Ni(OH)₂ Hollow‑Porous Architecture Enables Ultra‑Sensitive, Enzyme‑Free Glucose Detection
Abstract
Nickel hydroxide (Ni(OH)₂) has emerged as a promising non‑noble metal catalyst for glucose sensing, yet its practical utility is limited by sluggish electron transport and restricted mass diffusion. Here, we introduce a rationally designed hollow‑porous Ni(OH)₂ architecture (Ni(OH)₂‑HPA) prepared via a coordinating etching and precipitating (CEP) route that delivers a uniform cubic morphology with an internal void and a porous shell composed of ultrafine nanoparticles. The resulting material exhibits an exceptionally high specific surface area (54.7 m² g⁻¹), well‑ordered diffusion channels, and robust structural stability. When employed as a glucose sensor in alkaline medium, Ni(OH)₂‑HPA demonstrates a remarkable sensitivity of 1843 µA mM⁻¹ cm⁻², a detection limit of 0.23 µM, and a rapid response time of 1.4 s. These findings highlight the hollow‑porous design as an effective strategy for fabricating high‑performance, enzyme‑free glucose sensors.
Background
Accurate glucose monitoring is essential for clinical diagnostics, food safety, and environmental assessment. Conventional methods—including surface plasmon resonance, Fehling’s test, optical rotation, fluorescence, and electrochemical detection—offer varying degrees of sensitivity and practicality, but electrochemical sensing remains the most attractive due to its simplicity, low cost, and exceptional detection limits.
The performance of an electrochemical glucose sensor hinges on the electrocatalytic activity of its working electrode. Transition‑metal hydroxides, particularly Ni(OH)₂, have attracted attention owing to their abundant availability, cost‑effectiveness, and intrinsic redox activity (Ni²⁺/Ni³⁺) in alkaline solutions. Nonetheless, their catalytic performance is often hampered by limited electron conductivity and poor mass transport, preventing them from meeting industrial‑scale demands.
Nanostructuring has proven effective in enhancing electrocatalytic activity by modulating shape, size, and composition. Hollow‑porous nanostructures, in particular, offer high surface area, low density, and robust structural integrity, enabling efficient electron and mass transport while preventing particle aggregation. These attributes make them ideal candidates for constructing advanced glucose sensors.
In this study, we fabricate cubic Ni(OH)₂‑HPA using a Cu₂O template and the CEP methodology. By comparing the performance of intact Ni(OH)₂‑HPA with a mechanically broken counterpart (Ni(OH)₂‑BHPA), we demonstrate that the hollow‑porous design confers superior sensitivity, lower detection limit, and faster response, establishing a scalable route for high‑performance, enzyme‑free glucose sensing.
Methods/Experimental
Chemicals and Reagents
All reagents were analytical grade and used without further purification. Copper chloride dihydrate (CuCl₂·2H₂O, ≥ 99.0 %), nickel chloride hexahydrate (NiCl₂·6H₂O, ≥ 98.0 %), sodium thiosulfate pentahydrate (Na₂S₂O₃·5H₂O, ≥ 99.0 %), polyvinylpyrrolidone (PVP, Mw = 40 000), and sodium hydroxide (NaOH, ≥ 98.0 %) were sourced from Chengdu Kelong. Glucose (≥ 99.5 %), lactose (≥ 98.0 %), sucrose (≥ 99.5 %), fructose (≥ 99.0 %), l‑ascorbic acid (≥ 99.7 %), uric acid (≥ 99.0 %), and 5 wt % Nafion solution were purchased from Sigma‑Aldrich.
Synthesis of Ni(OH)₂ HPA
Cu₂O cubic templates were prepared following a previously reported procedure (see Additional file 1: Figure S1). In a typical synthesis, 10 mg of Cu₂O cubes and 4 mg of NiCl₂·6H₂O were dispersed in a 10 mL ethanol–water (1:1) mixture by sonication. After adding 0.33 g of PVP and stirring for 30 min, 4 mL of 1 M Na₂S₂O₃ was introduced dropwise. The mixture was maintained at 25 °C for 3 h, after which the products were collected by centrifugation, washed repeatedly, and dried at room temperature. For comparison, Ni(OH)₂‑BHPA was generated by ultrasonically disrupting Ni(OH)₂‑HPA for 2 h (see Additional file 1: Figure S2).
Materials Characterization
Phase identification was performed by X‑ray diffraction (XRD; Rigaku D/Max‑2400). Surface composition was examined via X‑ray photoelectron spectroscopy (XPS; ESCALAB250Xi). Morphology and microstructure were characterized using field‑emission scanning electron microscopy (FESEM; FEI Quanta 250, Zeiss Gemini 500) and high‑resolution transmission electron microscopy (HRTEM; FEI F20). Brunauer–Emmett–Teller (BET) analysis provided specific surface area (SSA) and pore size distribution.
Electrochemical Measurements
All measurements were conducted on a μIII Autolab workstation. The working electrode comprised a glassy carbon electrode (GCE, 3 mm diameter) coated with a Nafion‑impregnated slurry of Ni(OH)₂‑HPA or Ni(OH)₂‑BHPA (5 µL of 1 mg mL⁻¹ suspension in 0.05 % Nafion). The electrode was dried under nitrogen flow. A platinum foil and Ag/AgCl electrode served as counter and reference electrodes, respectively. Electrochemical performance was evaluated by cyclic voltammetry (CV), chronoamperometry (CA), and electrochemical impedance spectroscopy (EIS). EIS data were recorded from 0.01 to 100 kHz with a 5 mV perturbation amplitude.
Results and Discussions
Characterizations
Figure 1a displays the XRD pattern of Ni(OH)₂ products, confirming the hexagonal β‑Ni(OH)₂ phase (JCPDS 14‑0117). The weak diffraction peaks indicate a low crystallinity, consistent with the nanostructured nature of the material. XPS analysis (Figure 1b–d) reveals the presence of Ni 2p and O 1s signals characteristic of Ni²⁺ in Ni(OH)₂. The Ni 2p₃/₂ and Ni 2p₁/₂ peaks at 856.1 eV and 873.7 eV, respectively, exhibit a 17.6 eV separation typical of β‑Ni(OH)₂. The O 1s peak at 531.2 eV corresponds to Ni–O–Ni bonds.
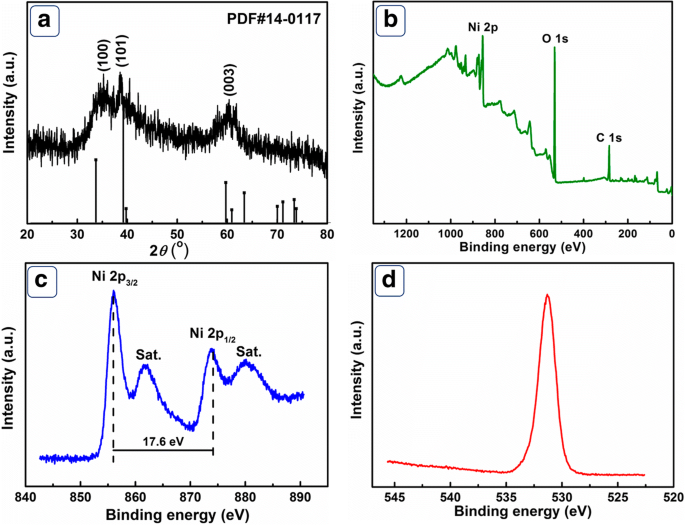
a XRD pattern of Ni(OH)₂. b XPS survey. c Ni 2p spectrum. d O 1s spectrum.
SEM images (Figure 2a) show uniform cubic particles, while Figure 2b confirms the presence of hollow cores in Ni(OH)₂‑HPA. The shell, assembled from densely packed nanoparticles, appears rough and porous. TEM (Figure 2c) corroborates the hollow morphology, and SAED patterns reveal the low crystallinity observed in XRD. The cubes measure ≈ 600 nm on each edge with a shell thickness of ≈ 50 nm, providing ample internal volume for ion transport.

a, b SEM; c, d TEM of Ni(OH)₂‑HPA. e Optical photos of the reaction solution over time. f TEM snapshots at different reaction stages. g Schematic of the growth mechanism.
Time‑resolved TEM and optical imaging (Figure 2e–f) reveal that S₂O₃²⁻ ions not only etch the Cu₂O core but also precipitate Ni(OH)₂ on the emerging shell. The concurrent release of OH⁻ ions facilitates the transformation of Ni²⁺ to Ni(OH)₂, yielding a well‑defined hollow‑porous structure. Controlled diffusion of S₂O₃²⁻ and OH⁻ from the interior ensures uniform shell growth.
The BET analysis (Figure 3) shows that Ni(OH)₂‑HPA possesses an SSA of 54.7 m² g⁻¹, far exceeding the 10.3 m² g⁻¹ of Ni(OH)₂‑BHPA. Pore size distributions indicate micropores (20–40 nm) and mesopores (60–85 nm) in the hollow sample, while the broken counterpart displays a less ordered distribution. The higher SSA and ordered porosity in Ni(OH)₂‑HPA facilitate efficient mass transport and provide abundant active sites.
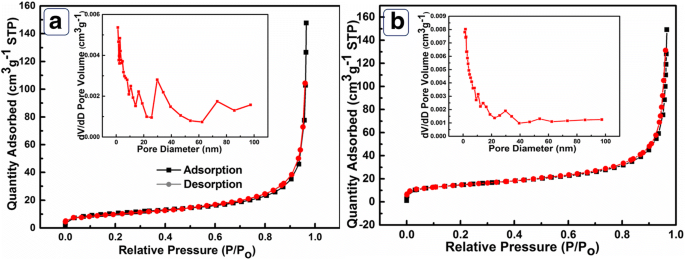
a N₂ adsorption–desorption isotherms for Ni(OH)₂‑HPA and Ni(OH)₂‑BHPA. b Corresponding pore size distributions.
Electrochemical Measurements
In 0.1 M NaOH, cyclic voltammetry (Figure 4a) demonstrates that Ni(OH)₂‑HPA delivers a higher redox peak current and a lower onset potential (0.41 V) than Ni(OH)₂‑BHPA. The enhanced current is attributed to the larger SSA and streamlined diffusion pathways of the hollow architecture. The electrochemical oxidation of glucose proceeds via the Ni(OH)₂/NiOOH redox couple (Scheme 1).
$$\text{Ni(OH)}_2+\text{OH}^-\rightarrow\text{NiOOH}+\text{H}_2\text{O}+e^-$$ $$\text{NiOOH}+\text{glucose}\rightarrow\text{Ni(OH)}_2+\text{gluconic acid}$$Electrochemical impedance spectroscopy (Figure 4b) confirms that Ni(OH)₂‑HPA exhibits lower charge‑transfer resistance (R_ct) and internal resistance (R_s) compared with Ni(OH)₂‑BHPA, while a higher Warburg impedance (Z_w) indicates superior mass‑transport capability.
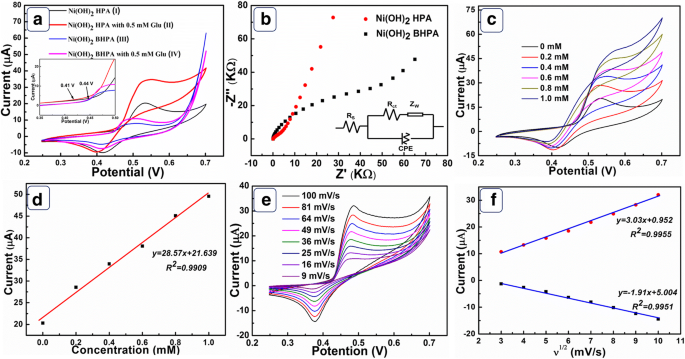
a CVs of Ni(OH)₂‑HPA and Ni(OH)₂‑BHPA with and without 0.5 mM glucose. b Nyquist plots and equivalent circuit. c CVs at varying glucose concentrations. d Linear relationship between peak current and glucose concentration. e CVs at different scan rates. f Peak current vs. √scan rate.
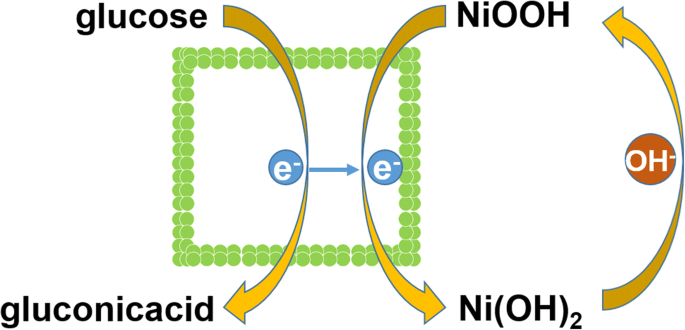
Scheme 1. Electrocatalytic mechanism for glucose oxidation on Ni(OH)₂‑HPA.
Chronoamperometry at 0.6 V (Figure 5c) shows that Ni(OH)₂‑HPA achieves a sensitivity of 1843 µA mM⁻¹ cm⁻² and a detection limit of 0.23 µM, outperforming Ni(OH)₂‑BHPA (632 µA mM⁻¹ cm⁻², 0.67 µM). The response time is 1.4 s, shorter than 1.8 s for the broken sample. Table 1 compares these metrics with other Ni(OH)₂‑based sensors, underscoring the superior performance of the hollow‑porous design.
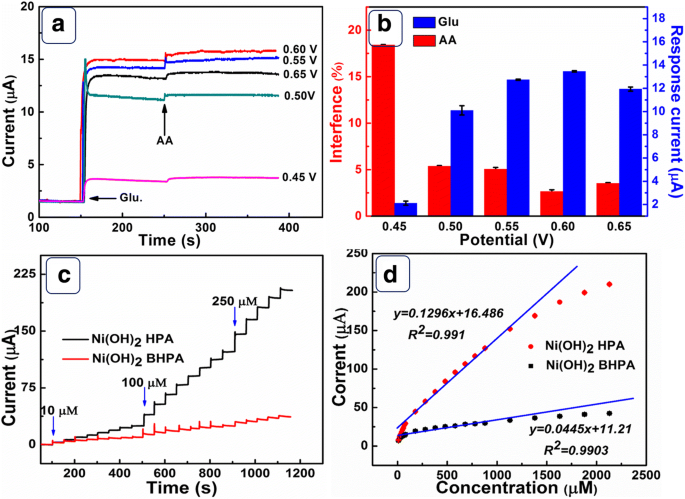
a Amperometric response at different potentials with 0.1 mM glucose and 0.01 mM AA. b Current response vs. potential. c CA curves at 0.6 V for Ni(OH)₂‑HPA and Ni(OH)₂‑BHPA. d Calibration plot.
Selectivity tests (Figure 6a) demonstrate that common interferents (lactose, sucrose, fructose, uric acid, ascorbic acid) produce less than 3.8 % interference, while the glucose signal remains at 98.1 % after the second addition. Long‑term stability (Figure 6b) shows that the sensor retains ≈ 93.5 % of its initial response after 2400 s. Reproducibility studies (Figures 6c–d) reveal relative standard deviations of 4.8 % for a single electrode and 5.3 % across five electrodes, confirming excellent consistency.
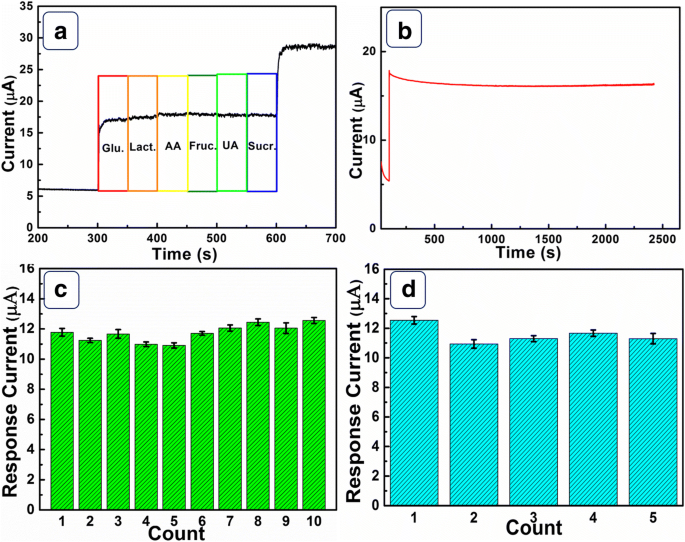
a Selectivity assessment. b Stability over 2400 s. c Ten repeat measurements of one electrode. d Reproducibility across five electrodes.
Conclusions
We have developed a facile, room‑temperature CEP strategy to fabricate uniform Ni(OH)₂ hollow‑porous cubes with a high SSA and well‑ordered diffusion channels. When applied as an electrochemical glucose sensor, the Ni(OH)₂‑HPA electrode delivers a sensitivity of 1843 µA mM⁻¹ cm⁻², a detection limit of 0.23 µM, and a response time of 1.4 s—outperforming the broken counterpart. The hollow‑porous architecture thus represents a scalable route for constructing high‑performance, enzyme‑free glucose sensors with practical analytical relevance.
Abbreviations
- AA:
l‑ascorbic acid
- BET:
Brunauer‑Emmett‑Teller
- CA:
Chronoamperometry
- CEP:
Coordinating etching and precipitating
- CNT:
Carbon nanotubes
- ECF:
Electrospun carbon nanofiber
- EIS:
Electrochemical impedance spectroscopy
- FESEM:
Field emission scanning electron microscope
- Fruc.:
Fructose
- GCE:
Glassy carbon electrode
- Glu.:
Glucose
- HPA:
Hollow porous architecture
- Lact.:
Lactose
- Ni(OH)2 BHPA:
Broken Ni(OH)₂ HPA
- oPPyNW:
Over‑oxidized polypyrrole nanowires
- PVP:
Polyvinyl pyrrolidone
- Rct:
Electron transfer resistance
- Rs:
Internal resistance
- RSD:
Relative standard deviation
- SAED:
Selected area electron diffraction
- SSA:
Specific surface area
- Sucr.:
Sucrose
- UA:
Uric acid
- XPS:
X‑ray photoelectron spectrometer
- XRD:
X‑ray diffraction
- Zw:
Warburg impedance
Nanomaterials
- Optimized Al₂O₃/MgO Emission Layers for Ultra‑High Gain Electron Multipliers
- Highly Sensitive Nonenzymatic Glucose Sensing with Hollow Porous Nickel Oxide
- Self‑Powered Bionic Cochlear Basilar‑Membrane Sensor Using Triboelectric Nanogenerator for High‑Resolution Frequency Selectivity
- High‑Performance NiO Flakes@CoMoO₄ Nanosheets Core‑Shell Electrode on Ni Foam for Advanced Supercapacitors
- MoS₂ Nanocomposite-Based Enzyme-Free Glucose Biosensors: A Cutting-Edge Approach
- Optimizing LaCoO3 with Sr Doping and rGO for Superior Energy Storage Performance
- Optimizing PCB Design for Manufacturing: Key Strategies & Best Practices
- Top 5 Design Strategies for Successful Reaction Injection Molding (RIM)
- Advanced TOMOPLEX Sensor Film for Real-Time Aerospace Monitoring
- Understanding Architecture Design: Principles, Practices, and Impact



