Fe3O4@C Core–Shell Nanoparticles: Hydrothermal Synthesis and Magnetic Removal of Heavy Metals from Water
Abstract
Core–shell Fe3O4@C nanoparticles were synthesized via a simple hydrothermal route followed by calcination at 450 °C. The resulting material displays a highly porous structure with a Brunauer–Emmett–Teller (BET) surface area of 238.18 m2 g−1, far exceeding typical Fe3O4 powders. When applied to aqueous solutions containing Pb2+, Cd2+, Cu2+ and Cr6+ at pH 3, the adsorbent achieved removal efficiencies of 100 %, 99.2 %, 96.6 % and 94.8 %, respectively. The magnetic core enables rapid and effortless separation of the particles from water, making this material a practical candidate for large‑scale water treatment. Moreover, the scalable synthesis approach offers a cost‑effective pathway to produce multifunctional core–shell nanostructures for environmental remediation, catalysis and energy applications.
Background
Heavy‑metal contamination poses serious risks to human health and ecosystems, prompting stringent regulations on industrial effluents. Conventional removal methods—ion exchange, coagulation, precipitation—often suffer from high operational costs, sludge generation, and incomplete metal removal. Nanostructured adsorbents with controlled morphology, such as hollow spheres, nanowires and nanotubes, have shown superior performance due to their high surface area and tunable chemistry. Core–shell architectures, in particular, combine the functional advantages of a magnetic core with a reactive shell, offering both high adsorption capacity and magnetic recoverability. Despite promising reports of Fe3O4–based core–shell composites, scalable, environmentally friendly synthesis routes remain limited. This study addresses that gap by presenting a straightforward hydrothermal procedure that yields Fe3O4@C hybrids with exceptional adsorption properties.
Experimental
Materials and Synthesis
Synthesis of Core–Shell Fe3O4 Hybrid Nanoparticle Aggregates
In a typical procedure, 0.72 g of Fe(NO3)3·9H2O, 0.0086 g of NH4H2PO4, 0.008 g of Na2SO4·10H2O and 3 g of glucose were dissolved in 90 mL of distilled water. After stirring for 10 min, the mixture was transferred to a 100 mL Teflon‑lined stainless‑steel autoclave and heated at 180 °C for 48 h. The resulting black precipitate was washed with deionized water and ethanol, dried at 65 °C overnight, and calcined at 450 °C (3 °C min−1) under a 4‑h CO/Ar flow. The calcination converts the Fe3+ precursor into Fe3O4 cores while carbonizing the organic matrix to form the shell, yielding Fe3O4@C core–shell aggregates (Scheme 1).

Synthesis route of the core–shell Fe3O4@C hybrid nanoparticle
Characterization
Phase composition was examined by X‑ray diffraction (Rigaku D/max‑A, Co Kα). Functional groups were identified via Fourier‑transform infrared spectroscopy (FTIR, Thermo Nicolet AVATARFTIR 360). Morphology and microstructure were visualized using SEM (AMRAY 1000B) and HR‑TEM (JEOL JEM‑2100, 200 kV). Nitrogen adsorption–desorption measurements (Micromeritics Tristar) provided BET surface areas and pore size distributions. Magnetic properties were measured with a vibrating‑sample magnetometer (VSM). Heavy‑metal adsorption experiments were conducted at room temperature: solutions containing 10 mg L−1 Pb2+, Cd2+, Cu2+ or Cr6+ were adjusted to pH 3, stirred with 20 mg of Fe3O4@C, and aliquots were collected at specified intervals for atomic absorption spectroscopy (AAS, Hitachi Z2000).
Heavy Metal Ion Removal Experiments
Adsorption tests were performed in 50 mL volumes with 20 mg of Fe3O4@C. Samples were sampled at 0, 0.5, 1, 1.5, 2, 4, 6, 10 and 24 h, filtered, and analyzed by AAS to determine residual metal concentrations.
Result and Discussion
Physicochemical Characteristics of Core–Shell Fe3O4@C Nanospheres
X‑ray diffraction patterns (Fig. 1) confirm the formation of crystalline Fe3O4 (fcc) and graphitic carbon (002). SEM images (Fig. 2) reveal uniform microspheres (~700 nm) with a distinct core–shell morphology. HRTEM (Fig. 2e) shows lattice spacing of 0.297 nm, matching the (220) plane of Fe3O4, and SAED confirms single‑crystal quality. The high BET surface area (238.18 m2 g−1) and mesoporous pore distribution (7.5–9.1 nm) (Fig. 4) are attributed to the carbon shell and calcination process.
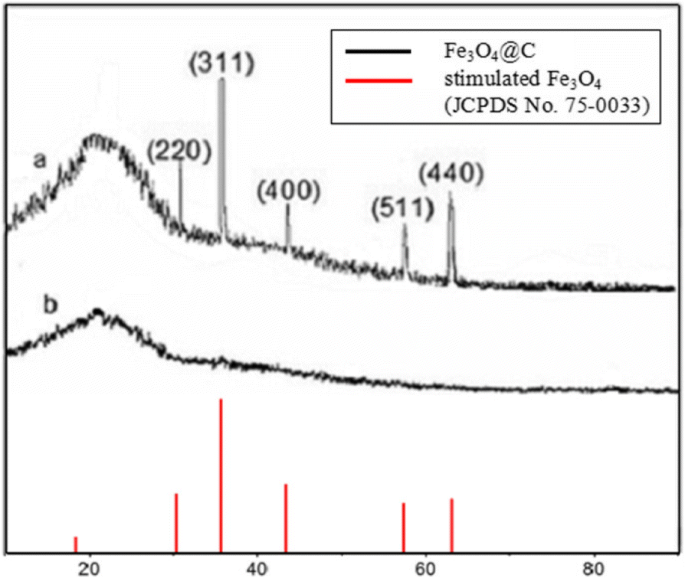
XRD patterns of core‑shell Fe3O4@C hybrid nanoparticle aggregates and its precursor (a—Fe3O4@C; b—precursor)

SEM image of prepared precursor (a). SEM images (b, c), TEM image (d), HRTEM micrograph (e), and SAED (f) of as‑synthesized core‑shell Fe3O4@C aggregates (calcined at 450 °C)
VSM measurements (Fig. 3) show a saturation magnetization of 53 emu g−1, sufficient for magnetic separation yet lower than bare Fe3O4 microspheres due to the carbon shell.
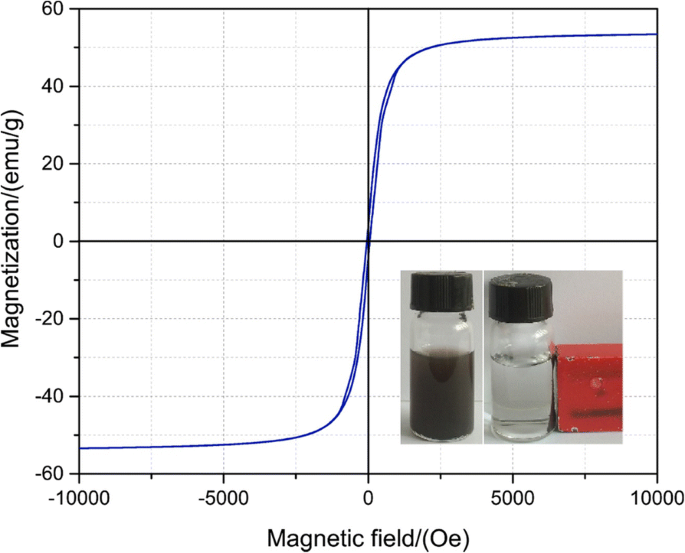
Magnetization loop measurements
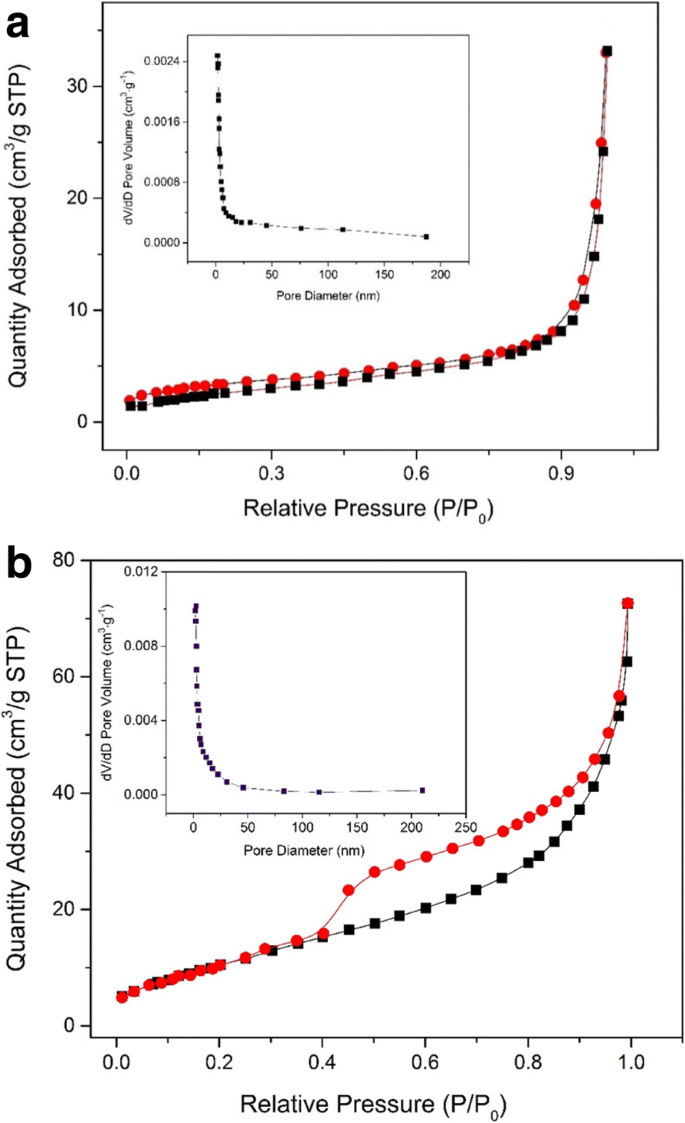
Nitrogen adsorption–desorption isotherm and BJH pore size distribution plot (inset) of the prepared sample before (a) and after (b) calcination
Uptake of Heavy Metal Ions by Fe3O4@C
Removal efficiencies (Fig. 5) reach 100 % for Pb2+, 99.2 % for Cd2+, 96.6 % for Cu2+, and 94.8 % for Cr6+ within 24 h at pH 3. The superior performance is attributed to the high surface area, abundant active sites and the magnetic core enabling rapid recovery.
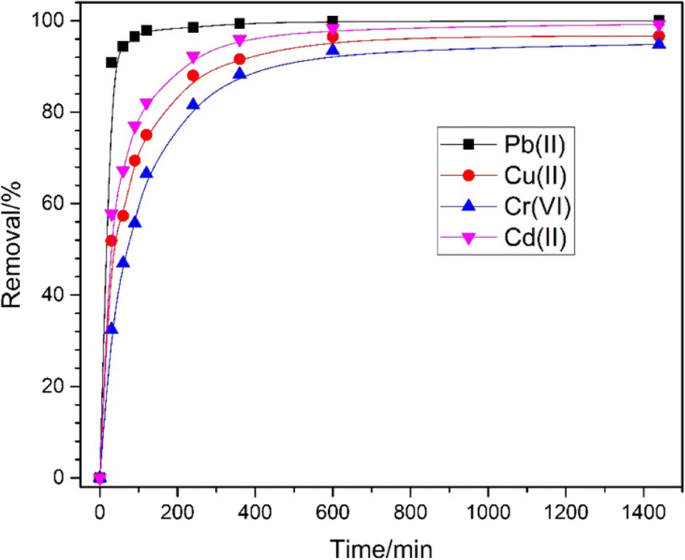
Relationship between the removal efficiency and time for the adsorption of Pb(II), Cd(II), Cu(II) and Cr(VI) by Fe3O4@C (400 mg L−1) at initial concentrations of 10 mg L−1
FTIR Spectra of the Heavy Metal Loaded Fe3O4@C
FTIR analysis (Fig. 6) shows significant shifts in the O–H stretching (~3475 cm−1), C=O (~1605 cm−1) and metal–oxygen bands after Pb2+ adsorption, confirming strong metal–ligand interactions.
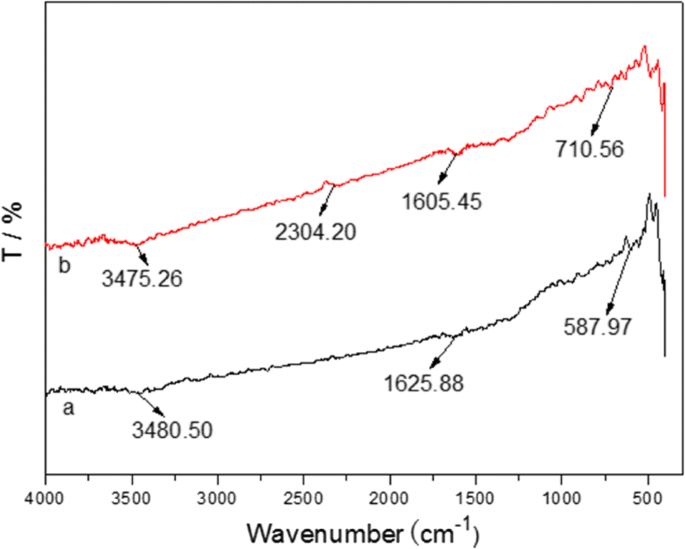
FTIR spectra of the prepared Fe3O4@C sample before (a) and after (b) adsorption of Pb(II)
Adsorption Kinetics
Adsorption follows a pseudo‑second‑order kinetic model (Eq. 1). The linear fit (R2 = 0.999) confirms chemisorption dominates the process. Kinetic parameters are listed in Table 1.
\[\frac{t}{q_t}=\frac{1}{k_2 q_e^2}+\frac{1}{q_e}t\]
Adsorption Isotherm
Equilibrium data were best described by the Freundlich isotherm (R2 = 0.9712), indicating heterogeneous surface adsorption. The Langmuir model did not fit the data adequately.
\[\lg q_e=\lg k_F+\frac{1}{n}\lg C_e\]
Thermodynamics Analysis
Arrhenius analysis yields an activation energy of 34.92 kJ mol−1, characteristic of physisorption. This suggests the process is energetically favorable and reversible.
\[\ln k_2=\frac{A}{n}-\frac{E_a}{RT}\]
Conclusions
Fe3O4@C core–shell nanoparticles were produced via a green hydrothermal method and calcination. The resulting material combines a high surface area, mesoporosity and magnetic recoverability, achieving near‑complete removal of Pb, Cd, Cu and Cr species from water. The scalable, low‑cost synthesis and excellent adsorption performance position Fe3O4@C as a promising candidate for industrial wastewater treatment and other environmental applications.
Nanomaterials
- Magnetite Nano‑Adsorbent from Mill Scale Waste Efficiently Removes Cu(II) from Water: Synthesis, Characterization, Adsorption Kinetics and Regeneration
- Optimizing Heavy Metal Removal with Iron‑Modified Magnetic Biochar Nanocomposites
- Enhanced Photocatalytic Performance of ZnO/In₂O₃ Hybrid Nanostructures via Hydrothermal Synthesis
- Palladium(II)-Imprinted Polymeric Nanospheres for Efficient Removal of Pd(II) from Water
- Optimized Fe³⁺‑Grafted BiOCl for Rapid Adsorption of Trace Cationic and Anionic Dyes in Aqueous Wastewater
- Titania‑Coated Silica Enhanced with Sodium Alginate: A Superior Sorbent for Cu(II), Zn(II), Cd(II), and Pb(II) Removal
- Rapid One‑Pot Synthesis of Monodisperse CoFe₂O₄@Ag Core–Shell Nanoparticles with Exceptional Plasmonic and Magnetic Properties
- Amorphous Nanoparticle Ferrofluids: Magnetoviscous Behavior & Hyperthermia Performance
- Magnetic Assembly of Monodisperse Noble Metal Microspheres for Reproducible, Recyclable SERS Substrates
- Advanced Approaches to Noble Metal Nanoparticle Synthesis



