Crystal Structure Drives Supercapacitive Performance of Hydrogenated TiO₂ Nanotube Arrays
Abstract
Engineering the crystal lattice of TiO₂ nanotube arrays (TNAs) is a powerful strategy to enhance their electrochemical performance. In this work, we fabricated hydrogenated TiO₂ TNAs (H@TNAs) with a dominant <001> orientation and tunable rutile‑anatase ratios through anodisation, controlled annealing, and electrochemical hydrogenation. Detailed structural analysis by TEM and refined XRD patterns revealed that the anatase grains preferentially grow along the <001> direction, forming plate‑shaped crystallites stacked vertically along the tube axis. This alignment, combined with hydrogen‑induced lattice disorder and partial rutile transformation, boosts charge carrier density and creates an efficient electron transport highway. As a result, H@TNAs-1 (450 °C, 3 h) achieved a specific capacitance of 20.86 mF cm⁻², while H@TNAs‑3 (650 °C, 2 h) reached 24.99 mF cm⁻², retaining 73 % capacitance at 0.625 mA cm⁻². The electrodes also exhibit excellent rate capability and 94–95 % capacity retention after 5 000 cycles. These findings underscore the critical role of crystal orientation and phase composition in designing high‑performance TiO₂‑based supercapacitors.
Introduction
TiO₂ is a cornerstone material in energy conversion and storage, prized for its low cost, non‑toxicity, and chemical stability. While TiO₂ nanotube arrays (TNAs) fabricated by anodic oxidation provide a high surface area and direct carrier pathways, their wide bandgap limits intrinsic conductivity (10⁻⁵–10⁻² S m⁻¹). Numerous strategies—heterostructuring, hetero‑atom doping, and surface functionalisation—have been explored to overcome this limitation. Among them, hydrogenation stands out for its simplicity and effectiveness in increasing free‑electron density via oxygen vacancy formation, thereby enhancing conductivity and electrochemical activity.
Crystalline phase (anatase vs. rutile) and preferential orientation (<001> vs. random) are pivotal determinants of carrier transport. Prior studies have shown that anatase TNAs with exposed {001} facets exhibit superior photocatalytic performance, yet their impact on supercapacitive behaviour remains underexplored, particularly in hydrogenated systems. This study systematically investigates how controlled crystallinity and orientation influence the electrochemical properties of H@TNAs, providing guidance for next‑generation electrode design.
Methodology
Materials
All reagents were analytical grade and used without further purification. A detailed table of raw materials is provided in the supplementary information.
Synthesis of Hydrogenated <001> Oriented TiO₂ Nanotubes
Titanium sheets (30 × 10 × 0.1 mm³) were ultrasonically cleaned in water, acetone, and ethanol (30 min each). TNAs were grown via two‑step anodisation in a 0.3 g NH₄F‑2 mL H₂O–98 mL ethylene glycol bath at 50 V, 30 °C, 1 h per step. The as‑grown arrays were amorphous and subsequently annealed in Ar at 450 °C (3 h) for anatase TNAs-1, or at 650 °C for 1–3 h to produce mixed‑phase TNAs-2 to TNAs‑4.
Hydrogenation was performed in a two‑electrode cell with 0.5 M Na₂SO₄. TNAs served as cathode, Pt as anode, 2 cm gap, 5 V, 30 s. All samples were designated H@TNAs‑1 through H@TNAs‑4 accordingly.
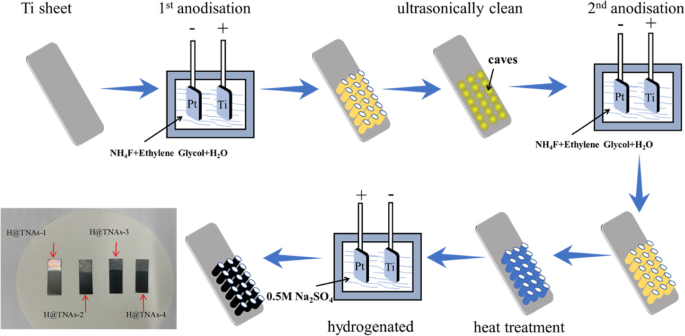
Schematic of synthesis and optical image of H@TNAs.
Characterisations
FESEM (Tescan MIRA3 LMH, 10 kV) examined morphology; XRD (Rigaku Smart Lab SE) recorded 10–100° 2θ with Cu Kα; XRD refinement used March–Dollase WPPF. TEM (JEOL 2100 F, 200 kV) and HR‑TEM provided microstructural detail. XPS (Escalab 250) assessed Ti valence states. Electrochemical tests employed a CHI660D workstation in a three‑electrode setup (0.5 M Na₂SO₄, SCE reference, Pt counter). CV, GCD, and EIS were performed within −0.3–0.5 V, scan rates 10–500 mV s⁻¹, current densities 0.025–0.5 mA cm⁻², and 0.1 Hz–1 MHz frequency ranges, respectively.
Results and Discussion
H@TNAs‑1 exhibited uniform tubes (≈85 nm diameter, 8.3 µm length) that retained their morphology after high‑temperature treatment (Figure 2). XRD confirmed anatase with intense (004) peaks, indicating strong <001> texture (r₍004₎ = 0.2721). TEM revealed plate‑shaped anatase grains stacked along the tube axis, and HR‑TEM showed parallel (001) lattice fringes, providing a direct electron highway.

SEM of H@TNAs‑1: (a) top view, (b) cross‑section.
XPS spectra (Figure 3a) displayed Ti⁴⁺ and Ti³⁺ peaks, confirming oxygen vacancies introduced by hydrogenation. The surface became amorphous, as evidenced by broadened features, which increases active sites and facilitates ion diffusion.
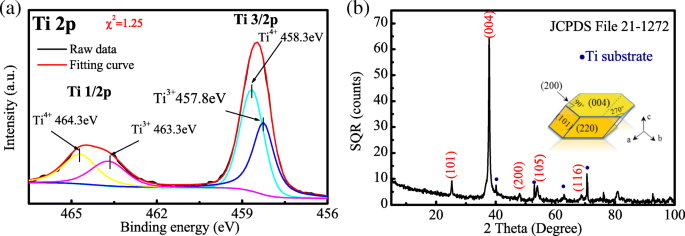
Ti 2p XPS of H@TNAs‑1 and XRD pattern.
Electrochemical evaluation showed quasi‑rectangular CV curves up to 500 mV s⁻¹ (Figure 5a), and symmetric GCD curves across all current densities (Figure 5b). The specific capacitance reached 20.86 mF cm⁻² at 0.025 mA cm⁻², retaining 87.9 % at 0.625 mA cm⁻². EIS displayed a near‑vertical line with negligible semicircle, indicating low charge‑transfer resistance (Rₛ ≈ 0.30 Ω).
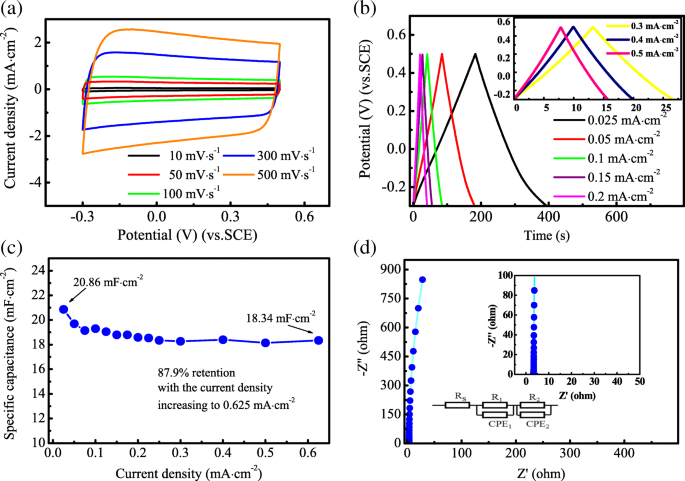
Supercapacitive performance of H@TNAs‑1: (a) CV curves, (b) GCD curves, (c) Nyquist plots, (d) capacitance vs. current density.
Introducing a controlled amount of rutile by annealing at 650 °C (H@TNAs‑2 to –4) maintained the <001> orientation while gradually increasing rutile content, as confirmed by XRD (Figure 8). TEM showed thinner amorphous layers with higher rutile fractions, reducing interfacial resistance but also decreasing oxygen vacancy concentration (Ti³⁺ fraction dropped from ~10 % to ~4 %).
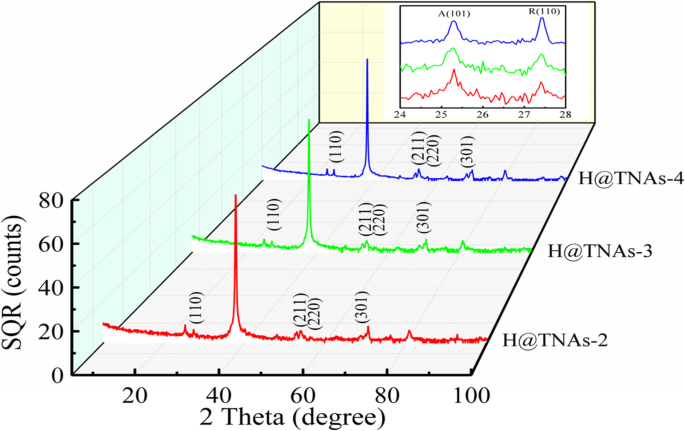
XRD patterns of H@TNAs‑2 to –4.
Electrochemical performance varied with phase composition: H@TNAs‑3 (2 h at 650 °C) delivered the highest capacitance of 24.99 mF cm⁻² and excellent rate capability (73 % retention at 0.625 mA cm⁻²). H@TNAs‑2 showed the highest absolute capacitance (28.23 mF cm⁻²) but suffered a rapid drop at high current density, attributed to residual lattice disorder. H@TNAs‑4, with minimal disorder, maintained high rate performance (only 12 % loss). EIS data (Figure 11e) confirmed that increased rutile reduced diffusion resistance, but excessive disorder increased impedance.
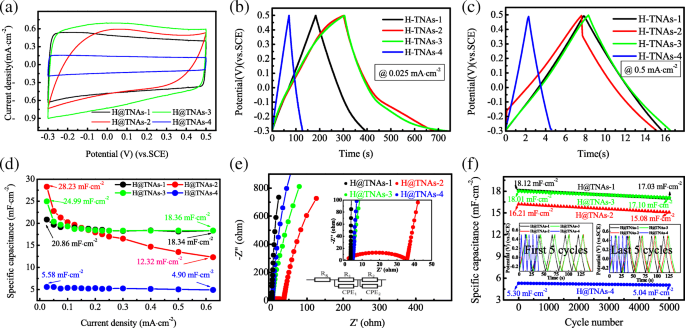
Supercapacitive properties of mixed‑phase H@TNAs: (a) CV, (b) GCD, (c) specific capacitance, (d) Nyquist, (e) cycling performance.
After 5 000 cycles at 0.3 mA cm⁻², all samples retained >93 % of their initial capacitance, demonstrating robust structural integrity. The combination of <001> oriented anatase platelets, controlled rutile bridges, and hydrogen‑induced amorphous layers synergistically enhances electron transport and ion accessibility.
Conclusion
This study demonstrates that tailoring the crystal structure—specifically, enforcing a <001> orientation and introducing a balanced rutile‑anatase phase mixture—dramatically improves the supercapacitive performance of hydrogenated TiO₂ nanotube arrays. The optimized H@TNAs‑3 electrode achieved 24.99 mF cm⁻² at 0.025 mA cm⁻², with excellent rate capability and cycling stability. The findings provide a clear blueprint for designing high‑efficiency TiO₂‑based supercapacitor electrodes through precise crystallographic engineering.
Availability of Data and Materials
All data supporting this work are available upon reasonable request from the corresponding author.
Abbreviations
- CV:
Cyclic voltammetry
- EIS:
Electrochemical impedance spectroscopy
- FESEM:
Field emission scanning electron microscopy
- H@TNAs:
Hydrogenated TiO₂ nanotube arrays
- H@TNAs‑1:
Annealed at 450 °C for 3 h before hydrogenation
- H@TNAs‑2:
Annealed at 650 °C for 1 h before hydrogenation
- H@TNAs‑3:
Annealed at 650 °C for 2 h before hydrogenation
- H@TNAs‑4:
Annealed at 650 °C for 3 h before hydrogenation
- HR‑TEM:
High‑resolution transmission electron microscopy
- SAED:
Selected area electron diffraction
- TEM:
Transmission electron microscopy
- TNAs:
TiO₂ nanotube arrays
- WPPF:
Whole powder pattern fitting
- XPS:
X‑ray photoelectron spectroscopy
- XRD:
X‑ray diffraction
Nanomaterials
- How Water Alters the Structure and Dielectric Behavior of Microcrystalline Cellulose
- Enhanced Photocatalytic Water Splitting with Au‑Plasmonic Nanoparticles on TiO₂/MoO₃ Nanotube Photoelectrodes
- Chromium‑Doped Titanium Dioxide: A Novel Colored Cool Pigment with High Near‑Infrared Reflectance
- Optimizing TiO₂ Nanotube Arrays via Soft–Hard Template for Superior Field Emission Performance
- Transition Metal‑Doped TiO₂ Nanoparticles: Surface‑Spectroscopic Insight into Catalytic Activity
- Comparative Analysis of Electrochemical, Biomedical, and Thermal Properties of Natural vs. Synthetic Nanomaterials
- Facile Thermal Decomposition Loading of CeO₂ Nanoparticles onto Anodic TiO₂ Nanotube Arrays
- Impact of Morphology and Crystal Structure on Titania Nanotube Thermal Conductivity
- Titania Nanotube Arrays via Electrochemical Anodization: Synthesis, Modifications, and Biomedical Applications
- Ga‑Doped LiNi0.5−xGa_xMn1.5O4 Spinel Cathodes: Enhanced High‑Temperature Stability and Rate Capability



