell Nanoparticles: Advancing Targeted Drug Delivery, Imaging, and Antimicrobial Therapies
- Polydopamine Core–Shell Nanoparticles with Redox‑Responsive Polymer Shells for Targeted Drug Delivery and Synergistic Chemo‑Photothermal Therapy
- Carbon Dots: Advanced Fluorescent Platforms for Sensitive Metal‑Ion Detection
- Silver‑Embedded Polymer Microfibers for Enhanced Up‑Conversion Optical Sensing
- General Hydrothermal Fabrication of Carbon‑Coated Noble Metal Nanocomposites with Photothermal Function
- Advanced Approaches to Noble Metal Nanoparticle Synthesis
- Chemical Film Conversion Coating for Sheet Metal: Enhancing Durability & Performance
- Essential Safety Guidelines for Metal Fabrication Work



