Enhanced Visible‑Light Photocatalysis via Cu2−xSe‑Modified Monoclinic BiVO4: 15.8‑Fold Activity Boost
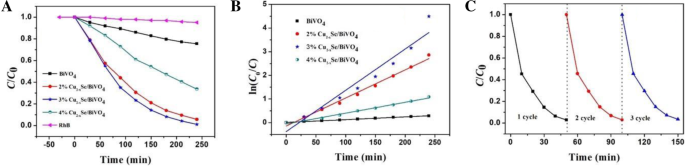
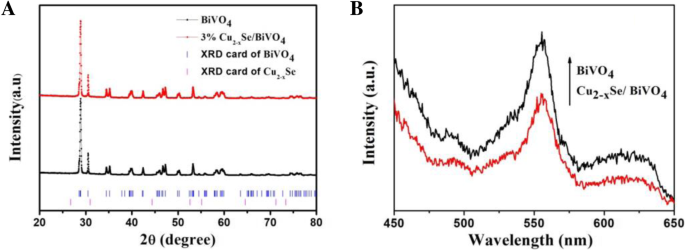
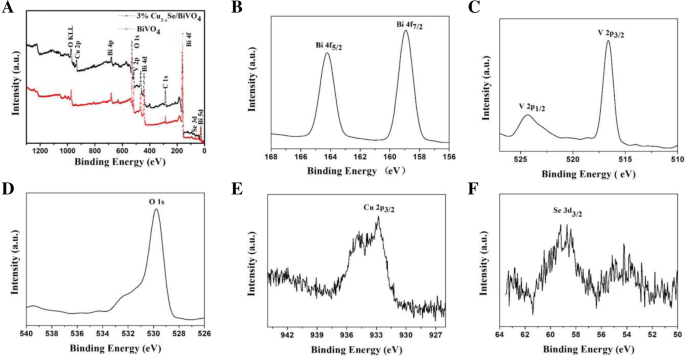
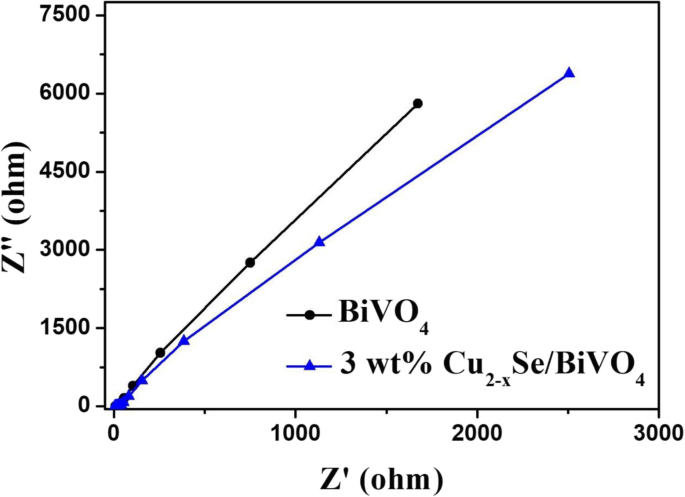
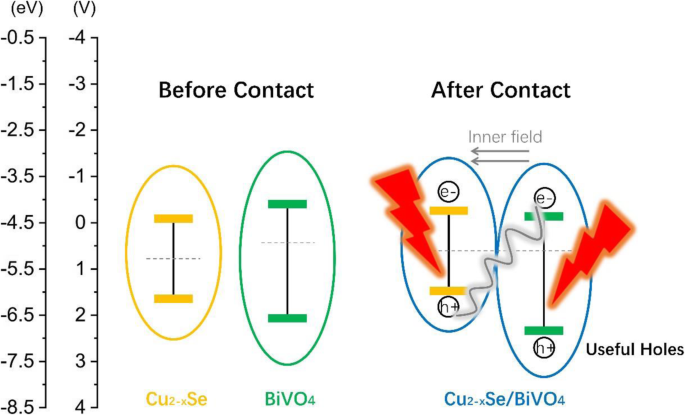
BiVO4 is a promising visible‑light photocatalyst, yet its performance is hampered by rapid electron–hole recombination. In this study we engineered a Cu2−xSe/BiVO4 heterostructure that leverages band‑structure mismatch to generate an internal electric field, driving efficient charge separation. The Cu2−xSe component, possessing a larger work function and lower Fermi level than BiVO4, attracts photogenerated electrons while donating holes in reverse. Photocatalytic tests on Rhodamine B (RhB) under visible light (>420 nm) reveal that a 3 wt% Cu2−xSe loading boosts activity by 15.8 times relative to pristine BiVO4. Adding a modest amount of H2O2 drives complete RhB degradation within 50 min, demonstrating the composite’s practical potential for environmental remediation. Industrial growth has intensified environmental pollution, prompting the need for sustainable remediation technologies. Photocatalytic decomposition of organic contaminants using solar energy is an attractive, green solution. Bi‑based semiconductors, especially monoclinic BiVO4 with a 2.4 eV band gap, absorb visible light efficiently and exhibit promising photocatalytic performance. However, fast electron–hole recombination limits their practical use. A proven strategy to mitigate this issue is the construction of heterojunctions that align the band edges of two semiconductors, thereby promoting charge separation. Cu2−xSe, a p‑type semiconductor with an indirect band gap of 1.4 eV, is well suited for visible‑light absorption. When combined with BiVO4, the higher work function of Cu2−xSe causes electrons to migrate from BiVO4 to Cu2−xSe, while holes move in the opposite direction. This charge redistribution establishes an internal electric field that facilitates selective recombination of the less energetic carriers, leaving highly oxidative holes in BiVO4 capable of generating hydroxyl radicals (•OH) that degrade complex organic molecules. Here we report, for the first time, the synthesis of Cu2−xSe/BiVO4 composites and their application in RhB photodegradation under visible‑light irradiation. The 3 wt% Cu2−xSe/BiVO4 sample exhibited a 15.8‑fold increase in activity compared with pure BiVO4, and complete RhB removal was achieved in 50 min when a small amount of H2O2 was introduced. These results establish Cu2−xSe as an effective cocatalyst for advanced composite photocatalysts. BiVO4 was prepared by a standard chemical‑precipitation route. Cu2−xSe was synthesized following our previously reported protocol. The two powders were dispersed in ethanol, stirred at 60 °C for 4 h, then heated to 80 °C to evaporate the solvent. Finally, the mixture was calcined at 160 °C for 6 h under flowing nitrogen to form the Cu2−xSe/BiVO4 composite. The schematic of this process is shown in Figure 1. The schematic diagram of formation for Cu2−xSe/BiVO4 composite X‑ray diffraction (XRD) was performed on a PANalytical X’pert Pro diffractometer using Cu Kα radiation. Morphology was examined by a Hitachi S‑4800 scanning electron microscope (SEM). X‑ray photoelectron spectroscopy (XPS) data were collected on a PHI‑5300 system. Photoluminescence (PL) spectra were recorded on a Cary Eclipse spectrophotometer. The photocatalytic activity was assessed in an XPA photochemical reactor. A 500 W Xe lamp with a 420 nm cut‑off filter simulated natural sunlight. In a typical experiment, 60 mg of the composite was dispersed in 60 mL of a 5 µM RhB solution. After 2 h of dark stirring to reach adsorption equilibrium, the suspension was illuminated; 6 mL aliquots were withdrawn every 10 min, centrifuged, and analyzed by a Shimadzu UV‑2450 spectrometer. Photocurrent measurements were carried out on a CHI 660E workstation. The working electrode was prepared by drop‑casting 10 mg of the catalyst with 20 µL Nafion onto a 0.196 cm2 ITO glass, followed by 1 h at 200 °C. A Pt foil served as the counter electrode, a Hg/HgCl₂ reference, and 0.5 M Na₂SO₄ as the electrolyte. Illumination matched the photocatalytic conditions (500 W Xe lamp, >420 nm). Figure 2a demonstrates that Cu2−xSe incorporation markedly improves RhB photodegradation. The optimal loading is 3 wt%, achieving the highest degradation rate constant (k = 0.0174 min−1), which is 15.8 times that of bare BiVO4 (k = 0.0011 min−1). Figure 2c shows that, with 103 µL H₂O₂ per 100 mL solution, the 3 wt% composite fully degrades RhB within 50 min, and the activity remains stable after three cycles. a Photocatalytic degradation of RhB over Cu2−xSe/BiVO4. b Degradation rate constants for varying Cu2−xSe contents. c Recycling tests with H₂O₂. SEM images (Figure 3) reveal that BiVO4 forms hexagonal bulk particles (0.2–1 µm), while Cu2−xSe sheets (≈300 nm thick, 4 µm long) are uniformly dispersed on BiVO4 surfaces. XPS confirms the presence of Cu2−xSe and the oxidation states of all elements (Figure 5). The SEM photograph of BiVO4 (a) and Cu2−xSe/BiVO4 (b) XRD patterns (Figure 4a) confirm the monoclinic phase of BiVO4 remains unchanged after Cu2−xSe loading, as the Cu content is too low for distinct diffraction peaks. PL spectra (Figure 4b) show a reduced emission intensity for the composite, indicating enhanced electron–hole separation. Electrochemical impedance spectroscopy (EIS, Figure 6) demonstrates a smaller Nyquist arc for the composite, reflecting lower charge‑transfer resistance and faster interfacial electron transport. The XRD data for BiVO4 and 3% Cu2−xSe/BiVO4 (a), the PL spectra for BiVO4 and Cu2−xSe/BiVO4 composites (b) The XPS spectra of Cu2−xSe/BiVO4 composite. a Survey, b Bi, c V, d O, e Cu, and f Se The EIS for BiVO4 and Cu2−xSe/BiVO4 under visible light irradiation in 0.5 M Na₂SO₄ solution The superior photocatalytic performance arises from an S‑scheme heterojunction formed between Cu2−xSe and BiVO4. Band alignment drives electrons from BiVO4 to Cu2−xSe and holes in the opposite direction, creating an internal field that selectively recombines the lower‑energy carriers. Consequently, high‑energy holes remain in BiVO4, efficiently generating •OH radicals that oxidize RhB. This mechanism explains the dramatic activity enhancement and the stability observed in repeated runs. The schematic diagram of photocatalytic mechanism We have successfully synthesized Cu2−xSe/BiVO4 composites that exhibit markedly enhanced visible‑light photocatalytic activity. The 3 wt% Cu2−xSe/BiVO4 sample achieves a 15.8‑fold increase in RhB degradation rate compared to pure BiVO4, and complete removal of RhB is attained within 50 min when a small amount of H₂O₂ is added. SEM, XPS, PL, and EIS analyses confirm efficient charge separation, reduced recombination, and lower interfacial resistance in the composite. These findings establish Cu2−xSe as an effective cocatalyst and provide a blueprint for designing high‑performance semiconductor photocatalysts for environmental applications. Rhodamine B Scanning electron microscope X‑ray diffractionAbstract
Introduction
Methods
Preparation of Cu2−xSe/BiVO4 Composites
Characterization
Photocatalytic Reaction
Photoelectrochemical Measurements
Results and Discussion

Conclusion
Abbreviations
Nanomaterials
- Ag Nanoparticle-Enhanced BiV1‑xMoxO4 Composites: Synergistic Photocatalytic Performance under Visible Light
- Enhanced Photocatalytic Degradation of Rhodamine B Using an ATP/TiO₂/Ag₃PO₄ Ternary Nanocomposite Under Simulated Solar Light
- Ag3PO4/BiFeO3 Heterojunctions: Superior Visible‑Light Photocatalytic Degradation of Acid Orange 7
- Visible‑Light Photocathodic Protection of 304 Stainless Steel Using Bi₂Se₃‑Sensitized TiO₂ Nanotube Films
- Ag/BiPbO₂Cl Nanosheet Composites: 3.6‑Fold Enhancement of Visible‑Light Photocatalytic Activity
- Enhanced Visible‑Light Photocatalysis via In‑Situ Z‑Scheme BiOBr0.3I0.7/Ag/AgI Nanocomposites
- Partially BiVO4-Modified ZnO Porous Nanosheets: Solar‑Driven Photocatalysis with Superior Charge Separation
- Enhanced Visible-Light Photocatalysis by Anchoring Plasmonic Ag@AgCl Nanocrystals on ZnCo₂O₄ Microspheres
- High‑Performance Flexible Photocatalytic Paper: Cu2O and Ag Nanoparticle‑Decorated ZnO Nanorods for Visible‑Light Degradation of Organic Dyes
- Enhanced Visible‑Light Photocatalysis via Z‑Scheme Ag3PO4/TiO2 Heterojunctions



