Enhanced Photocatalytic Degradation of Rhodamine B Using an ATP/TiO₂/Ag₃PO₄ Ternary Nanocomposite Under Simulated Solar Light
Abstract
A highly efficient ternary photocatalyst was engineered by integrating silver orthophosphate (Ag3PO4), attapulgite (ATP), and TiO2. The resulting heterojunction promotes effective separation of photogenerated electrons and holes, markedly boosting photocatalytic performance. Structural analysis via SEM, XRD, and UV‑vis diffuse reflectance confirmed a lath‑particle morphology and the presence of all three components. Compared to binary composites, ATP/TiO2/Ag3PO4 achieved superior Rhodamine B (RhB) degradation under simulated solar irradiation, while simultaneously enhancing the stability of Ag3PO4 and reducing its required loading. This work offers a practical blueprint for designing Ag‑based photocatalysts with lower cost and higher durability.
Background
Organic pollutants pose a persistent threat to environmental health, and photocatalytic degradation has emerged as a promising, environmentally benign remediation strategy. Since Fujishima’s pioneering discovery of TiO2-driven water splitting in 1972, TiO2 has been extensively studied for its chemical stability, non‑toxicity, and low cost. However, its wide band gap (3.2 eV) limits visible‑light absorption, curbing its practical efficiency.
In contrast, semiconductors such as Ag3PO4, Bi2MoO6, WO3, and g‑C3N4 exhibit strong visible‑light activity. Ag3PO4, in particular, delivers high photo‑oxidation performance but suffers from poor photostability due to Ag+ reduction to metallic Ag and high material cost. Numerous composite strategies (e.g., TiO2/Ag3PO4, Ag3PO4/graphene) have been pursued to mitigate these drawbacks.
Attapulgite (ATP) is a rod‑shaped, hydrated magnesium–aluminum silicate featuring a high specific surface area and excellent adsorption capability. Its rod morphology makes it an attractive scaffold for anchoring photocatalytic nanoparticles, yet ternary ATP‑based composites remain underexplored.
In this study, we synthesized an ATP/TiO2/Ag3PO4 ternary nanocomposite via a simple two‑step route. Comprehensive characterization revealed a robust heterojunction that not only improves charge separation but also stabilizes Ag3PO4 and reduces silver usage, while achieving rapid RhB degradation under simulated solar light.
Experimental section
Materials
ATP nanofibers (average diameter <100 nm, length <1 µm) were supplied by Jiangsu Qingtao Energy Science and Technology Co., Ltd. Rhodamine B (RhB, A.R.), EDTA disodium salt, tert‑butanol, stearyl trimethyl ammonium chloride, silver nitrate, and disodium dihydrogen phosphate dihydrate were obtained from Macklin. TiO2 anatase nanoparticles (5–10 nm, 99.8 % purity) were sourced from Aladdin.
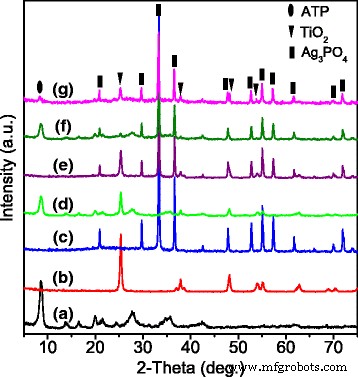
XRD patterns: a ATP, b TiO2, c Ag3PO4, d ATP/TiO2, e Ag3PO4/TiO2, f ATP/Ag3PO4, g ATP/TiO2/Ag3PO4
Synthesis of samples
The ternary composite was fabricated in two steps. First, ATP nanorods and TiO2 nanoparticles (mass ratio 5:2) were dispersed in deionized water and stirred for 4 h, allowing TiO2 to adsorb onto ATP surfaces. After centrifugation, washing, and drying at 60 °C for 6 h, ATP/TiO2 was obtained. Next, Ag3PO4 nanoparticles were precipitated onto ATP/TiO2 via a simple co‑precipitation: 20 mL 0.1 M AgNO3 was mixed with 0.7 g ATP/TiO2 in 50 mL water under sonication for 30 min; then 20 mL 0.1 M Na2HPO4 was added dropwise in the dark for 40 min. The resulting light‑yellow precipitate was centrifuged, washed with absolute ethanol, and dried at 60 °C for 12 h, yielding ATP/TiO2/Ag3PO4. Analogous procedures produced Ag3PO4, Ag3PO4/ATP, Ag3PO4/TiO2, and ATP/TiO2 samples.
Characterization
XRD data were collected on a Rigaku D/max‑RB diffractometer (40 kV, 30 mA). SEM imaging was performed on an INSPECTF FEI instrument. UV‑vis diffuse reflectance spectra were recorded on a Hitachi U‑3010 spectrophotometer with BaSO4 as reference.
Photocatalytic experiment
Photocatalytic degradation of RhB (5 mg L−1) was carried out under simulated solar irradiation using a 300 W Xe lamp (150 mW cm−2). 50 mg of ATP/TiO2/Ag3PO4 was added to 100 mL RhB solution and stirred in the dark for 40 min to reach adsorption–desorption equilibrium. Upon lamp illumination, aliquots (4 mL) were withdrawn at set intervals, centrifuged (10,000 rpm, 10 min), and analyzed by UV‑vis spectroscopy (λmax=554 nm). Degradation efficiency was calculated as %D = (1–A/A0) × 100 %.
Results and discussion
Characterization of the ATP‑Ag3PO4‑TiO2 composites
Figure 1 confirms the crystalline integrity of each component: ATP matches JCPDS #21–0958 (monoclinic), TiO2 shows anatase peaks, and Ag3PO4 aligns with JCPDS #06‑0505. No impurity phases appear in the composites, indicating successful fabrication. In the ternary pattern (Fig. 1g), the ATP peaks are weak, suggesting TiO2 and Ag3PO4 nanoparticles coat the ATP rods.
SEM images (Fig. 2) reveal ATP as sub‑micron rods (<1 µm) with <100 nm diameter. TiO2 nanoparticles (~40 nm) attach uniformly to ATP surfaces, forming ATP/TiO2. In the ternary composite, the rods are completely covered by both TiO2 and ~50 nm Ag3PO4 spheres, producing a lath‑particle architecture that maximizes interfacial contact.

SEM images of a ATP, b ATP/TiO2, and c ATP/TiO2/Ag3PO4 powders
Absorption spectra
UV‑vis spectra (Fig. 3a) show Ag3PO4 absorbs up to ~500 nm, TiO2 absorbs only in the UV, and ATP has weak absorption. The ternary composite combines these features, yielding strong UV absorption from TiO2 and ATP and extended visible‑light response from Ag3PO4. Band‑gap energies were extracted from Tauc plots: TiO2 3.20 eV (indirect), Ag3PO4 2.49 eV (direct), ATP 3.75 eV (direct). The composite exhibits two absorption edges—385 nm (Eg 3.64 eV) and 510 nm (Eg 2.49 eV)—demonstrating retention of both TiO2 and Ag3PO4 characteristics.
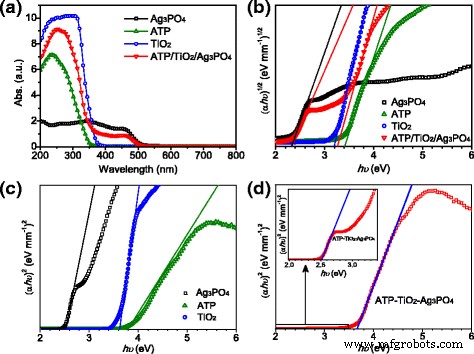
a UV‑vis absorption spectra and b plots of (αhν)1/2 vs. (hν) for Ag3PO4, ATP, TiO2 and ATP/TiO2/Ag3PO4; c plots of (αhν)2 vs. (hν) for Ag3PO4, ATP, TiO2; d plots of (αhν)2 vs. (hν) for ATP/TiO2/Ag3PO4 and the inset shows a magnified view of (d).
Photocatalytic activities
Figure 4a displays the time‑dependent UV‑vis spectra of RhB during irradiation with ATP/TiO2/Ag3PO4. The 554 nm peak nearly vanishes after 20 min, indicating >90 % degradation. In comparison (Fig. 4b), pure TiO2 and ATP achieve <50 % removal after 60 min, whereas Ag3PO4 performs better but suffers from stability loss. The ternary composite reaches 81.1 % degradation after only 3 min and >99 % after 20 min—outperforming all binary counterparts while requiring less Ag content.
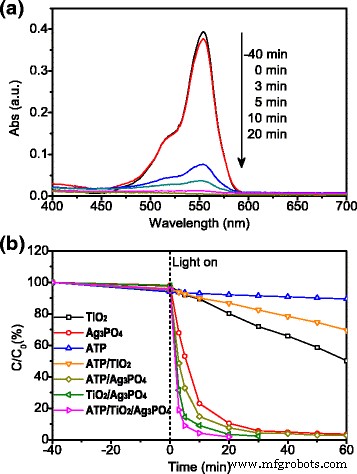
a UV‑vis spectra of RhB degradation by ATP/TiO2/Ag3PO4 over time; b comparative degradation of RhB with various photocatalysts under simulated solar light.
Stability tests (Fig. 5) show that Ag3PO4 loses activity after successive cycles due to Ag accumulation on its surface. In contrast, ATP/TiO2/Ag3PO4 retains >95 % of its initial performance over five cycles, confirming the protective effect of TiO2 and ATP in the heterojunction.
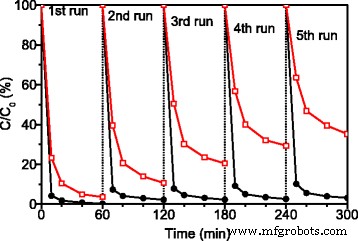
Repeated RhB degradation with Ag3PO4 (red open squares) vs. ATP/TiO2/Ag3PO4 (black solid circles) under simulated solar irradiation.
Possible mechanism in photocatalytic process
Scavenger experiments (Fig. 6) identified the dominant reactive species: holes (h+) and superoxide radicals (O2•–) are essential, while hydroxyl radicals (•OH) play a minor role. The proposed mechanism (Fig. 7) involves electron transfer from TiO2 CB (−0.5 eV) to Ag3PO4 CB (+0.45 eV), and hole migration from Ag3PO4 VB (+2.97 eV) to TiO2 VB (+2.70 eV). This charge separation reduces recombination, enhances O2 reduction to O2•–, and protects Ag+ from photoreduction, thereby improving both activity and stability.
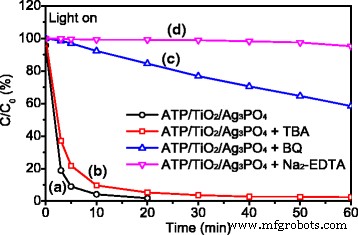
Reactive species trapping experiments for ATP/TiO2/Ag3PO4.
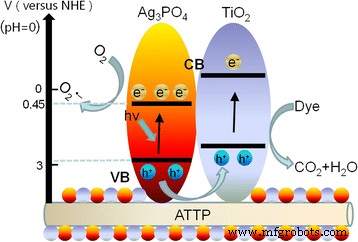
Proposed photocatalytic mechanism of ATP/TiO2/Ag3PO4 composites.
Conclusions
We successfully fabricated an ATP/TiO2/Ag3PO4 ternary nanocomposite via a straightforward two‑step process. The resulting heterojunction markedly improves charge separation, leading to rapid and stable RhB degradation under simulated solar light. Compared with pure Ag3PO4, the composite requires less silver, reduces cost, and offers superior durability. These findings provide a scalable strategy for designing Ag‑based photocatalysts with enhanced performance and longevity.
Abbreviations
- ATP
Attapulgite
- BQ
Benzoquinone
- CB
Conduction band
- Na2-EDTA
Disodium ethylenediaminetetraacetate
- RhB
Rhodamine B
- TBA
Tert‑butanol
- VB
Valence band
Nanomaterials
- Nano‑Heterojunctions: Boosting Solar Cell Efficiency with Colloidal Quantum Dots
- Rapid‑Breakdown Anodized Titania Nanotubes Deliver Superior Sunlight‑Driven Photocatalytic Decolorization of Organic Dyes
- Ag3PO4/BiFeO3 Heterojunctions: Superior Visible‑Light Photocatalytic Degradation of Acid Orange 7
- Enhanced Visible‑Light Photocatalysis via In‑Situ Z‑Scheme BiOBr0.3I0.7/Ag/AgI Nanocomposites
- Optimizing ZnO‑Based Nanohybrids: How Materials, Heterojunctions, and Crystal Orientation Enhance Methyl Orange Degradation
- Enhanced Visible‑Light Photocatalysis via Cu2−xSe‑Modified Monoclinic BiVO4: 15.8‑Fold Activity Boost
- Partially BiVO4-Modified ZnO Porous Nanosheets: Solar‑Driven Photocatalysis with Superior Charge Separation
- Enhanced Visible‑Light Photocatalysis via Z‑Scheme Ag3PO4/TiO2 Heterojunctions
- Visible‑Light‑Driven Solar Hydrogen Production with Cost‑Effective Stannic Oxide
- CQD‑Decorated BiFeO3 Nanoparticles Deliver Superior Visible and Near‑Infrared Photocatalysis for AO7 Degradation and Cr(VI) Reduction



