Rapid‑Breakdown Anodized Titania Nanotubes Deliver Superior Sunlight‑Driven Photocatalytic Decolorization of Organic Dyes
Abstract
Rapid‑breakdown anodization (RBA) in perchloric acid yields highly crystalline titania nanotube (TNT) powder. Annealing between 250 °C and 550 °C modifies the nanotube morphology, phase composition and band‑gap energy, while retaining an open‑ended tubular structure only up to 250 °C. The as‑prepared TNTs exhibit a 3.04 eV band‑gap, which narrows to 2.88 eV after high‑temperature annealing. X‑ray photoelectron spectroscopy (XPS) confirms TiO₂ surface chemistry with residual chlorine that diminishes upon heating. Photoluminescence (PL) indicates reduced electron‑hole recombination in samples annealed at 350 °C and 450 °C, with the 450 °C powder delivering the highest photocurrent. When used as photocatalysts, the TNTs demonstrate modest dye decolorization under UV light but achieve complete removal of both anionic methyl orange (MO) and cationic rhodamine B (RhB) under natural sunlight. The superior sunlight performance stems from the narrow band‑gap, high specific surface area, and mixed anatase/brookite crystal structure, outperforming commercial anatase TiO₂. These RBA‑derived TNTs therefore represent a promising, scalable platform for solar‑driven water remediation.
Background
One‑dimensional titania nanotubes (TNTs) are prized for their high specific surface area, tunable crystal phases, and exceptional optical properties, making them ideal for gas sensing, dye‑sensitized solar cells, electrochromic devices, and photocatalysis [1–4]. While TNTs can be synthesized via anodization, hydrothermal, sol‑gel, or template methods, the time‑consuming anodization of titanium foil followed by mechanical peeling has limited large‑scale production [5–8]. Rapid‑breakdown anodization (RBA) circumvents this bottleneck by converting Ti metal into TiO₂ nanotubes within seconds in a halide‑rich electrolyte [7,9]. The process generates a thin oxide layer that is instantaneously etched by chloride ions, producing localized pits that expand into nanotube bundles. After brief growth, the bundles detach and can be collected as powder, offering a scalable route to TNTs [10–12]. RBA‑derived TNT powders have been explored for dye‑sensitized solar cells, hydrogen evolution, and photocatalytic pollutant degradation [12–19]. However, their performance under visible light, especially natural sunlight, remains underreported.
Titania’s suitability for water purification stems from its non‑toxicity, thermal stability, and cost‑effectiveness [25]. TNTs, with their tubular geometry and large surface area, can accelerate photocatalytic degradation of organic dyes (e.g., MO, RhB) [25–27]. The band‑gap of TNTs (~3.0–3.2 eV) depends on the crystal phase (anatase, brookite, rutile) and can be tuned by annealing, which simultaneously alters morphology and specific surface area [3,11]. Anatase offers an indirect band‑gap and longer electron‑hole lifetimes than rutile or brookite, while brookite’s presence can improve charge separation when mixed with anatase [28–30]. Most studies report efficient UV‑driven degradation, but TNTs are generally inactive under visible light [29]. Modifying the electronic structure via anionic or cationic doping has been employed to shift activity into the visible spectrum [30–32].
In this work, we synthesize undoped TNT powders by RBA, anneal them at 250 °C–550 °C, and evaluate their photocatalytic activity for MO and RhB decolorization under UV and natural sunlight. To our knowledge, this is the first comprehensive study of RBA‑derived TNT powders under real‑world solar irradiation, revealing that they can achieve complete dye removal while outperforming commercial anatase TiO₂.
Methods/Experimental
Preparation of the TNTs
TNT powder was synthesized by RBA in 0.1 M HClO₄ (Sigma‑Aldrich, 70 %) with a 20 V potential, following the protocol of Zhong et al. [11]. The as‑prepared powders were then annealed in air for 3 h at 250 °C, 350 °C, 450 °C, or 550 °C to investigate the effects of temperature on structure and photocatalytic performance.
Characterization methods
Transmission electron microscopy (TEM; Tecnai F‑20G2 FEG S‑Twin GIF, 200 kV) revealed nanotube dimensions and morphology. X‑ray diffraction (XRD; PANalytical X’pert Pro, Co‑Kα, 20°–110° 2θ) determined crystalline phases. Raman spectroscopy (Horiba Jobin‑Yvon Labram HR, 514 nm, 50 mW) provided complementary phase information. Ultraviolet–visible diffuse reflectance spectroscopy (DRS; Agilent Cary 5000, 200–800 nm) was used to calculate band‑gap energies via the Kubelka–Munk transform. Fourier transform infrared spectroscopy (FTIR; Nicolet 380 ATR, 525–4000 cm⁻¹) assessed surface functional groups. X‑ray photoelectron spectroscopy (XPS; Kratos AXIS Ultra, Al Kα, 1486.6 eV) quantified surface elemental composition and chemical states. Photoluminescence (PL; Perkin Elmer LS 50B, 330 nm excitation) probed charge carrier recombination. Photocurrent and incident photon‑to‑current efficiency (IPCE) measurements were performed on TNT films deposited on fluorine‑doped tin oxide (FTO) glass, using a three‑electrode setup (Ag/AgCl reference, Pt counter) under 150 W Xe lamp irradiation (300–400 nm). Electrochemical impedance spectroscopy (EIS; Gamry Reference 600+, 200 kHz–100 mHz, 15 mV AC) evaluated electron transfer kinetics.
Photocatalysis of TNTs
Decolorization experiments were carried out with 10 mg L⁻¹ aqueous solutions of methyl orange (MO) or rhodamine B (RhB). Each suspension contained 100 mg TNT powder in 100 mL dye solution. After a 3‑h adsorption period in the dark, the mixtures were irradiated either with two 11 W Philips PL‑S UV lamps (350–400 nm, 1 mW cm⁻²) or under natural sunlight (Espoo, Finland, 60° N, 24° E, midday June–July 2017). Control experiments without catalyst assessed photolysis. At defined intervals, the catalyst was removed by 0.45 µm filtration and centrifugation; dye concentrations were measured spectrophotometrically (MO: λ_max = 465 nm; RhB: λ_max = 554 nm). Pseudo‑first‑order rate constants (κ₁) were derived from ln(C/C₀) = –κ₁t plots.
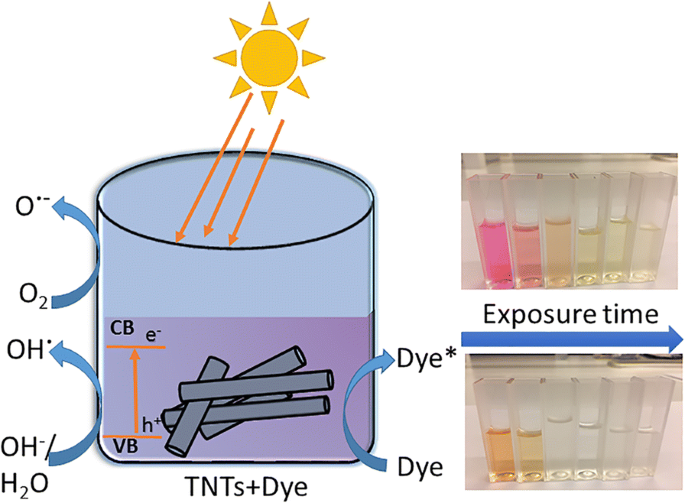
Schematic illustration of decolorization of organic dyes under natural solar light irradiation by undoped TNTs
Results and discussion
Morphology and structure
RBA generates single‑walled TNT bundles with open tops, closed bottoms, 11–18 nm pores, and 18–35 µm lengths in seconds. Annealing preserves this morphology only up to 250 °C; at 350 °C and above, nanotubes collapse into nanorods and nanoparticles, reducing the specific surface area from 179 m² g⁻¹ to 35 m² g⁻¹ (Table 2). XRD and Raman confirm anatase dominance at 250 °C, 350 °C, and 450 °C, with brookite present at 450 °C and rutile emerging at 550 °C. Empirical phase fractions derived from peak intensities reveal 64 % anatase/36 % brookite at 450 °C and 34 % anatase/66 % rutile at 550 °C.

TEM images of TNTs. a As‑prepared TNTs (open top, closed bottom); b 250 °C; c 350 °C (nanorod transition); d 450 °C
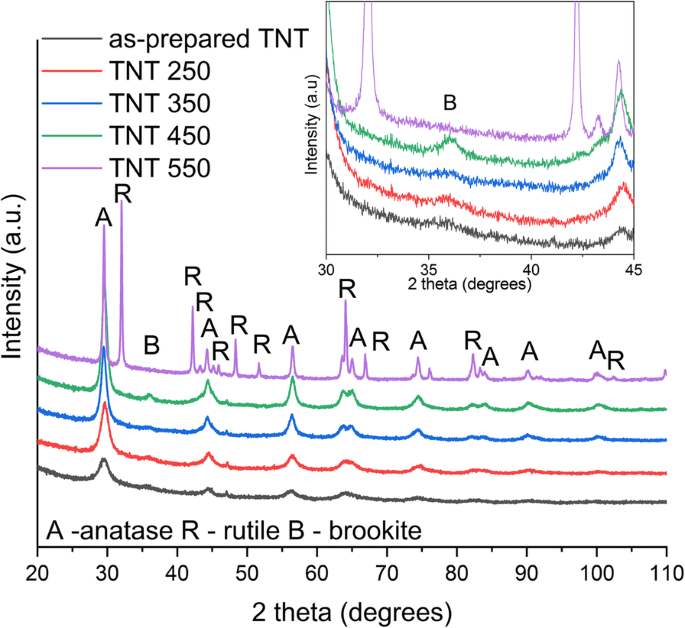
XRD of as‑prepared and annealed TNTs (Co‑Kα)
Band‑gap energies derived from Kubelka–Munk plots are 3.04 eV (as‑prepared and up to 350 °C) and 3.14 eV/2.88 eV for 450 °C/550 °C, respectively (Fig. 4). The redshift correlates with the emergence of rutile at 550 °C. FTIR spectra show O–H bending (1620–1640 cm⁻¹) and stretching (3000–3500 cm⁻¹) bands, diminishing with temperature, indicating loss of surface hydroxyls (Fig. 5). XPS confirms Ti⁴⁺/O bonding and a high‑energy Cl 2p doublet (198.6/200.1 eV) that diminishes from 2.3 % to 0.3 % atomic % after annealing (Fig. 6). The Ti/O ratio remains constant across temperatures. PL spectra exhibit a dominant band at 419 nm (2.96 eV); intensity decreases from as‑prepared to 350 °C, with the lowest recombination in the 550 °C sample, consistent with increased charge separation (Fig. 7). IPCE measurements reveal the 450 °C powder (anatase/brookite) achieving the highest photocurrent and IPCE (1.37× higher than 350 °C, 3.95× higher than 550 °C) (Fig. 8), highlighting the beneficial mixed‑phase effect.
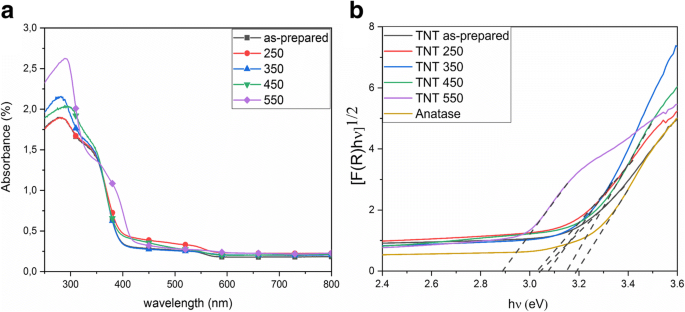
UV/Vis diffuse reflectance: a absorbance; b Kubelka–Munk function (band‑gap estimation)
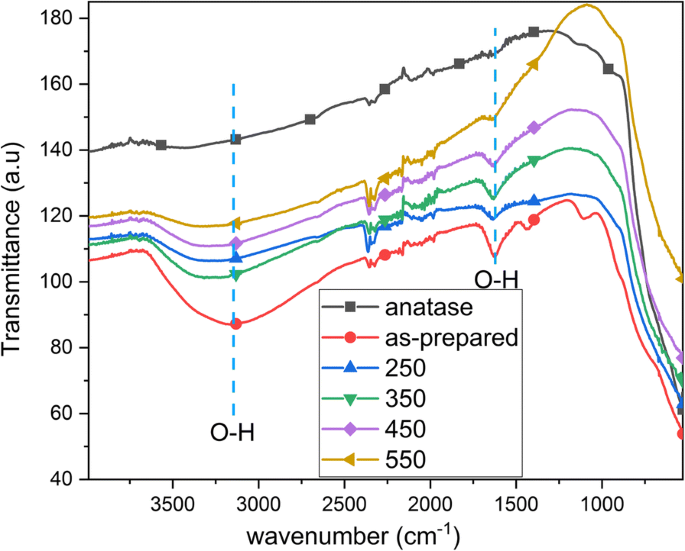
FTIR of as‑prepared TNTs, annealed TNTs (250, 350, 450, 550 °C), and reference anatase powder
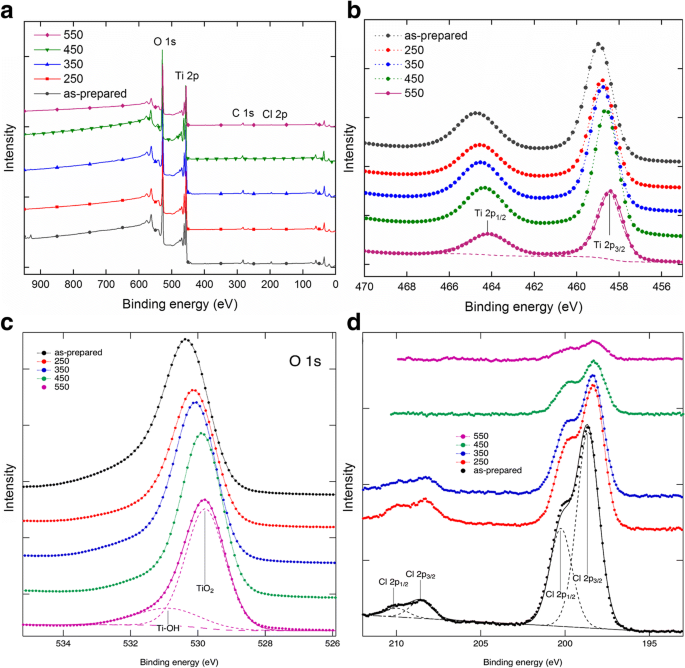
XPS: a survey; b Ti 2p; c O 1s; d Cl 2p
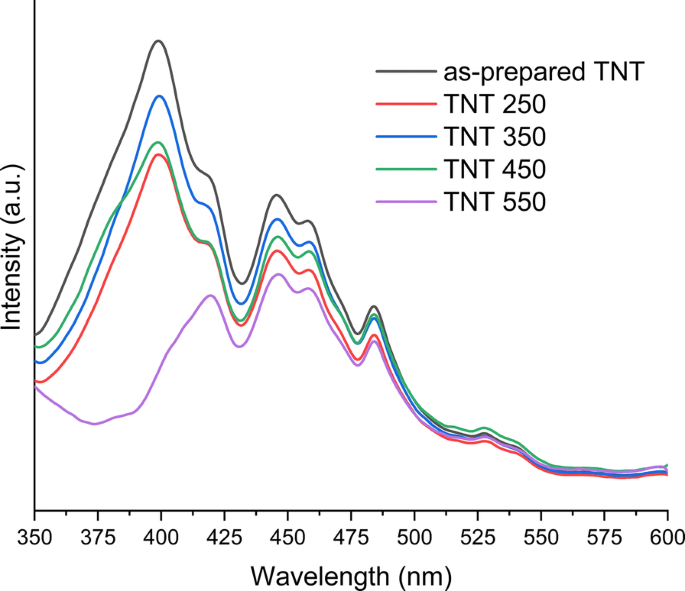
Photoluminescence emission of as‑prepared and annealed TNTs
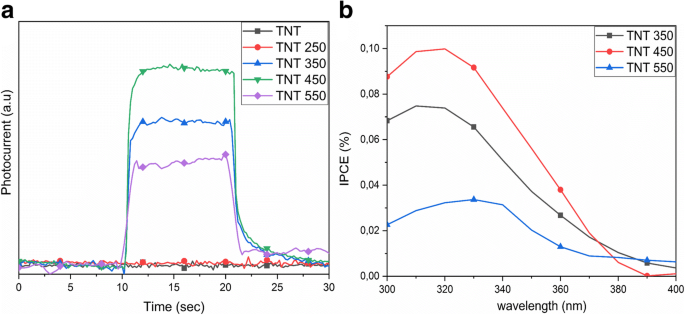
a Photocurrent at 320 nm; b IPCE of TNT 350, 450, 550
Photocatalytic decolorization of TNTs
Adsorption tests show the as‑prepared TNTs adsorb ~28 % of MO within 3 h, whereas all other samples adsorb ≤15 % of both dyes (Fig. 9). Under UV light, the anatase reference decolorizes both MO and RhB within 3 h. The best UV performance among TNTs is TNT 350 (47 % MO, 38 % RhB). In contrast, under natural sunlight the as‑prepared TNT, TNT 250, and reference anatase achieve complete MO decolorization (≤1 mg L⁻¹), while TNT 450 reaches 87 % MO after 2 h. RhB decolorization is fastest with the as‑prepared TNT (κ₁ = 1.29 h⁻¹, R² = 0.93), surpassing the anatase reference (1.22 h⁻¹, R² = 0.89). Overall, TNTs outperform their reference under sunlight, contradicting the common view that TiO₂ is ineffective in the visible spectrum.
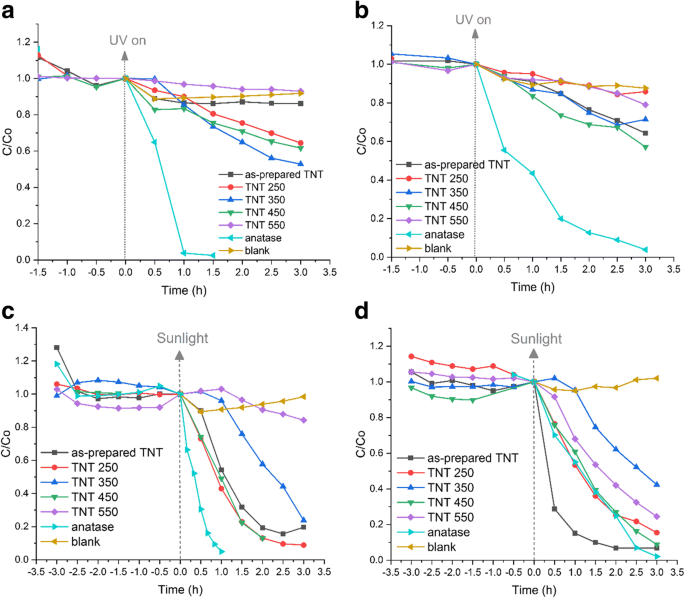
Decolorization of a MO and b RhB under UV light; c MO, d RhB under sunlight (3 h)
Pseudo‑first‑order kinetics (Table 2) reveal the anatase reference as the fastest UV catalyst (MO: 2.78 h⁻¹; RhB: 1.05 h⁻¹). TNT 350 achieves 0.24 h⁻¹ (MO) and TNT 450 0.18 h⁻¹ (RhB) under UV. In sunlight, TNT 450 delivers 1.05 h⁻¹ (MO) and the as‑prepared TNT 1.29 h⁻¹ (RhB). The superior sunlight activity of the as‑prepared TNT is attributed to its high surface area, abundant –OH groups, and unique degradation pathway (N‑de‑ethylation of RhB to Rhodamine, observed only with this sample). The mixed‑phase TNT 450 shows enhanced electron‑hole separation, as evidenced by its higher IPCE, compensating for its reduced surface area.
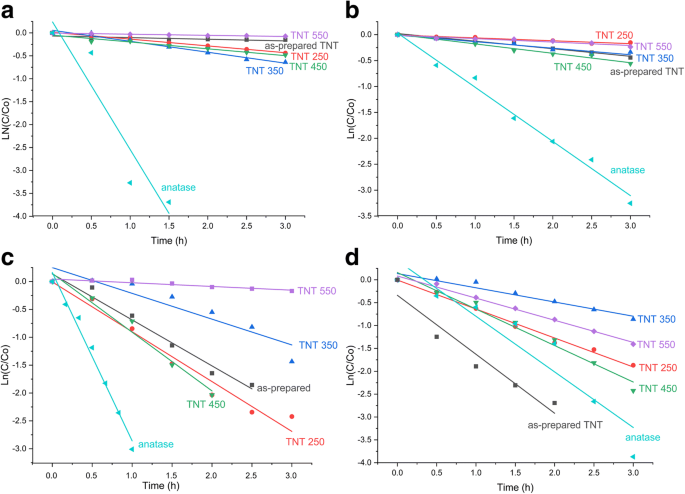
Pseudo‑first‑order plots: a MO, b RhB (UV); c MO, d RhB (sunlight)
Comparative analysis shows that the as‑prepared TNT (anatase, 179 m² g⁻¹) and TNT 450 (anatase/brookite, 70 m² g⁻¹) differ in surface area, chlorine impurity, and electron‑hole lifetime. The as‑prepared TNT’s high –OH content promotes N‑de‑ethylation under visible light, while the mixed‑phase TNT 450’s favorable band alignment facilitates charge separation. UV‑Vis spectra of RhB degradation (Fig. 11) confirm that sunlight induces a 498 nm shift, indicating Rhodamine formation, whereas UV irradiation only reduces absorbance. MO degradation shows only a decrease in absorbance, consistent with azo group cleavage.
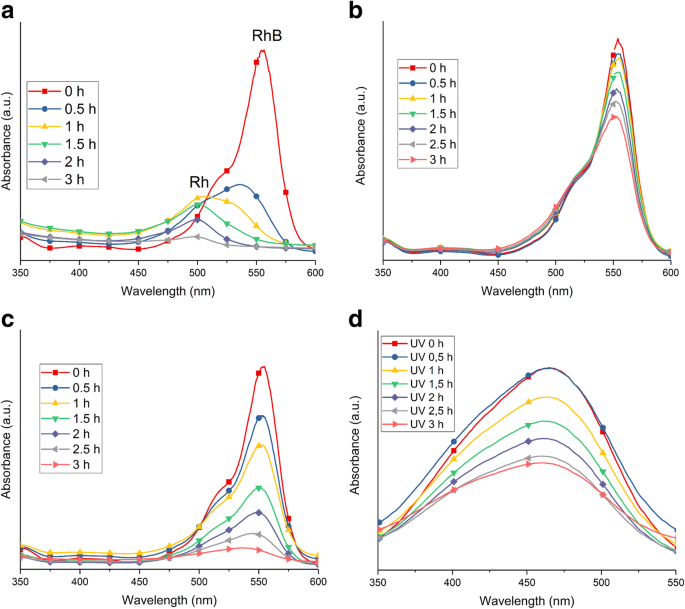
UV‑Vis spectra of RhB and MO decolorization: a as‑prepared TNT (sunlight); b as‑prepared TNT (UV); c TNT 450 (sunlight); d TNT 350 (MO)
Conclusions
Rapid‑breakdown anodization yields scalable TNT powders whose properties can be finely tuned by annealing. Band‑gap narrowing from 3.04 eV to 2.88 eV and phase evolution (anatase → anatase/brookite → anatase/rutile) accompany a transition from tubular to nanorod morphology and a 75 % drop in specific surface area. XPS confirms a reduction in chlorine contamination with temperature, while PL and IPCE data highlight reduced electron‑hole recombination in mixed‑phase samples, particularly TNT 450. Photocatalytic tests demonstrate that all TNT powders outperform commercial anatase TiO₂ under natural sunlight, achieving complete decolorization of MO and superior RhB degradation with the as‑prepared TNT. The findings reveal that RBA‑derived TNTs can harness the full solar spectrum, offering a practical and efficient route for photocatalytic water remediation.
Nanomaterials
- Enhanced Photocatalytic Degradation of Rhodamine B Using an ATP/TiO₂/Ag₃PO₄ Ternary Nanocomposite Under Simulated Solar Light
- Impact of Morphology and Crystal Structure on Titania Nanotube Thermal Conductivity
- Designing Plasmonic Nanoparticle Strategies for Enhanced Organic Solar Cell Performance
- Optimizing ZnO‑Based Nanohybrids: How Materials, Heterojunctions, and Crystal Orientation Enhance Methyl Orange Degradation
- Enhanced Visible‑Light Photocatalysis via Cu2−xSe‑Modified Monoclinic BiVO4: 15.8‑Fold Activity Boost
- Partially BiVO4-Modified ZnO Porous Nanosheets: Solar‑Driven Photocatalysis with Superior Charge Separation
- High‑Performance Flexible Photocatalytic Paper: Cu2O and Ag Nanoparticle‑Decorated ZnO Nanorods for Visible‑Light Degradation of Organic Dyes
- CQD‑Decorated BiFeO3 Nanoparticles Deliver Superior Visible and Near‑Infrared Photocatalysis for AO7 Degradation and Cr(VI) Reduction
- GaAs Nanoantenna Solar Absorber: Ultra‑Broadband Light Trapping for High‑Efficiency Thin Cells
- Self‑assembled SnIn4S8/TiO₂ Nanotube Photoanode for Enhanced Visible‑Light Photocathodic Protection of Q235 Steel



