Paramagnetic Behavior of Fullerene-Derived Nanomaterials and Their Polymer Composites: A Comprehensive EPR Study
Abstract
Electron paramagnetic resonance (EPR) was employed to investigate the paramagnetic evolution of fullerene soot (FS), fullerene black (FB), and their composites with Phenylon C-2 (PhC‑2). For the first time, a pronounced amplification of EPR signals was recorded in FB, FS, and composite samples subjected to vacuum pumping at temperatures between 20 °C and 300 °C. The effect is attributed to interactions between carbon defects and adsorbed gases, predominantly oxygen.
The paramagnetic ensemble in FB, FS, and the composites is heterogeneous, comprising three distinct spin subsystems (1, 2, and 3). Each contributes a characteristic signal—L1, L2, and L3—to the overall EPR profile. The dominant, broad L3 signal originates from two‑dimensional (2D) electrons localized on the surfaces of carbon flakes. Theoretical modeling of the L3 line shape yielded decay constants for each component following exposure to ambient air. Bulk composites exhibit a markedly slower signal decay, reflecting their reduced gas permeability at room temperature.
Background
Carbon‑based nanomaterials—graphene, nanotubes, fullerenes, onion‑like carbon (OLC), nanodiamonds (ND), and carbon dots—have attracted intense research interest over the last decade. Their diverse sizes and hybrid sp1, sp2, and sp3 bonding configurations underpin unique electronic, mechanical, and chemical properties that are exploited across materials science, energy storage, biomedicine, and environmental remediation [1–10].
Fullerene derivatives, in particular, occupy a pivotal position among nanocarbon materials due to their versatile synthesis routes and functionalization capabilities. They can undergo structural transformations, such as the conversion of soot or fullerene black into OLC, or the integration of ND within OLC shells [11–14].
Recent developments have expanded fullerene applications into biology, medicine, nanocomposite fabrication, electromagnetic shielding, and beyond [15–20]. Their physical‑chemical behavior is governed by electronic structure, defect density, surface area, and other parameters. For instance, OLC nanoparticles synthesized in the presence of oxygen exhibit superior microwave‑absorption performance [18]. The high defect density and non‑planar “pyramidalization” of fullerene‑type structures markedly influence reactivity [21–24].
EPR spectroscopy is a primary tool for probing electron spin dynamics in fullerene‑like materials. Previous studies of FS and FB revealed EPR signatures with g‑values ranging from 2.0022 to 2.0023 and linewidths ΔHpp≈2 G [25–28]. Paramagnetic radical concentrations were estimated at Ns≈1021 g−1 for FS and 3 × 1018 g−1 for FB [25,27]. These parameters remained largely unchanged in the presence of molecular oxygen, except for FB, where Ns increased by an order of magnitude after evacuation at 150 °C [27].
Understanding the interaction of fullerene‑derived materials with oxygen is essential, especially in light of recent reports linking EPR behavior to gas‑defect coupling [29,30]. The present work aims to elucidate the nature of paramagnetic defects in FS and FB, the mechanisms of oxygen interaction, and the influence of a phenolic polyamide matrix (PhC‑2) on these processes. PhC‑2, characterized by strong hydrogen‑bonded interactions, has shown promise for enhancing heat resistance and mechanical strength in space‑grade composites [31,32].
Methods
Fullerene C60, FS, and FB were sourced from NeoTechProduct (St. Petersburg, Russia) and used without further modification. FS was produced via arc‑evaporation of graphite, yielding a black powder with a bulk density of ~0.25 g cm−3 and ~10 % fullerene content. FB was derived by extracting fullerenes from FS using o‑xylol followed by steam post‑treatment, resulting in a product with ≤0.3 % C60 [https://www.neotechproduct.ru/main_page].
The PhC‑2 matrix is a linear heterocyclic copolymer containing amide groups (–HNCO–) flanked by phenyl rings, synthesized by emulsion polycondensation of metaphenylene‑diamine with a 3:2 molar mixture of isophthalic and terephthalic anhydrides.
Composite samples (PhC‑2/FS and PhC‑2/FB) were prepared by mixing in a rotating electromagnetic field, followed by compression molding at 598 K and 40 MPa. Filler loadings were 1.5 wt.% and 3 wt.%.
EPR measurements were performed at X‑band (ν ≈ 9.4 GHz) using a Radiopan X‑2244 spectrometer with 100 kHz field modulation. The g‑factor accuracy was ±2 × 10−4 for linewidths ΔHpp≤10 G. Spin density Ns was accurate to ±50% absolute and ±20% relative. Samples were studied in ambient air and under controlled oxygen partial pressures achieved by vacuum pumping at temperatures ranging from 20 °C to 170 °C. After evacuation, samples were transferred to the spectrometer cavity without altering the vacuum conditions.
Results and Discussion
Figure 1a displays the room‑temperature EPR spectra of C60, FS, and FB. All spectra share a g‑factor of 2.0024 ± 2 × 10−4. C60 shows a single Lorentzian line, whereas FS and FB require a sum of two Lorentzian components. Spin concentrations and relative contributions are tabulated in Table 1. The inset presents the FB spectrum at 30 K.
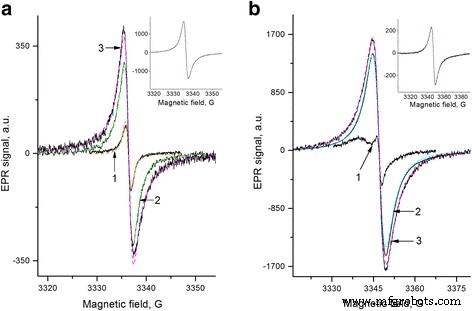
EPR spectra of fullerene, FS, FB, and Phenylon C‑2 composites. a. Initial samples: 1 = C60, 2 = FS, 3 = FB at room temperature. Dashed lines are fitted signals (Table 1). ν = 9350 MHz. Inset: FB spectrum at 30 K. b. Phenylon C‑2 composites (3 % filler): 1 = C60, 2 = FS, 3 = FB. The supplementary broad line (1) belongs to Phenylon C‑2. ν = 9375 MHz, gain ×5. Inset: PhC‑2/FS at 30 K (fitted).
Figure 1b shows the ESR spectra of Phenylon C‑2 composites with 3 % C60, FS, and FB. The inset displays the FB composite at 30 K. Parameters extracted from fitting are provided in Table 1. All samples were also examined under vacuum at temperatures 20–300 °C.
Figure 2a reveals a substantial EPR intensity increase for FS upon evacuation at room temperature. Evacuation at higher temperatures amplifies the signal by over 30×, primarily due to broad wings in the spectrum (Table 1). A comparable, albeit weaker, effect is observed in composites after high‑temperature evacuation (Figure 2b, Table 1).
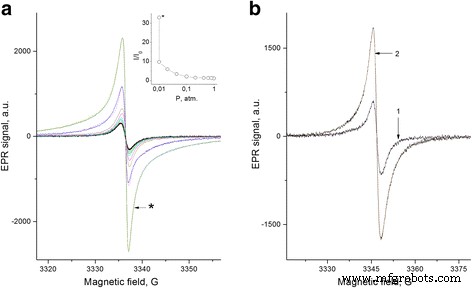
EPR spectra of FS under varying oxygen pressures and PhC‑2/FS composites. a. Pressure series (1–0.001 atm) during 0.5 h pumping at 160 °C; dashed line—fit with 3 Lorentzians. Inset: total EPR intensity vs. oxygen pressure. b. PhC‑2/FS before (1) and after (2) 1 h pumping at 160 °C. Dashed lines—fit (Table 1).
The decay of the signal upon re‑exposure to ambient air was monitored for FB after 0.5 h evacuation at 300 °C. Figure 3 separates the spectrum into L1, L2, and L3 components, each characterized by distinct linewidths (0.9, 3.0, and 24 G). L3 was modeled using the theoretical 2D electron spin system (see Discussion).
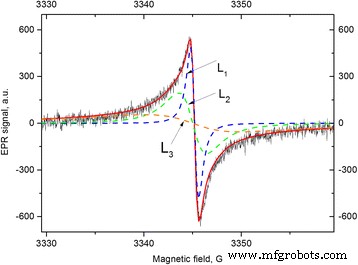
Decomposition of the FB EPR spectrum after evacuation. Components L1, L2, and L3 are labeled. ν = 9375 MHz.
Figure 4 tracks the intensity decay of L1, L2, and L3 over time after air contact. The initial rapid decline (seconds to minutes) is followed by a slower, hours‑long relaxation toward equilibrium.
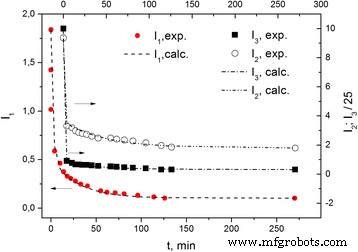
Decay of L1, L2, and L3 intensities after air exposure. t = 0 corresponds to the state in Figure 3.
Bulk composites (∼1.5 × 3 × 3 mm3) exhibit a markedly slower decay (Figure 5), with characteristic times exceeding those of powders by an order of magnitude due to limited gas permeability.
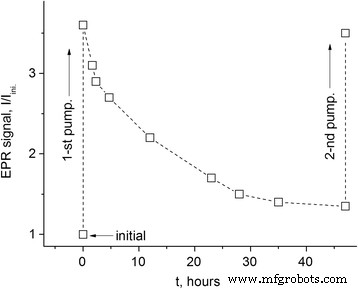
Decay of the total EPR signal in a bulk PhC‑2/FS composite after 1 h evacuation at 160 °C.
Annealing FS at 550 °C under weak vacuum (Figure 6) yielded a drastically different EPR profile: a single Lorentzian line with ΔH≈7–8 G and negligible intensity change upon subsequent evacuation. This contrasts with the pronounced pumping‑out effect observed at 20–300 °C, underscoring the temperature‑dependent evolution of paramagnetic characteristics [33,34].
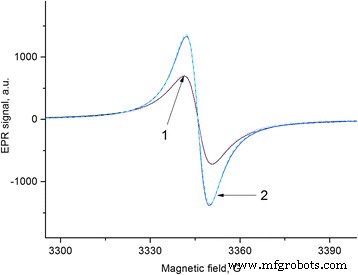
EPR spectra of FS before and after 1 h annealing at 550 °C, and after 24 h ambient exposure.
Detailed analysis (Section 6) elucidates the origins of L1, L2, and L3. L1, with g = 2.0024 and ΔH = 0.9 G, arises from fullerene‑derived sp2/sp3 defect sites (C120O). L2, broader (ΔH ≈ 3 G) and g = 2.0025, is attributed to edge sp3 dangling bonds on carbon flakes. L3, the broadest (ΔH ≈ 24 G) and g = 2.0025, originates from 2D electrons on the flake surfaces, consistent with dipole‑dipole broadening indicative of ~1 nm spin separations. The time‑dependent intensities of each component fit bi‑exponential decays, reflecting distinct binding energies of adsorbed oxygen to the various defect sites.
Composite behavior (Section 7) shows that the gas‑permeability of PhC‑2 dramatically slows the pumping‑out effect compared to the fillers alone. The L3 component remains prominent in composites, yet its amplitude and linewidth are moderated by the polymer matrix. This suggests that the matrix can mitigate oxygen diffusion and preserve the paramagnetic landscape.
Conclusions
Fullerene soot and fullerene black exhibit strong, reversible interactions with environmental gases, leading to near‑complete quenching (~95 %) of paramagnetic signals upon air exposure. Evacuation between 20 °C and 300 °C restores the full EPR signature, revealing a three‑component spectrum linked to specific structural features: L1 (fullerene‑derived defects), L2 (edge sp3 sites), and L3 (2D surface electrons). Theoretical modeling of L3 aligns well with experimental data. Decay kinetics of each component were quantified, demonstrating rapid initial relaxation followed by slower, long‑term equilibration.
In bulk PhC‑2 composites, the same phenomena occur but with reduced clarity due to limited oxygen diffusion. Nonetheless, the presence of fullerene fillers enhances mechanical and electronic performance while preserving a distinctive paramagnetic response.
These findings open avenues for deploying fullerene‑based nanomaterials as high‑sensitivity oxygen sensors in biomedical and environmental applications.
Abbreviations
- EPR:
Electron paramagnetic resonance
- ESR:
Electron spin resonance
- FB:
Fullerene black
- FS:
Fullerene soot
- ND:
Nano‑diamond
- Ns:
Spin concentration
- OLC:
Onion‑like carbon
- PC:
Paramagnetic center
- PhC-2:
Phenylon C‑2
- RT:
Room temperature
Nanomaterials
- Graphene‑Polymer Composites for High‑Performance Supercapacitors: A Comprehensive Review
- Water‑Soluble Antimony Sulfide Quantum Dots: Efficient Synthesis and Superior Photo‑Electric Performance
- How Synthesis Route Shapes the Structure, Magnetism, and Hyperthermia Performance of La₁₋ₓSrₓMnO₃ Nanoparticles
- How Nanoparticle Properties Drive Their Toxicity: A Comprehensive Review
- Zinc Oxide Nanoparticles: Antimicrobial Properties, Mechanisms, and Applications
- PVP‑Enhanced SnO₂ Nanoflowers Deliver Ultra‑Fast, Highly Selective H₂S Sensing
- Influence of Reactant Stoichiometry on Morphology and Structure of CH3NH3PbI3 Perovskite Films
- Impact of Zirconia Surface Modifications on Polymer Composite Tensile Strength
- Size-Dependent Photoelectrical Behavior of Individual Silicon Nanowires
- Exploring Metal Types: Key Properties and Applications



