Self‑Powered Bionic Cochlear Basilar‑Membrane Sensor Using Triboelectric Nanogenerator for High‑Resolution Frequency Selectivity
Abstract
Sensorineural hearing loss remains the most prevalent chronic disability, affecting individuals of all ages. Traditional cochlear implants are bulky, require external power supplies, and limit patient comfort. We introduce a novel bionic cochlear sensor that emulates the basilar membrane’s frequency‑selective mechanics and harvests acoustic energy through a triboelectric nanogenerator (TENG). The device incorporates nine trapezoidally arranged silver electrodes on dual polytetrafluoroethylene (PTFE) membranes, achieving distinct resonance peaks across the audible spectrum (20–3000 Hz). Its self‑powered operation, derived from sound‑induced vibrations, offers a compact and user‑friendly alternative for hearing rehabilitation.
Background
Worldwide, sensorineural hearing loss arises from age, noise exposure, ototoxic drugs, and trauma, compromising frequency discrimination and daily communication. The cochlea’s basilar membrane separates sound waves by frequency and translates mechanical vibrations into electrical impulses that stimulate auditory nerves. Conventional implants convert acoustic signals to electrical pulses via an external processor and implanted electrode array, yet they burden patients with visible hardware and external power sources.
Recent micro‑ and nano‑scale acoustic sensors have attempted to replicate cochlear selectivity using piezoelectric or microbeam technologies, but they suffer from low voltage output, complex fabrication, and limited frequency coverage. Triboelectric nanogenerators (TENGs) offer a compelling alternative, converting mechanical motion into significant electrical signals with minimal complexity. Their sensitivity to frequency variations makes them ideal for designing a self‑powered, frequency‑selective cochlear substitute.
Methods/Experimental
The sensor design mirrors the cochlear basilar membrane’s trapezoidal geometry, allowing local resonance frequency to shift along the membrane. The device comprises two 120 mm × 60 mm × 4 mm acrylic plates with a central trapezoidal slit (baseline 30 mm, topline 10 mm, height 100 mm). Two 20 µm‑thick PTFE membranes, each 120 mm × 60 mm, are positioned on either side of a 20 µm Kapton spacer, which defines a narrow gap and sets the acoustic pressure threshold. Nine 4 × 8 mm silver electrodes (≈200 nm thick) are sputter‑deposited on the upper PTFE surface, numbered #1 (bottom) to #9 (top). The electrodes are 10 mm apart in‑plane, ensuring minimal mechanical interference.
Vibration responses were measured with a laser Doppler vibrometer (LDV) across 100–3000 Hz, while triboelectric voltage output was recorded using a preamplifier and oscilloscope. A calibrated speaker delivered sinusoidal acoustic waves at varying pressures. COMSOL Multiphysics simulations modeled membrane displacement patterns at selected frequencies (300 Hz, 1000 Hz, 2000 Hz) to validate experimental observations.
Conceptual diagram of the cochlear basilar membrane and its trapezoidal structure.
Results and Discussion
Increasing sound pressure linearly amplified both the vibration amplitude at electrodes #2, #5, and #8 and the corresponding triboelectric voltage (Fig. 3). This linearity confirms the sensor’s capability to quantify acoustic intensity through electrical readout.
Frequency‑dependent tests revealed distinct resonance peaks for each electrode: electrode #2 peaked at 200 Hz, #5 at 1030 Hz, and #8 at 1850 Hz, with peak voltages reaching 104 mV. The resonance shift toward higher‑numbered electrodes with increasing frequency reproduces the basilar membrane’s tonotopic organization (Fig. 4d). Both vibration amplitude and voltage output spectra overlapped closely, underscoring the sensor’s dual mechanical‑electrical transduction efficiency.
COMSOL simulations (Fig. 5) demonstrated that the PTFE membrane’s maximum displacement moves from the baseline toward the topline as frequency rises, corroborating experimental findings. The triboelectric mechanism operates by contact electrification between the upper PTFE and silver‑coated lower PTFE under sound pressure, followed by electrostatic induction as the membranes separate, generating measurable voltage pulses (Fig. 6).
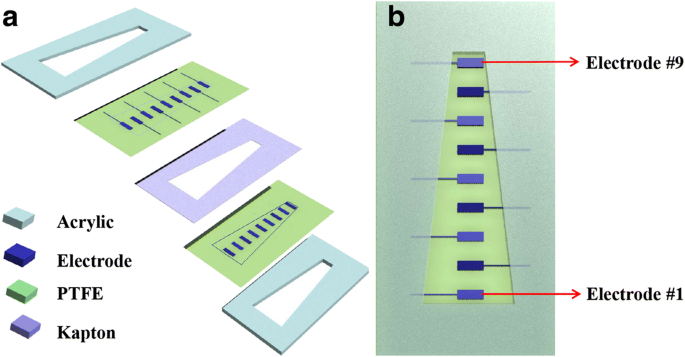
Device architecture: (a) 3‑D view of key components; (b) top view with electrode numbering.
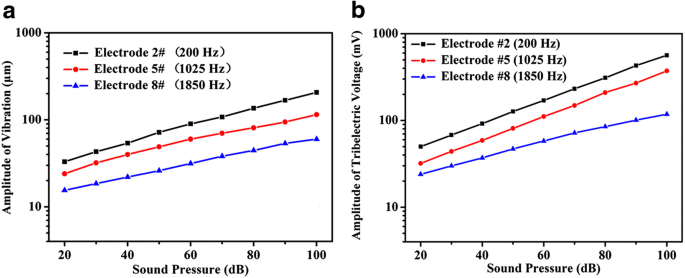
Relationship between acoustic pressure and (a) vibration amplitude, (b) triboelectric voltage.
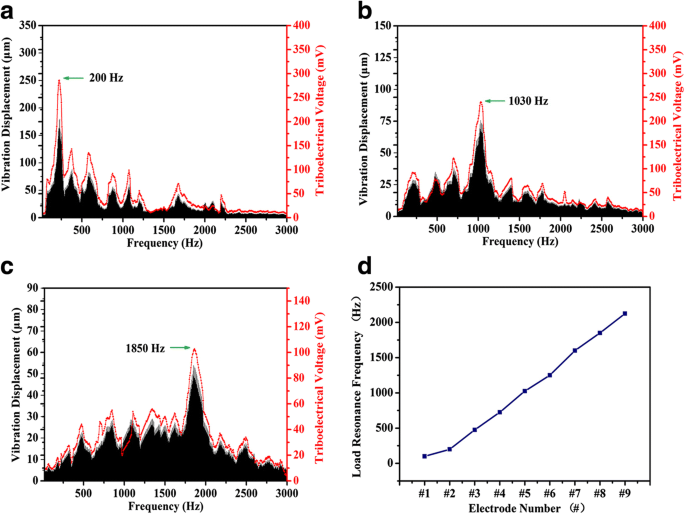
Frequency response of electrodes #2, #5, and #8: vibration amplitude (black) and triboelectric voltage (red). (d) Resonance frequency vs. electrode number.
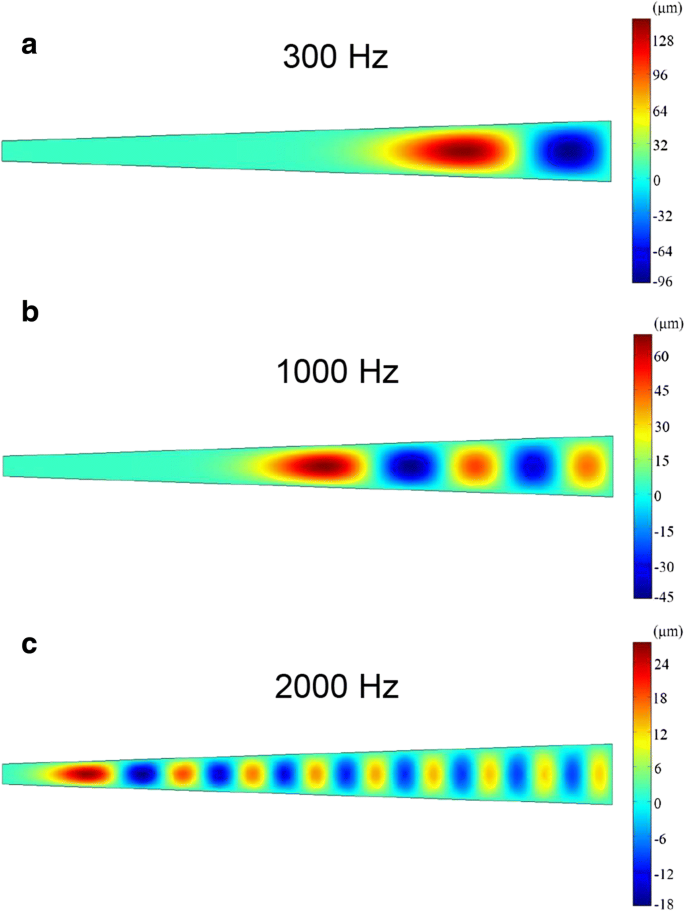
COMSOL simulation of PTFE membrane displacement at 300 Hz, 1000 Hz, and 2000 Hz.
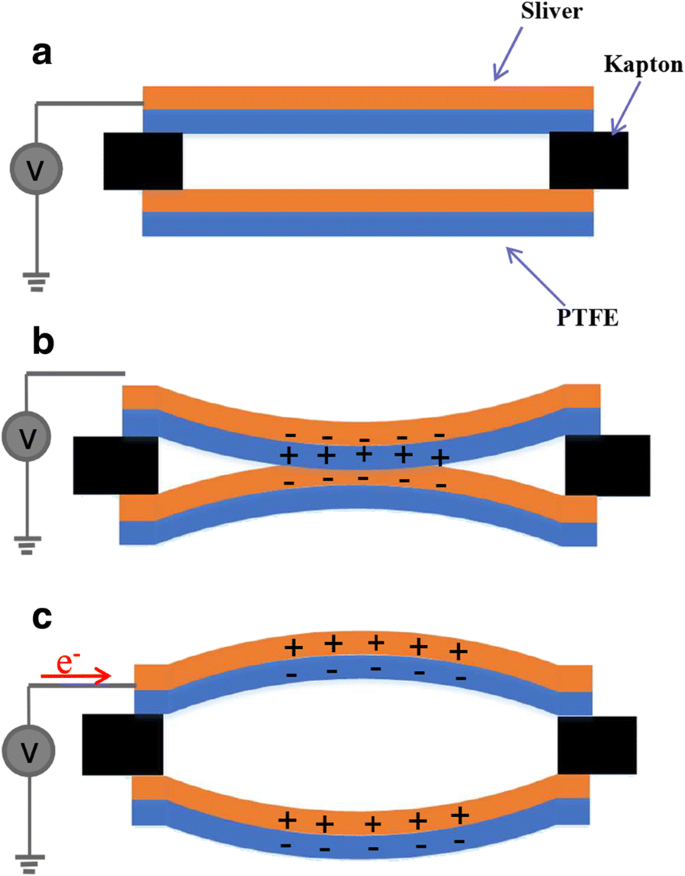
Working principle: (a) rest state, (b) contact state under sound, (c) separation state generating voltage.
Conclusions
We have demonstrated a self‑powered, frequency‑selective cochlear surrogate that replicates the basilar membrane’s mechanics using a trapezoidal PTFE membrane integrated with a triboelectric nanogenerator. The nine‑electrode array provides distinct resonance peaks across the audible range, enabling precise frequency discrimination. Experimental and finite‑element results confirm that local vibration amplitude and triboelectric voltage peak at corresponding frequencies, mimicking cochlear tonotopy. This low‑cost, implant‑ready platform offers a promising alternative for treating sensorineural hearing loss without the need for external power or bulky hardware.
Abbreviations
- LDV
Laser Doppler vibrometer measuring system
- PM
Polytetrafluoroethylene membrane
- PTFE
Polytetrafluoroethylene
- TENG
Triboelectric nanogenerator
Nanomaterials
- Highly Stretchable, Electrically Conductive TPU–PANI Nanofibrous Membrane for Advanced Flexible Strain Sensors
- High‑Sensitivity Flexible Strain Sensor Using Poly(vinylidene fluoride) Piezoelectric Film
- Eco‑Friendly Starch‑Paper Triboelectric Nanogenerator for Real‑Time Human Sweat Sensing
- Ni(OH)₂ Hollow‑Porous Architecture Enables Ultra‑Sensitive, Enzyme‑Free Glucose Detection
- Ultra-Stable Pt–Ni Jagged Nanowire Sensor for Highly Sensitive Caffeic Acid Detection
- Hydrophilic‑Substrate‑Driven Self‑Polarization of PVDF for Ultra‑Low Noise Pyroelectric Sensors
- Wearable Wireless Triboelectric Nanogenerator for Real‑Time Respiratory Monitoring
- 2D Woven Core‑Shell Fiber Triboelectric Nanogenerator Harvests Body Motion into 6 V, 575 nA, Powering LEDs and Capacitors
- Binder‑Free Electrospun Fiber Electrodes for High‑Performance Lithium‑Ion Batteries via Simple Rolling Assembly
- Advanced TOMOPLEX Sensor Film for Real-Time Aerospace Monitoring



