Visible‑Light‑Driven Solar Hydrogen Production with Cost‑Effective Stannic Oxide
Abstract
Visible‑light‑responsive stannic oxide was produced via a simple one‑pot solvothermal route using SnCl₂·2H₂O and methanol. X‑ray diffraction confirmed a low‑crystalline SnO₂ phase, while UV–vis spectroscopy revealed an absorption edge near 530 nm, indicating strong visible‑light activity. In photoelectrochemical tests on FTO electrodes, the material generated significant anodic photocurrents under λ > 420 nm illumination and produced hydrogen (with methanol as an electron donor) and oxygen (with AgNO₃ as an electron acceptor) without the need for additional co‑catalysts. The enhanced visible‑light performance is attributed to self‑doping of Sn²⁺ into the Sn⁴⁺ lattice, creating intermediate energy levels that narrow the effective band gap.
Introduction
Harnessing solar energy to split water and generate clean hydrogen is a cornerstone of renewable energy strategies. Photocatalytic and photoelectrochemical water splitting offer resource‑sustainable, environmentally benign, and cost‑effective pathways compared to conventional thermochemical processes. The challenge lies in engineering semiconductor photocatalysts with band gaps that absorb a substantial portion of the solar spectrum, band edges aligned for water oxidation and reduction, high quantum yields, and long‑term stability.
While binary oxides such as TiO₂, ZnO, and Fe₂O₃ have been extensively studied, their wide band gaps limit visible‑light utilization. Ternary oxides (e.g., SrTiO₃, NaTaO₃) and complex oxynitride or sulfide systems have shown improved performance, yet many still fall short of simultaneously meeting all criteria. SnO₂, a well‑known n‑type semiconductor with a band gap of 3.6–3.8 eV, typically requires coupling with other materials to enhance visible‑light response.
Here we report a facile one‑pot solvothermal synthesis of SnO₂‑x that demonstrates intrinsic visible‑light photocatalytic activity and photoelectrochemical water‑splitting performance without the need for composite fabrication or co‑catalyst loading.
Methods
Reagents
All reagents were of analytical grade and used as received. Deionized water (18.25 MΩ·cm) served as the solvent for electrode preparation and photocatalytic measurements.
Preparation of SnO₂‑x Powder
SnCl₂·2H₂O (0.02 mol) was dissolved in 100 mL methanol and stirred for 30 min. The solution’s pH was adjusted from 1.0 to 3.0 using 0.02 mol L⁻¹ NH₃·H₂O, yielding a white precipitate. After 2 h of stirring, the mixture was transferred to a 200 mL Teflon‑lined autoclave and heated at 423 K for 20 h. The resulting yellow slurry was washed with water and ethanol, then dried at 343 K for 12 h to obtain the target powder.
Preparation of SnO₂‑x Electrode
Electrophoretic deposition onto fluorine‑doped tin oxide (FTO) glass produced porous thin films (~2 µm thick). The deposition bath contained 40 mg of the synthesized powder and 15 mg iodine in 40 mL acetone, sonicated for 3 min. The coated area was ~1.5 × 4 cm.
Photocatalytic Evaluation
Photoelectrochemical measurements used a three‑electrode setup: the prepared electrode as working, Pt mesh as counter, and Ag/AgCl as reference. The electrolyte was 0.1 M Na₂SO₄ (pH 4.05), purged with Ar for 10 min. Illumination came from a 300 W Xe lamp filtered with a Hoya L‑42 cut‑off filter (λ < 420 nm blocked).
Photocatalytic hydrogen and oxygen evolution were measured in a side‑irradiation Pyrex reactor with a closed gas circulation system. 0.2 g of powder was dispersed in 200 mL of solution, illuminated by the same Xe lamp, and evolved gases were analyzed by TCD gas chromatography using Ar as carrier.
Characterization
X‑ray powder diffraction (Cu Kα), scanning electron microscopy (SEM), transmission electron microscopy (TEM), high‑resolution TEM (HRTEM), Brunauer–Emmett–Teller (BET) surface area analysis, UV–vis diffuse reflectance, and X‑ray photoelectron spectroscopy (XPS) were employed to probe structure, morphology, surface area, optical properties, and elemental states.
Results and Discussion
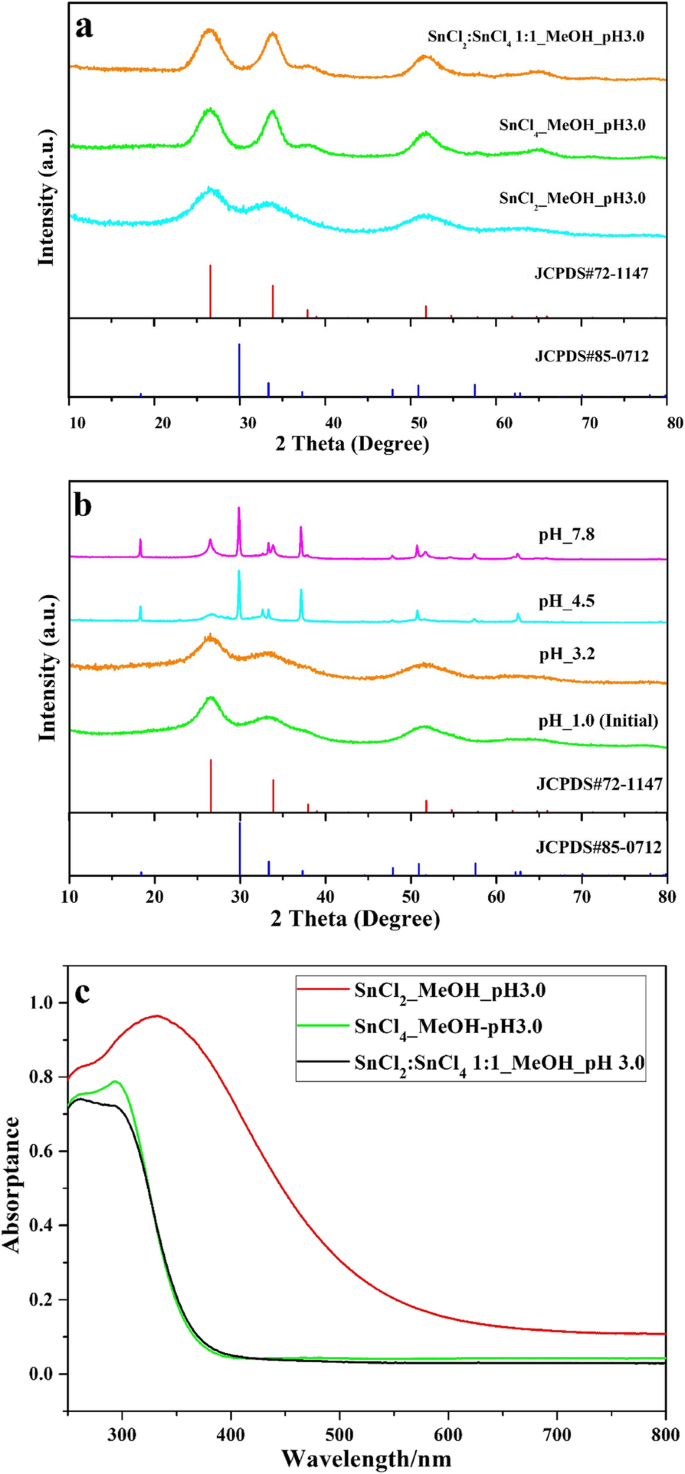
XRD confirmed a low‑crystalline SnO₂ phase (JCPDS #72‑1147). UV–vis spectra showed an absorption edge at ~570 nm, corresponding to a band gap of 2.17 eV, revealing substantial visible‑light absorption.
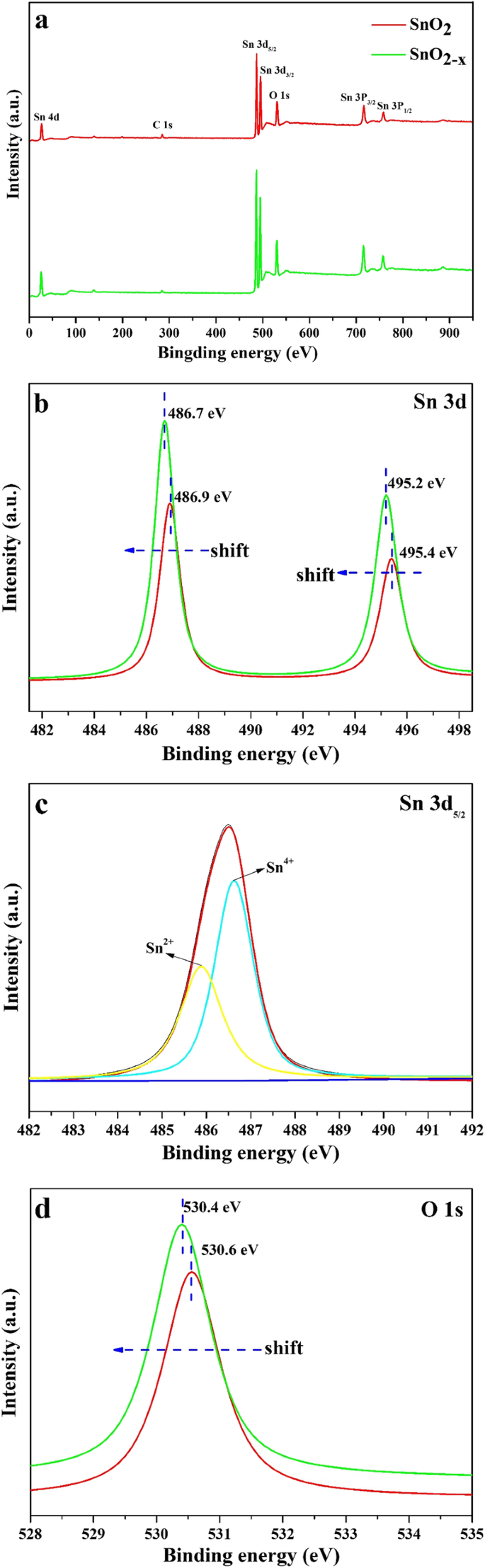
When the precursor was SnCl₂·2H₂O in methanol and the pH was tuned to 3.0, the resulting material displayed the desired SnO₂‑x phase. XPS analysis revealed a slight shift (~0.2 eV) in the Sn 3d binding energies, indicative of Sn²⁺ incorporation into the Sn⁴⁺ lattice. Deconvolution of the Sn 3d₅/₂ peak separated contributions from Sn⁴⁺ (486.8 eV) and Sn²⁺ (485.8 eV), confirming self‑doping and the formation of oxygen vacancies. The O 1s peak also shifted by ~0.2 eV, supporting the presence of vacancies that enhance visible‑light absorption.
SEM and TEM images showed uniform spherical particles (~1–2 µm) with a BET surface area of ~100 m² g⁻¹. HRTEM revealed lattice fringes with 0.33 nm spacing, corresponding to the (110) plane of cassiterite SnO₂.
Photoelectrochemical tests revealed a significant anodic photocurrent under λ > 420 nm illumination, with an onset potential below 0 V versus RHE, indicating that the conduction band lies above the H⁺/H₂ reduction potential and enabling bias‑free water splitting. Hydrogen and oxygen evolution experiments confirmed that the material could split water under visible light without any co‑catalyst; loading 1 wt.% Pt via in‑situ photodeposition further enhanced the rates.
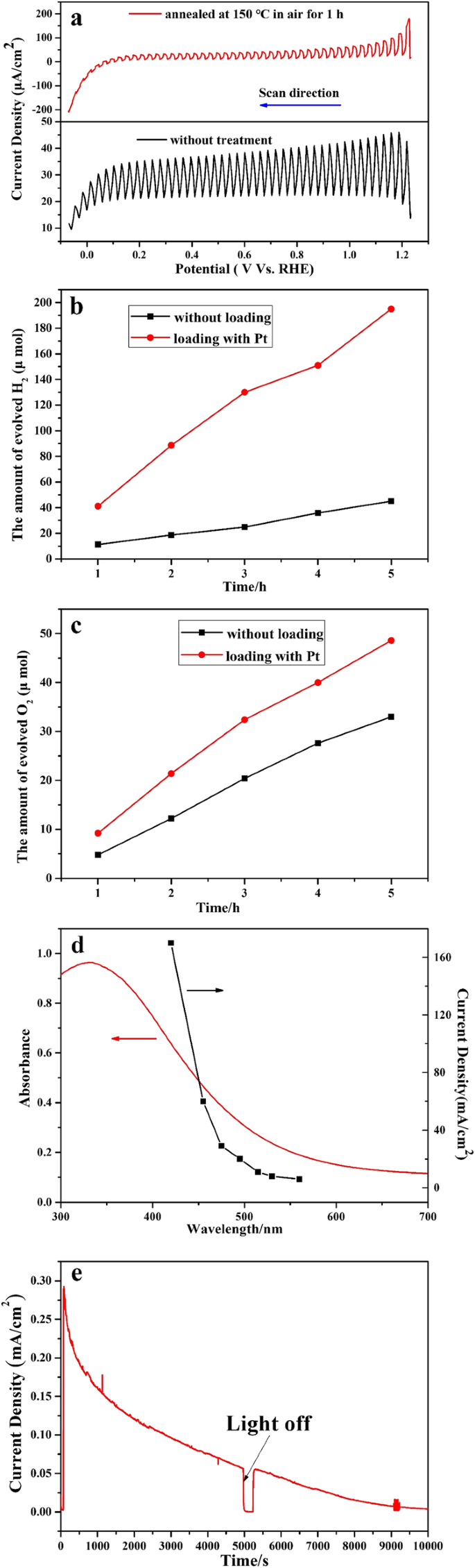
The visible‑light activity is attributed to the creation of intermediate states between the Sn 5s orbital of Sn²⁺ and the O 2p orbitals, effectively narrowing the band gap. However, the photocurrent decays after prolonged illumination, likely due to oxidation of Sn²⁺ to Sn⁴⁺, indicating a stability challenge that future work must address.
Conclusion
We have demonstrated a scalable, low‑cost synthesis of SnO₂‑x that exhibits intrinsic visible‑light photocatalytic and photoelectrochemical water‑splitting performance. The activity arises from self‑doping of Sn²⁺ and the resulting oxygen vacancies, which introduce sub‑bandgap states. While the material shows promise for solar‑hydrogen generation, further optimization of stability and activity is underway.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article.
Nanomaterials
- Enhanced Photocatalytic Degradation of Rhodamine B Using an ATP/TiO₂/Ag₃PO₄ Ternary Nanocomposite Under Simulated Solar Light
- Optimizing Broadband Light Absorption in Thin‑Film Silicon Solar Cells with Double‑Sided Pyramid Gratings
- Rapid‑Breakdown Anodized Titania Nanotubes Deliver Superior Sunlight‑Driven Photocatalytic Decolorization of Organic Dyes
- Visible‑Light Photocathodic Protection of 304 Stainless Steel Using Bi₂Se₃‑Sensitized TiO₂ Nanotube Films
- Enhanced Visible‑Light Photocatalysis via In‑Situ Z‑Scheme BiOBr0.3I0.7/Ag/AgI Nanocomposites
- Enhanced Visible‑Light Photocatalysis via Cu2−xSe‑Modified Monoclinic BiVO4: 15.8‑Fold Activity Boost
- Partially BiVO4-Modified ZnO Porous Nanosheets: Solar‑Driven Photocatalysis with Superior Charge Separation
- Large‑Area Monolayer Reduced Graphene Oxide Assembly via Deep UV Irradiation in Solution
- CQD‑Decorated BiFeO3 Nanoparticles Deliver Superior Visible and Near‑Infrared Photocatalysis for AO7 Degradation and Cr(VI) Reduction
- Self‑assembled SnIn4S8/TiO₂ Nanotube Photoanode for Enhanced Visible‑Light Photocathodic Protection of Q235 Steel



