Ag/BiPbO₂Cl Nanosheet Composites: 3.6‑Fold Enhancement of Visible‑Light Photocatalytic Activity
Abstract
We report the hydrothermal synthesis and photo‑reduction of Ag/BiPbO2Cl nanosheet composites. TEM, XRD, and UV‑Vis diffuse‑reflection spectroscopy confirm the morphology, crystal structure, and optical response of the materials. A 0.5 wt % Ag loading yields photocatalytic activity 3.6 times higher than pure BiPbO2Cl when degrading methyl orange (MO) under visible light. The improvement arises from an internal electromagnetic field, extended visible‑light absorption, superior electrical conductivity, and a lower Ag Fermi level.
Background
Environmental pollution from organic contaminants has driven the development of semiconductor photocatalysts, notably ZnO and TiO2, which mainly absorb ultraviolet light. To harness the abundant visible spectrum, bismuth‑based semiconductors such as BiPbO2Cl—known for a narrow band gap, internal electric fields, and hybrid band structures—are attractive, yet suffer from rapid electron–hole recombination.
Integrating noble metals or graphene with photocatalysts can mitigate recombination by acting as electron sinks. Ag, Au, and Pt have demonstrated such effects. Here, we combine Ag with BiPbO2Cl to enhance visible‑light photocatalysis.
Methods
Preparation of Ag/BiPbO2Cl Nanosheet Composites
BiPbO2Cl nanosheets were synthesized via a one‑step hydrothermal route (see ref. 13). For Ag loading, 1 mmol BiPbO2Cl was dispersed in 20 mL deionized water, then AgNO3 was added at 0.25, 0.5, or 0.75 wt %. The mixture was irradiated with a 500‑W Xe lamp (cut‑off filter <420 nm) under stirring at room temperature for 3 h. The resulting powders were washed, dried at 80 °C, and stored for analysis.
Photocatalytic Activities
Photocatalytic performance was evaluated using a 500‑W Xe lamp (cut‑off filter >420 nm) in an XPA photochemical system. 50 mg of catalyst was dispersed in 50 mL 10 mg L−1 MO solution, stirred in the dark for 1 h to reach adsorption equilibrium, then illuminated. UV‑Vis spectra were recorded with a Shimadzu UV‑2700.
Sample Characterization
XRD (Cu Kα, 1.54178 Å) was performed on a PANalytical X’Pert Pro. SEM (Hitachi S‑4800) and TEM (JEOL JEM‑2011) examined morphology. UV‑Vis diffuse‑reflection spectra were obtained on a Shimadzu UV‑2450. XPS (Pekin Elmer PHI‑5300) provided surface chemistry, and PL spectra (Shimadzu RF‑5301, 320 nm excitation) assessed charge‑carrier recombination.
Results and Discussion
MO degradation under visible light (>420 nm) showed a clear dependence on Ag content. The 0.5 wt % Ag/BiPbO2Cl composite achieved the highest activity, with a degradation rate constant of 0.0158 min−1, 3.6 times that of pristine BiPbO2Cl (0.0044 min−1) (Fig. 1a,b). Excess Ag reduces the BiPbO2Cl surface area and thus diminishes photocatalytic sites.
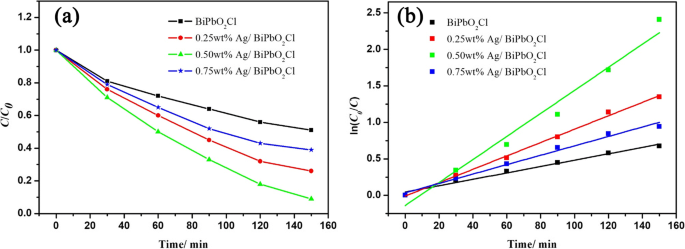
a Photocatalytic degradation of MO; b Degradation kinetics.
SEM images reveal BiPbO2Cl as ~12 nm‑thick nanosheets, while Ag nanoparticles (~10 nm) are uniformly distributed on the composite surface (Fig. 2). HRTEM confirms the presence of Ag (Fig. 2c). XRD patterns (Fig. 2d) show no new phases, indicating the low Ag content. EDS mapping (Fig. 3) confirms homogeneous Ag distribution.
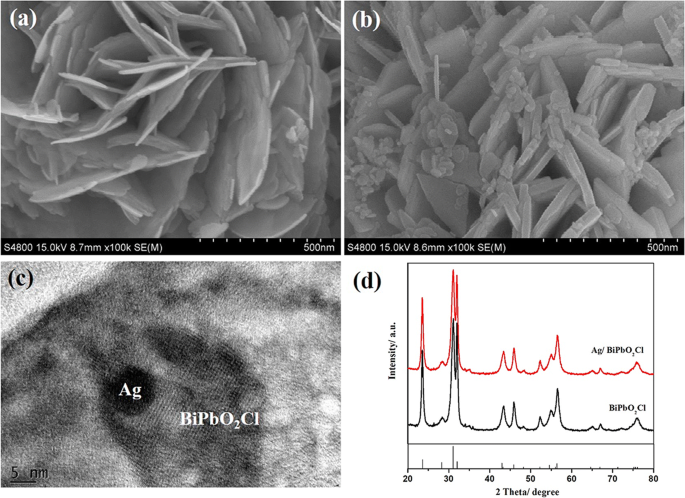
SEM of BiPbO2Cl (a) and 0.5 wt % Ag/BiPbO2Cl (b). c HRTEM; d XRD.

EDS elemental mapping of Ag/BiPbO2Cl.
XPS spectra (Fig. 4) confirm the presence of Bi3+, Pb2+, O2−, Cl−, and metallic Ag (Ag0). The Ag 3d peaks at 368.1 and 374.3 eV match the literature value for Ag0 (ref. 23).
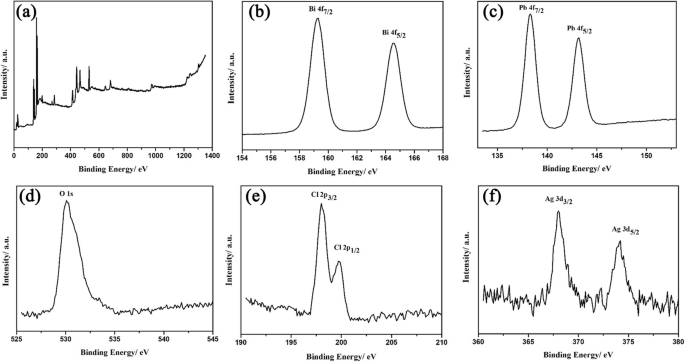
The XPS spectra of Ag/BiPbO2Cl composites: a survey; b Bi 4f; c Pb 4f; d O 1s; e Cl 2p; f Ag 3d.
UV‑Vis absorption (Fig. 5a) shows enhanced visible‑light absorption (450–800 nm) due to surface plasmon resonance of Ag, increasing the light‑harvesting range. The band gap narrows from 2.05 eV (pure BiPbO2Cl) to 1.68 eV (0.5 wt % Ag) (Fig. 5b). PL spectra (Fig. 5c) display a marked intensity reduction, evidencing suppressed electron–hole recombination.
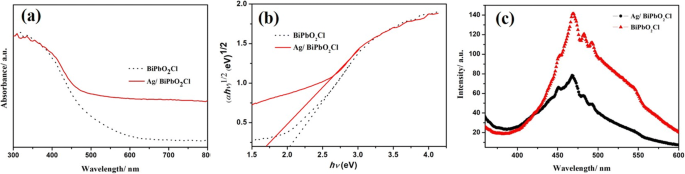
UV‑Vis absorption (a) and PL emission (b) of BiPbO2Cl and 0.5 wt % Ag/BiPbO2Cl.
Mechanistically, the Ag loading extends visible‑light response, establishes an internal electromagnetic field by aligning Fermi levels (Ag lower than BiPbO2Cl), and enhances conductivity. Electrons excited in BiPbO2Cl rapidly transfer to Ag, where they reduce O2 to •O2⁻, while holes oxidize water to •OH (Fig. 6). The combined action of these radicals drives MO decomposition into CO2 and H2O.
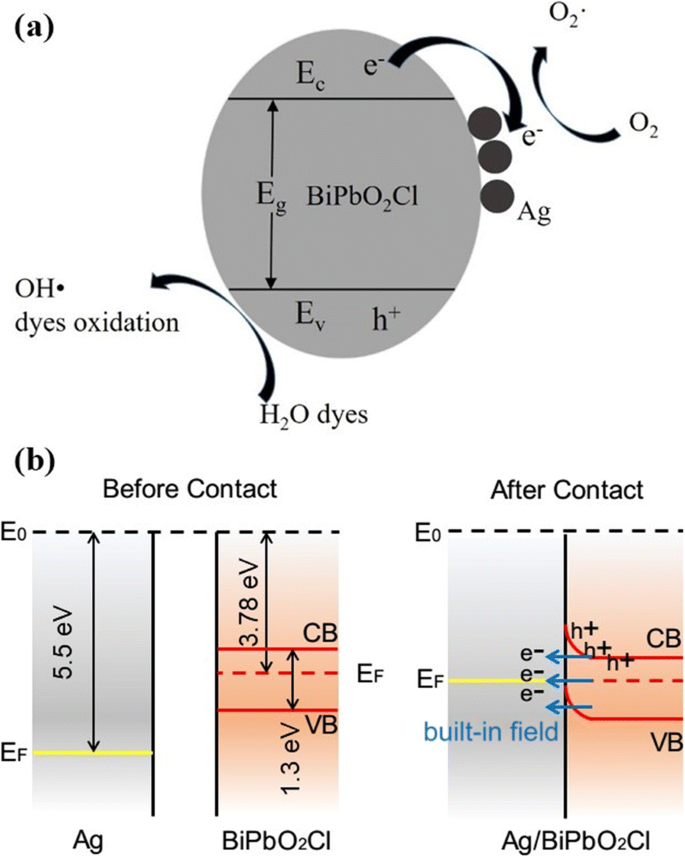
a Photocatalytic mechanism; b Band alignment at the Ag/BiPbO2Cl interface.
Conclusions
Hydrothermal synthesis followed by photo‑reduction produced Ag/BiPbO2Cl nanosheet composites with 0.5 wt % Ag loading that outperform pure BiPbO2Cl by 3.6× in visible‑light photocatalytic degradation of MO. The enhanced performance stems from extended light absorption, reduced charge‑carrier recombination, and the creation of an internal electromagnetic field due to Ag’s lower Fermi level and superior conductivity.
Nanomaterials
- Thermoset Composite Properties: A Game Changer for HVAC Systems
- Enhanced Visible‑Light Photocatalysis of Methylene Blue and Phenol with N‑Doped ZnO/g‑C₃N₄ Nanocomposites
- Enhanced Visible‑Light Photocatalytic Degradation of Rhodamine B Using Bi<sub>4</sub>Ti<sub>3</sub>O<sub>12</sub>/Ag<sub>3</sub>PO<sub>4</sub> Heterojunction Nanocomposites
- Ag Nanoparticle-Enhanced BiV1‑xMoxO4 Composites: Synergistic Photocatalytic Performance under Visible Light
- Efficient One‑Step Photo‑Ultrasonic Synthesis of rGO/Ag₃PO₄ Quantum‑Dot Composites for Enhanced Visible‑Light Photocatalysis
- Ag3PO4/BiFeO3 Heterojunctions: Superior Visible‑Light Photocatalytic Degradation of Acid Orange 7
- Enhanced Visible‑Light Photocatalysis via In‑Situ Z‑Scheme BiOBr0.3I0.7/Ag/AgI Nanocomposites
- Enhanced Visible‑Light Photocatalysis via Cu2−xSe‑Modified Monoclinic BiVO4: 15.8‑Fold Activity Boost
- Hierarchical Au@CdS‑CdS Nanoflowers: Stepwise Fabrication and Superior Photocatalytic Performance
- Enhanced Visible-Light Photocatalysis by Anchoring Plasmonic Ag@AgCl Nanocrystals on ZnCo₂O₄ Microspheres



