Using GaN/Fe Nanoparticles to Magnetically Guide Endothelial Cells in Vitro
Abstract
We report the synthesis and cellular uptake of multifunctional GaN/Fe nanoparticles and demonstrate their utility as magnetic guides for endothelial cells. The particles are produced by growing a GaN shell on ZnO/Fe2O3 cores via hydride vapor phase epitaxy (HVPE) followed by core decomposition at high temperature. Transmission electron microscopy confirms that porcine aortic endothelial cells internalize the GaN/Fe nanoparticles into vesicles without compromising cell viability. These inert, magnetically responsive particles can be used to steer cells in external magnetic fields, offering a new strategy for targeted cell delivery and tissue engineering.
Background
Nanotechnology has emerged as a powerful tool in oncology, largely through nanoparticle-based drug carriers [1,2]. However, ligand coating, drug encapsulation, and covalent attachment can limit delivery efficiency and raise safety concerns. An alternative strategy involves using nanoparticles as direct biological modulators—targeting and guiding cells themselves [3]. For instance, magnetic nanoparticles can direct endothelial cells to sites of arterial injury when exposed to an external magnetic field. Beyond therapeutic applications, such guided cells can aid in in‑vitro cell sorting and scaffold functionalization [4]. Here we present the first evidence that endothelial cells can internalize GaN/Fe nanoparticles and that this uptake enables controlled spatial positioning of the cells.
Methods
Nanoparticle Synthesis
Thin GaN layers were grown on ZnO nanoparticles alloyed with Fe2O3 using a two‑step HVPE process. An initial nucleation layer was deposited at 600 °C for 5 min, followed by a growth phase at 800 °C for 10 min to decompose the ZnO core and improve GaN crystalline quality. The process employed metallic gallium, NH3, HCl, and H2 as carrier gases at flow rates of 20, 600, and 3500 sccm, respectively [5,6].
Cell Culture
Primary porcine aortic endothelial cells were isolated by gentle scraping of the aortic wall, then cultured in EGM™-2 (Lonza) at 37 °C, 5% CO2. Passages 3–8 were used, and cells were transduced with GFP via lentivirus [7]. Cell detachment for experiments was performed with TrypLE™Select(1X) (Gibco®).
XTT Assay
Cell viability was assessed 24 h after adding nanoparticles. XTT reagent (0.1 ml in 5 ml XTT) was mixed with fresh EGM2 medium (2:1) and incubated for 4 h at 37 °C, 5% CO2. Absorbance was measured on a Paradigm multi‑mode plate reader.
Cell Counting
After 48 h of nanoparticle exposure, cells were fixed in 4% paraformaldehyde, washed with PBS, and stained with DAPI (1:7500). Images were captured from six wells per condition using a Zeiss fluorescence microscope and quantified with DotCount v1.2 [8].
Transmission Electron Microscopy
Cells incubated for 24 h with 50 µg/ml GaN/Fe nanoparticles were fixed in 2% glutaraldehyde/2% formaldehyde, post‑fixed in 1% OsO4, dehydrated, and embedded in EPON. Ultra‑thin sections (~50 nm) were stained with 4% uranyl acetate and lead citrate, then examined on an FEI Tecnai 20 (200 kV).
Results and discussion
GaN/Fe nanoparticles were engineered by coating ZnO/Fe2O3 cores with GaN, followed by core removal. The resulting particles exhibit a GaN shell (~20–100 nm) embedded with Fe, conferring magnetic responsiveness while retaining the piezoelectricity of GaN. SEM, TEM, XRD, Raman, and EDX analyses confirm core decomposition, GaN growth, and a Fe content of ~50% [Figure 1].
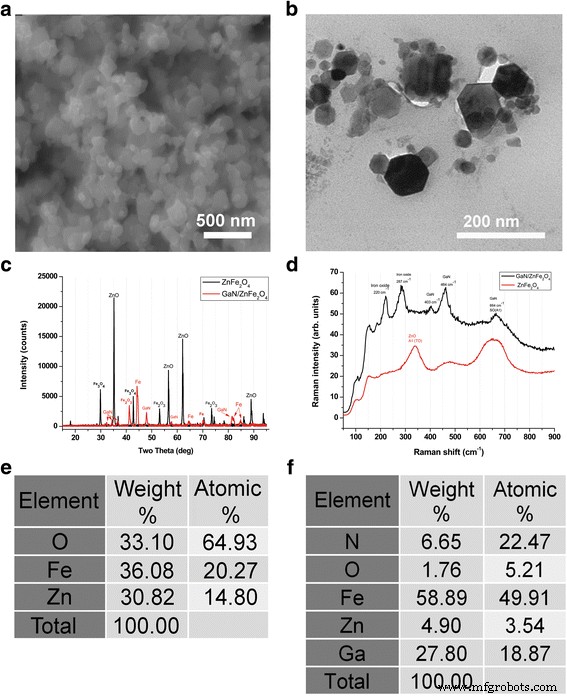
Analysis of nanoparticles. a SEM of GaN on ZnO/Fe2O3 cores. b TEM of GaN/Fe particles. c XRD of ZnFe2O4 vs. GaN/ZnFe2O4. d Raman spectra before and after GaN growth. e EDX of ZnO/Fe2O3. f EDX after GaN growth.
When incubated with porcine aortic endothelial cells, the nanoparticles were predominantly internalized into vesicles, with no cytoplasmic or nuclear localization (Fig. 3a). Uptake occurred via classical pathways—micropinocytosis, clathrin‑mediated, and caveolin‑mediated endocytosis—likely dominated by caveolae in endothelial cells [9,10,11]. Viability assays revealed a modest, concentration‑dependent reduction in metabolic activity at 50 µg/ml, while lower doses (<10 µg/ml) maintained >90% viability (Fig. 2).
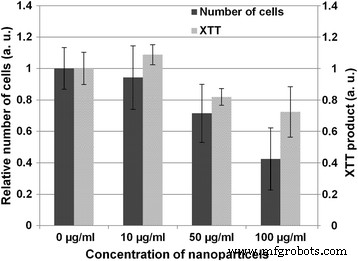
Impact of nanoparticles on cell viability. XTT reduction after 1 day of incubation with various concentrations. Results are expressed relative to untreated controls (mean ± SD, n = 6).
Given the high Fe loading, the particles exhibit strong ferromagnetism, while the GaN shell preserves piezoelectricity. These dual properties enable remote actuation via ultrasound‑induced polarization and magnetic manipulation. To validate magnetic guidance, endothelial cells loaded with 50 µg/ml GaN/Fe were detached, re‑suspended, and plated onto EGM™-2. When the culture plate was positioned beneath permanent magnets, the cells displayed a highly non‑random distribution that matched the magnetic field map, whereas control cells showed random placement (Fig. 4). This demonstrates that GaN/Fe nanoparticles can be used to direct cells in a predictable manner.
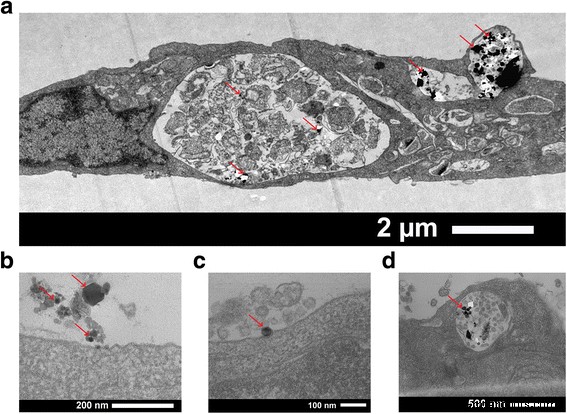
TEM images of a single endothelial cell after 1 day with GaN/Fe nanoparticles. a Vesicular distribution. b–d Uptake pathways (red arrows denote high‑density particles).
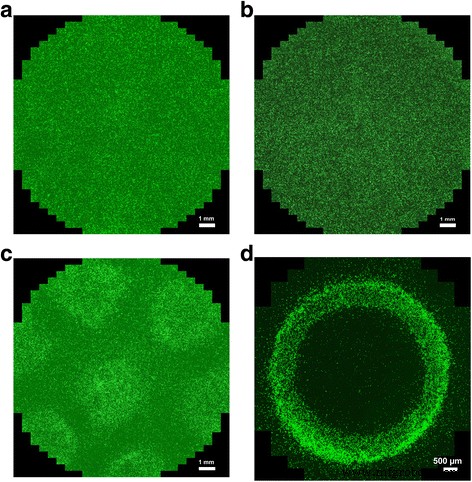
Guiding of nanoparticle‑laden endothelial cells with magnetic fields. a Cells with nanoparticles, no field. b Cells without nanoparticles, field applied. c Cells with nanoparticles under seven neodymium magnets. d Cells with nanoparticles under a ring magnet.
Conclusions
We have established that GaN/Fe nanoparticles are safely internalized by endothelial cells and stored within vesicles. Their magnetic responsiveness allows precise, field‑driven positioning of the cells, opening avenues for targeted cell delivery and the fabrication of engineered tissues. Additionally, the intrinsic piezoelectricity of GaN offers a platform for remote electrical stimulation of cellular functions. Ongoing work will explore in vivo applications and the integration of piezoelectric activation for therapeutic modulation.
Abbreviations
- EDX:
Energy‑dispersive X‑ray analysis
- EGM™-2:
Endothelial Growth Medium 2
- Fe:
Iron
- Fe2O3:
Iron(III) oxide
- GaN:
Gallium nitride
- GFP:
Green fluorescent protein
- H2:
Hydrogen
- HCl:
Hydrogen chloride
- NH3:
Ammonia
- OsO4:
Osmium tetroxide
- PBS:
Phosphate‑buffered saline
- SEM:
Scanning electron microscopy
- TEM:
Transmission electron microscopy
- XRD:
X‑ray diffraction
- ZnO:
Zinc oxide
Nanomaterials
- Ag Nanoparticle-Enhanced BiV1‑xMoxO4 Composites: Synergistic Photocatalytic Performance under Visible Light
- Gold Nanoparticle–Modified 6‑Mercaptopurine and Rabies‑Derived Peptide Enhance SH‑SY5Y Neural Cell Proliferation and Neurite Outgrowth
- PEG-CoFe₂O₄ Nanoparticles: Assessing Toxicity and Curcumin’s Protective Effect
- Optimizing Perovskite Solar Cell Efficiency with Size‑Controlled Ag Nanoparticles in a TiO₂ Compact Layer
- Targeted Lipid Nanoparticles Functionalized with Transferrin Enhance Paclitaxel Efficacy in Leukemia Cells
- Lithocholic Acid–Functionalized Gold Nanoparticles Induce Selective Apoptosis in Liver Cancer Cells
- Lanthanum Strontium Manganite Nanoparticles: Airway Epithelial Cell Toxicity and Mucus Secretion Impact
- Enhanced Gene Delivery Using Gold Nanoparticles Functionalized with Chitosan, N‑Acylated Chitosan, and Chitosan Oligosaccharide
- Reducing Cytotoxicity of Silver Nanoparticle Coatings with Graphene Oxide: Enhanced Antibacterial Performance and Biocompatibility of an Ag–GO Nanocomposite
- Hydroxyapatite Nanoparticles Enhance MSC Osteogenesis via Endothelial Cell Interaction



