Lithocholic Acid–Functionalized Gold Nanoparticles Induce Selective Apoptosis in Liver Cancer Cells
Abstract
Gold nanoparticles (AuNPs) are renowned for their biocompatibility, optical properties, and drug‑delivery potential. In this study, we engineered AuNPs with a polyethylene glycol (PEG) spacer and lithocholic acid (LCA) ligand via carboxylate coupling, yielding AuNP@MPA‑PEG‑LCA. Cytotoxicity assays revealed that AuNP@MPA‑PEG‑LCA exhibited pronounced selectivity, markedly inhibiting multiple hepatocellular carcinoma (HepG2, SMMC‑7721) cells while sparing normal hepatocytes (QSG‑7701) and breast cancer cells (MCF‑7). Apoptotic induction was confirmed through nuclear condensation (Hoechst 33342), annexin V‑FITC binding, mitochondrial membrane potential (MMP) loss (JC‑1), and acridine orange/ethidium bromide (AO/EB) staining. The nanoparticles triggered reactive oxygen species (ROS) accumulation, leading to mitochondrial dysfunction and programmed cell death. These findings demonstrate that AuNP@MPA‑PEG‑LCA is a promising, targeted nanotherapeutic for liver cancer.
Background
Gold nanoparticles possess unique optical resonance, chemical stability, and an extensive surface area that make them attractive for biomedical applications, including drug delivery, imaging, and photothermal therapy [1–5]. Their spherical geometry facilitates efficient cellular uptake and minimizes off‑target effects [6–8]. Functionalization of AuNPs with targeting ligands or polymers further enhances their therapeutic index, particularly for oncology [9–14].
Lithocholic acid (LCA), a secondary bile acid, has been reported to exert potent antitumor activity against liver cancer via apoptosis [29–30]. As a naturally occurring molecule, LCA can serve as a ligand to guide AuNPs to hepatic tissues, improve solubility, and potentially modulate intracellular signaling pathways [20–28].
Combining the biocompatibility of AuNPs with the tumor‑suppressive properties of LCA may yield a selective anticancer agent that exploits ROS‑mediated mitochondrial pathways to trigger apoptosis in hepatocellular carcinoma cells.
Methods
Materials
All reagents were purchased from Sigma‑Aldrich unless noted otherwise. Cell culture reagents (RPMI‑1640, FBS) were obtained from Invitrogen. Human hepatocellular carcinoma lines (HepG2, SMMC‑7721), normal hepatocytes (QSG‑7701), and breast cancer cells (MCF‑7) were acquired from the Shanghai Institute for Biological Sciences.
Synthesis of AuNP@MPA
Citrate‑capped AuNPs (average diameter 4.0 ± 0.5 nm) were synthesized by the Turkevich method [38] using HAuCl₄ and NaBH₄. The resulting nanoparticles were subsequently capped with 3‑mercaptopropionic acid (MPA) at pH 11 to provide carboxylate groups for further conjugation.
Conjugation with PEG
AuNP@MPA (0.0875 mmol) was activated with NHS (0.0875 mmol) and EDC (0.035 mmol) in 50 mM MES buffer, followed by addition of NH₂‑PEG₁₀₀₀‑NH₂ (0.045 mmol). After 24 h at 25 °C, the PEGylated nanoparticles (AuNP@MPA‑PEG) were isolated by centrifugation.
Attachment of Lithocholic Acid
AuNP@MPA‑PEG was mixed with LCA (0.045 mmol) in anhydrous DMF and stirred for 24 h at 25 °C. The final product, AuNP@MPA‑PEG‑LCA, was purified by repeated centrifugation.
Transmission Electron Microscopy (TEM)
Particle morphology and size were examined on a JEOL JEM‑200CX TEM operating at 200 kV. Nanoparticle suspensions were drop‑cast onto 300 mesh copper grids.
Cytotoxicity Assays
Cell viability after 48 h exposure to AuNPs (0.2–1.0 mg mL⁻¹) was measured using the MTT assay. Absorbance at 570 nm was recorded on a Tecan Infinite M200 plate reader.
Apoptotic Morphology (Hoechst 33342)
HepG2 cells were treated with 0.5 mg mL⁻¹ AuNP@MPA‑PEG‑LCA for 24 and 48 h, then stained with 10 µg mL⁻¹ Hoechst 33342 for 30 min. Nuclear morphology was imaged with a Leica‑SP8 confocal microscope.
Early Apoptosis (Annexin V‑FITC)
After 6 h treatment with 0.5 mg mL⁻¹ AuNP@MPA‑PEG‑LCA, HepG2 cells were stained with annexin V‑FITC and analyzed by flow cytometry (FACSCalibur, BD). Confocal images were also acquired.
Mitochondrial Membrane Potential (MMP)
JC‑1 dye (10 µg mL⁻¹) was used to assess MMP changes in HepG2 cells treated with AuNP@MPA‑PEG‑LCA (6 h). Both flow cytometry and confocal microscopy were employed.
Reactive Oxygen Species (ROS)
Intracellular ROS were quantified using H₂DCF‑DA (10 µM) after 6 h exposure to 0.5 mg mL⁻¹ AuNP@MPA‑PEG‑LCA. Fluorescence was measured by flow cytometry and confocal microscopy.
Results and Discussion
Nanoparticle Characterization
The AuNP@MPA‑PEG‑LCA maintained a spherical shape with an average diameter of 16.0 ± 1.2 nm, as confirmed by TEM (Figure 1c). The PEG and LCA layers contributed to the increased hydrodynamic size compared to the core AuNP@MPA (4.0 nm). These dimensions are ideal for enhanced permeability and retention (EPR) effect in tumor tissues.
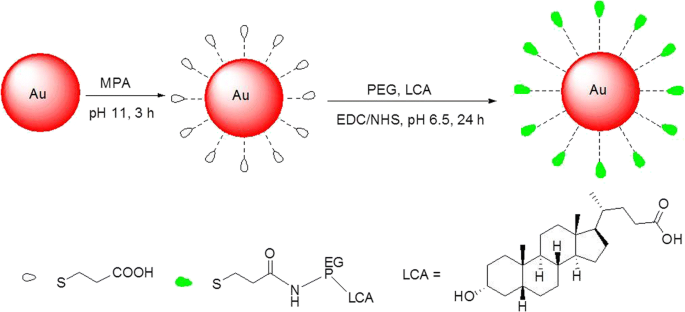
Schematic representation of synthesis of the AuNP@MPA‑PEG‑LCA

TEM images of a AuNP@MPA, b AuNP@MPA‑PEG, and c AuNP@MPA‑PEG‑LCA
Selective Cytotoxicity
MTT assays (Figure 2) showed negligible cytotoxicity of AuNP@MPA across all cell lines. In contrast, AuNP@MPA‑PEG‑LCA dose‑dependently reduced viability of HepG2 and SMMC‑7721 cells, with IC₅₀ values below 0.6 mg mL⁻¹, while QSG‑7701 and MCF‑7 cells were minimally affected. This selective profile underscores the targeting capability conferred by LCA.
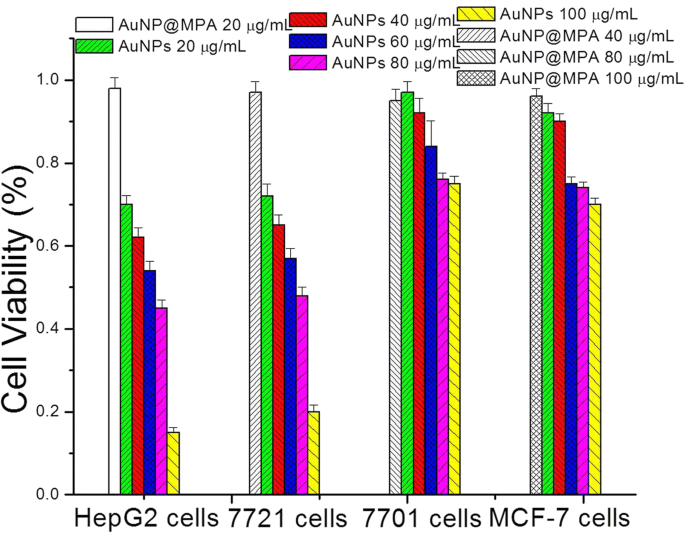
Cell viability of AuNP@MPA and AuNP@MPA‑PEG‑LCA (AuNPs) incubated with HepG2, SMMC‑7721, QSG‑7701, and MCF‑7 cells for 48 h
Apoptotic Morphology
Hoechst staining (Figure 3) revealed progressive nuclear condensation, fragmentation, and chromatin margination in HepG2 cells treated with AuNP@MPA‑PEG‑LCA over 48 h, indicative of late apoptosis. Control cells and AuNP@MPA‑treated cells maintained uniform nuclear morphology.
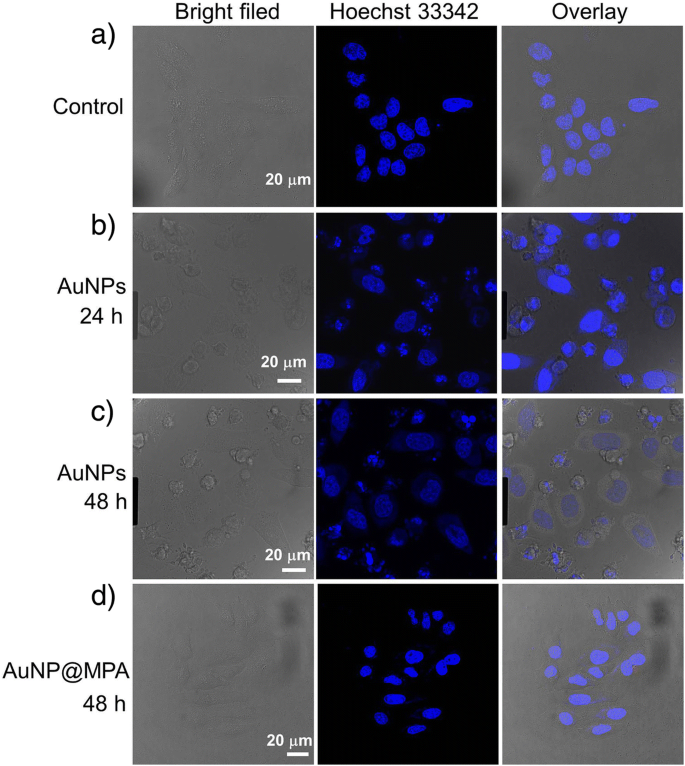
Morphologic characteristics of cell nucleus of HepG2 cells stained with Hoechst 33342. HepG2 cells were incubated with AuNP@MPA‑PEG‑LCA (0.5 mg mL⁻¹) for a 0 h, b 24 h, and c 48 h, and d AuNP@MPA (0.5 mg mL⁻¹) incubated for 48 h. Apoptotic cells displayed condensed and fragmented nuclei, and shrinkage of cell volume; scale bar 20 µm
Annexin V‑FITC Confirmation
Annexin V staining (Figure 4) demonstrated a clear translocation of phosphatidylserine to the outer leaflet of the plasma membrane in AuNP@MPA‑PEG‑LCA‑treated cells. Flow cytometry quantified a 38.45 % apoptosis rate versus 8.16 % in controls, confirming early apoptotic induction.
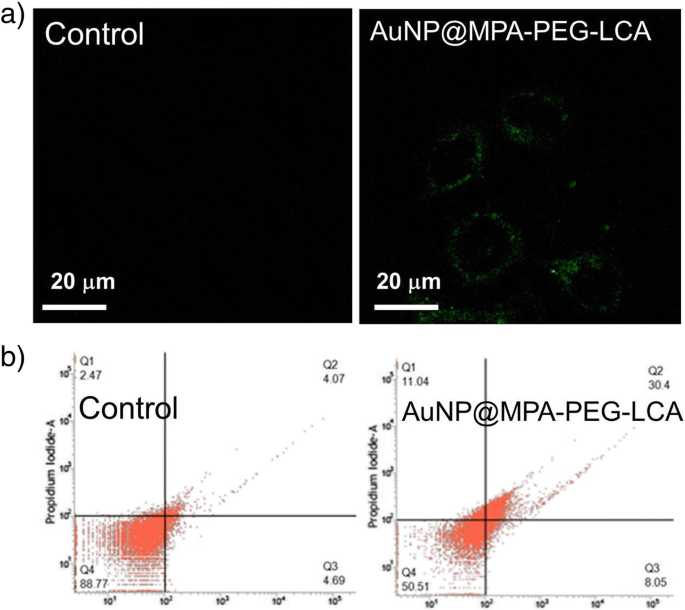
The apoptosis results of a confocal images and b flow cytometry data of HepG2 cells treated with AuNP@MPA‑PEG‑LCA(0.5 mg mL⁻¹). Cells were stained with annexin V‑FITC (excitation at 488 nm and emission at 500–560 nm); scale bar 20 µm
Mitochondrial Depolarization
JC‑1 analysis (Figure 5) revealed a loss of red aggregate fluorescence and an increase in green monomer signal, indicating MMP collapse in treated cells. Flow cytometry confirmed a significant reduction in the red/green ratio, corroborating mitochondrial dysfunction as a downstream event of apoptosis.
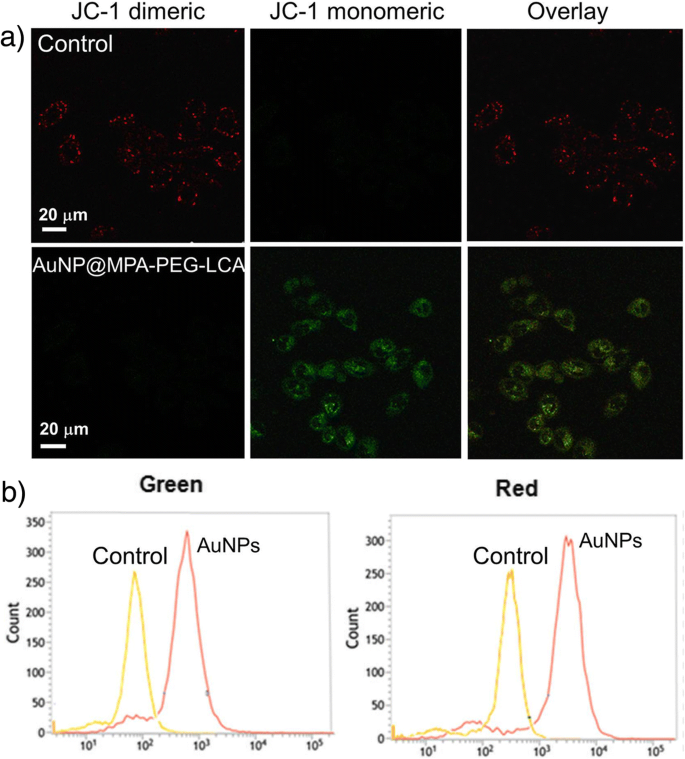
a Fluorescence images of JC‑1 labeled cells viewed by confocal microscopy and b effects of AuNP@MPA‑PEG‑LCA on MMP analyzed by flow cytometry
ROS Generation
H₂DCF‑DA staining (Figure 6) showed a marked increase in green fluorescence in AuNP@MPA‑PEG‑LCA‑treated HepG2 cells, reflecting elevated ROS production. Flow cytometric quantification confirmed the trend, suggesting that oxidative stress precedes mitochondrial depolarization and apoptosis.
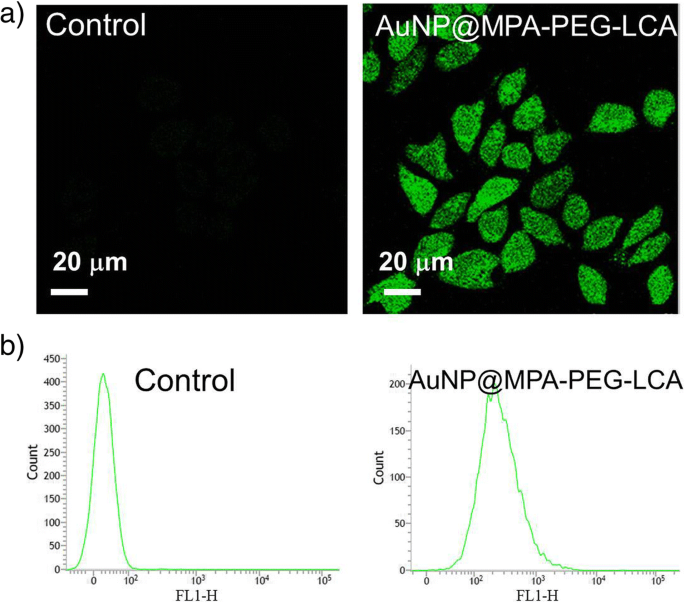
Analysis of ROS production after HepG2 cells was treated with AuNP@MPA‑PEG‑LCA for 6 h. The content of ROS in HepG2 cells was investigated by a confocal microscopy (excitation at 488 nm and emission at 530 nm) and b flow cytometry (excitation at 488 nm and emission at 525 nm)
Conclusions
We successfully fabricated AuNP@MPA‑PEG‑LCA nanoparticles (~16 nm) that selectively inhibit hepatocellular carcinoma cell growth by triggering apoptosis. Comprehensive assays—nuclear morphology, annexin V binding, JC‑1 MMP loss, and ROS elevation—confirm that the therapeutic action is mediated through ROS‑induced mitochondrial dysfunction. These results position AuNP@MPA‑PEG‑LCA as a promising, targeted nanomedicine for liver cancer treatment.
Abbreviations
- AuNPs:
Gold nanoparticles
- DMF:
Dimethylformamide
- EDC:
1-(3‑Dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride
- FBS:
Fetal bovine serum
- H₂DCF‑DA:
2′,7′‑Dichlorofluorescein diacetate
- HepG2:
Human hepatocellular liver carcinoma cells
- JC‑1:
5,5′,6,6′‑Tetrachloro‑1,1′,3,3′‑tetraethylbenzimidazolylcarbocyanine iodide
- LCA:
Lithocholic acid
- MCF‑7:
Human breast cancer cells
- MES:
4‑Morpholineethanesulfonic acid
- MMP:
Mitochondrial membrane potential
- MPA:
3‑Mercaptopropionic acid
- MTT:
3-(4,5‑Dimethyl‑2‑thiazolyl)-2,5‑diphenyltetrazolium bromide
- NHS:
1‑Hydroxypyrrolidine‑2,5‑dione
- PEG:
Polyethylene glycol
- PS:
Phosphatidylserine
- QSG‑7701:
Human normal hepatocyte cells
- ROS:
Reactive oxygen species
- SMMC‑7721:
Human hepatocellular liver carcinoma cells
- TEM:
Transmission electron microscopy
Nanomaterials
- Visible‑Light‑Assisted Au Nanoparticle‑Modified Glassy Carbon Electrodes for Sensitive Uric Acid Detection
- Optimizing Gold Nanoparticle Placement in TiO₂ Enhances Dye‑Sensitized Solar Cell Efficiency
- Using GaN/Fe Nanoparticles to Magnetically Guide Endothelial Cells in Vitro
- Gold Nanoparticle–Modified 6‑Mercaptopurine and Rabies‑Derived Peptide Enhance SH‑SY5Y Neural Cell Proliferation and Neurite Outgrowth
- PEG-CoFe₂O₄ Nanoparticles: Assessing Toxicity and Curcumin’s Protective Effect
- Green‑Synthesized Protein‑Coated Gold Nanoparticles from Tricholoma crassum: Antimicrobial, Apoptotic, and Gene‑Delivery Potentials
- Optimized Mitoxantrone Delivery Using Cholesterol‑Modified Pullulan Nanoparticles: Size‑Dependent Antitumor Efficacy Against Bladder Cancer
- Laser‑Assisted MAPLE Deposition of Antibody‑Functionalized Upconversion Nanoparticles Enhances Cell Culture Performance
- Reducing Cytotoxicity of Silver Nanoparticle Coatings with Graphene Oxide: Enhanced Antibacterial Performance and Biocompatibility of an Ag–GO Nanocomposite
- Ultrasmall Superparamagnetic Iron Oxide Nanoparticles for Stem Cell Tracking in Parkinson’s Therapy



