Laser‑Assisted MAPLE Deposition of Antibody‑Functionalized Upconversion Nanoparticles Enhances Cell Culture Performance
Abstract
A high‑performance cell culture substrate relies on an optimal surface that supports cell function and communication. Recent work demonstrates that nanostructured coatings can enhance cell adhesion, yet conventional solution‑based methods often introduce contaminants. Matrix‑assisted pulsed laser evaporation (MAPLE) offers a clean, non‑destructive route to deposit biopolymers onto a variety of substrates. In this study, we synthesized NaGdF4:Yb3+,Er3+ upconversion nanoparticles (UCNPs) and their immunoglobulin‑G (IgG) functionalized counterparts via a single‑pot method. The UCNPs exhibit an average diameter of 50 ± 8 nm, and TEM images confirm the presence of IgG on their surfaces. Using a 532 nm Nd:YAG laser (10 Hz), we deposited UCNPs—with and without IgG—onto glass‑bottom culture dishes by MAPLE. Human umbilical vein endothelial cells (HUVECs) were then seeded on these substrates and compared with gelatin‑coated glass controls. Cell viability assays revealed no cytotoxicity, while morphological analysis showed improved adhesion and proliferation on both UCNP‑coated surfaces. These findings demonstrate that MAPLE‑deposited, antibody‑functionalized UCNPs can serve as biocompatible, high‑performance coatings for cell culture applications.
Introduction
Epithelial cells line the internal and external surfaces of the human body, playing a dual role as a physical barrier and active participant in physiological processes such as barrier function, signaling, and immune defense [1]. In tissue engineering, the interaction between cells and substrate surfaces is a critical determinant of cell adhesion, morphology, and function. Traditional protein coatings—collagen, gelatin, fibronectin—have long been employed to enhance cell attachment on glass or plastic surfaces. Recent advances show that nanostructured coatings can modulate cell behavior by providing topographical cues and increasing effective surface area, thereby improving adhesion and proliferation [2–4].
Upconversion nanoparticles (UCNPs) doped with lanthanide ions, such as NaGdF4:Yb3+,Er3+, exhibit efficient near‑infrared (NIR) excitation and visible or NIR emission, offering advantages for deep‑tissue imaging and biosensing due to low phototoxicity and high photostability [5–9]. Surface functionalization of UCNPs with biomolecules—avidin, DNA, antibodies—has enabled targeted imaging, sensing, and cell labeling [19–21]. However, limited work has explored antibody‑functionalized UCNPs as surface coatings for cell culture.
Conventional deposition techniques (sol‑gel, spin coating, dip coating) suffer from uncontrolled thickness, solvent residue, and potential denaturation of biomolecules. Matrix‑assisted pulsed laser evaporation (MAPLE) circumvents these issues by entrapping the target material in a frozen solvent matrix; laser energy is absorbed by the matrix, leading to gentle transfer of the material onto the substrate without direct ablation of the biomolecule [24,25]. MAPLE has been applied in drug delivery, organic electronics, and biomedical coatings [31–35], yet its impact on cell‑culture substrates remains underexplored.
In this work, we report the synthesis of NaGdF4:Yb3+,Er3+ UCNPs and their IgG‑functionalized analogues, followed by MAPLE deposition onto glass‑bottom culture dishes. We then evaluate the biocompatibility and influence on human umbilical vein endothelial cell (HUVEC) behavior, providing a foundation for future tissue engineering applications.
Methods/Experimental
Materials
All reagents were analytical grade: Gd(NO3)3·6H2O (99.9 %), Yb(NO3)3·5H2O (99.9 %), Er(NO3)3·5H2O (99.9 %), NaF (≥ 99 %), branched polyethylenimine (PEI, Mw ≈ 800), anti‑human IgG (Fab‑specific, FITC‑labelled), DAPI, Phalloidin‑TRITC, and solvents (ethylene glycol, isopropanol). The materials were purchased from Sigma‑Aldrich, Fisher Scientific, and Caledon.
Synthesis of UCNPs With and Without IgG by a One‑Pot Process
UCNPs (NaGdF4:Yb3+,Er3+) were synthesized via a modified hydrothermal route. In brief, 720 mg Gd(NO3)3·6H2O, 170 mg Er(NO3)3·5H2O, 160 mg Yb(NO3)3·5H2O, and 20 mL ethylene glycol were mixed in a three‑neck flask. 0.7 g PEI was added, followed by dropwise addition of 336 mg NaF dissolved in 10 mL ethylene glycol. The mixture was heated to 200 °C under nitrogen and refluxed for 6 h. The product was collected by centrifugation, washed with ethanol and water, and dried at 60 °C.
For IgG functionalization, 15 mg UCNP powder was dispersed in 15 mL distilled water, then 5 mL borate‑buffered saline (pH 8) and 20 µg IgG were added. The mixture was stirred at room temperature for 2 h, then washed and pelleted by centrifugation. The conjugate, UCNPs‑IgG, was stored in aqueous buffer.
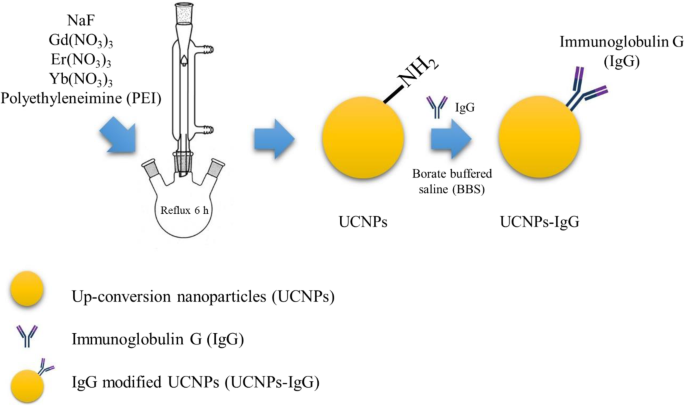
Schematic of one‑pot synthesis of UCNPs and UCNPs‑IgG
Deposition of UCNPs With/Without IgG by MAPLE
MAPLE deposition was performed using a MAPLE 2000 system (PVD Products, Inc.) with a 532‑nm Nd:YAG laser. The nanoparticle solution (1 wt % in isopropanol) was frozen in a liquid‑nitrogen‑cooled target holder. Laser parameters: 10 Hz, 200 µs pulse width, 150 mJ cm–2 fluence, 0.63 cm2 spot area. Glass substrates, pre‑coated with 2 wt % gelatin, were positioned 4.5 cm from the target and maintained at 25 °C under 1 × 10–6 Torr. The target and substrate holders rotated at 10 rpm and 25 rpm, respectively, during a 2‑h deposition. The resulting films were characterized by EDX, FTIR, and photoluminescence (PL) spectroscopy.
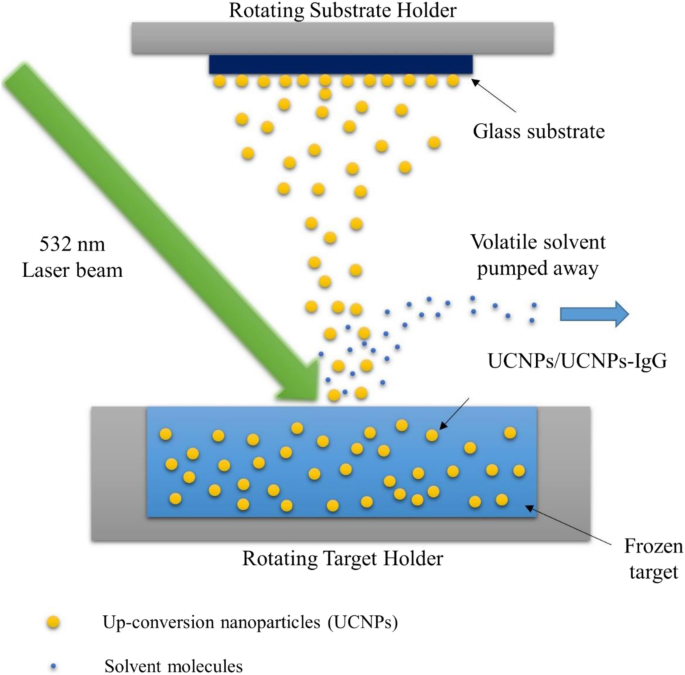
Schematic of MAPLE deposition of UCNPs and UCNPs‑IgG
Materials Characterization
Pre‑deposition UCNPs and UCNPs‑IgG were examined by transmission electron microscopy (TEM, Philips CM‑10, 80 kV). Fourier‑transform infrared (FTIR) spectra were recorded on a Bruker Vector 22 (600–4500 cm–1, 4 cm–1 resolution, 64 scans). PL spectra were measured with a QuantaMaster 40 spectrofluorometer (Horiba). Post‑deposition films were inspected by EDX (Hitachi S‑3400 N with INCA PentaFET‑x3) and confocal microscopy (Zeiss LSM 5 Duo). Cytocompatibility was evaluated via MTT assay on HUVECs cultured for 24 h.
Cell Behavior Study
HUVECs (ATCC) were seeded at ~1 × 10⁵ cells per dish on glass substrates (control gelatin, UCNPs, UCNPs‑IgG). After 24 h incubation at 37 °C, cells were fixed with 4 % formalin, stained with Phalloidin‑TRITC (actin) and DAPI (nuclei), and imaged by confocal microscopy. Cell area, connection length, cell length, and cell count were quantified using ImageJ.
Cytotoxicity Assay
MTT (3‑(4,5‑dimethylthiazol‑2‑yl)‑2,5‑diphenyl‑tetrazolium bromide) was added to the culture medium after 24 h, incubated for 3 h, and absorbance measured at 490 nm using a Bio‑Tek EL340I reader. Experiments were performed in triplicate.
Results and Discussion
Characterization of UCNPs and UCNPs‑IgG Before MAPLE Deposition
TEM images confirm the cubic morphology of UCNPs with an average diameter of 50 ± 8 nm. High‑resolution TEM reveals a lattice spacing of 0.312 nm, corresponding to the (111) plane of cubic NaGdF4 (JCPDS 27‑0697). After IgG conjugation, the particle size increases to 54 ± 8 nm, and the antibody layer (~10 ± 5 nm) is visible as a peripheral halo (Fig. 3b).
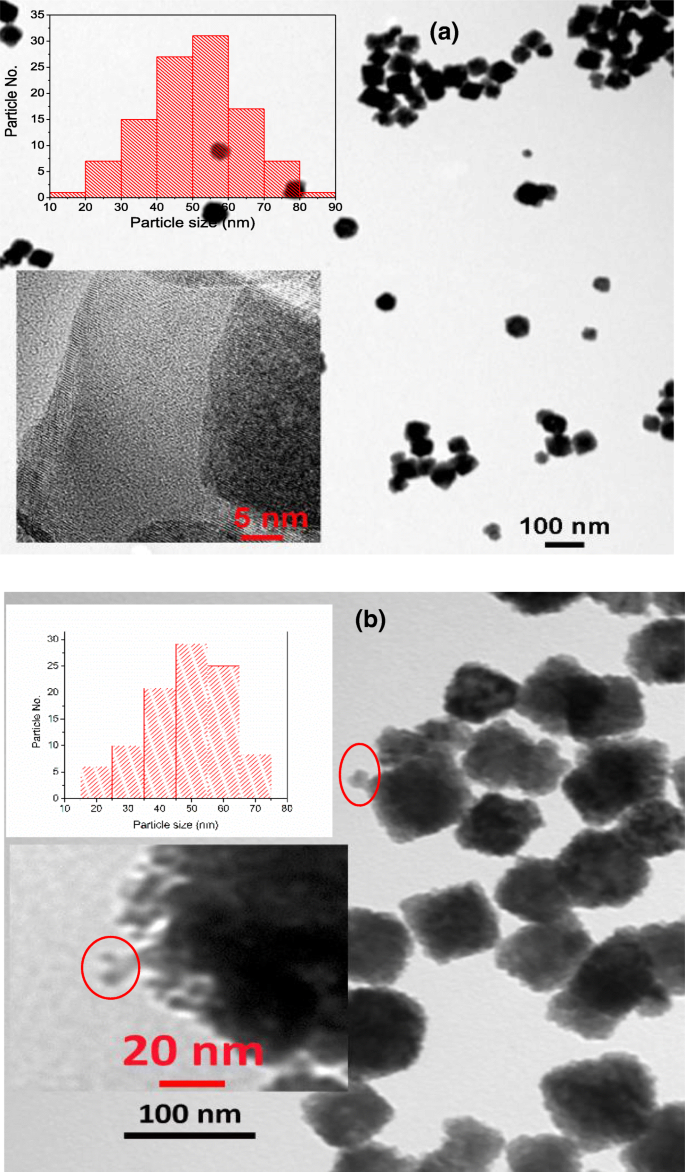
TEM micrographs of a UCNPs and b UCNPs‑IgG before MAPLE deposition
Powder XRD shows characteristic peaks at 32°, 37°, 54°, and 65° (111, 200, 220, 311), confirming the cubic phase (Fig. 4).
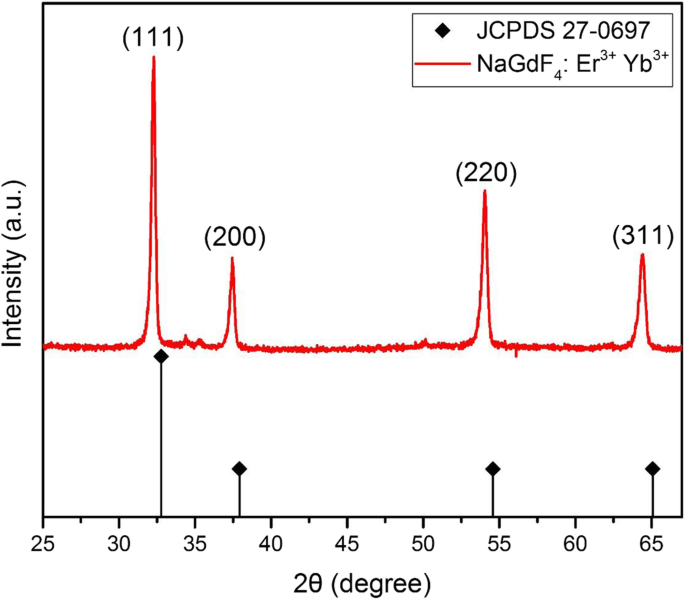
XRD profile of NaGdF4:Yb3+,Er3+ upconversion nanoparticles
FTIR spectra reveal amine peaks (1515 cm–1, 1511 cm–1) from PEI and IgG, –CH2–/C‑C stretches (2987, 2900, 1400 cm–1), and carboxyl and amide bands (1249, 1650 cm–1) confirming IgG attachment (Fig. 5).
FTIR spectra of UCNPs‑IgG and UCNPs, respectively, made by one‑pot process
Characterization of UCNPs After MAPLE Deposition
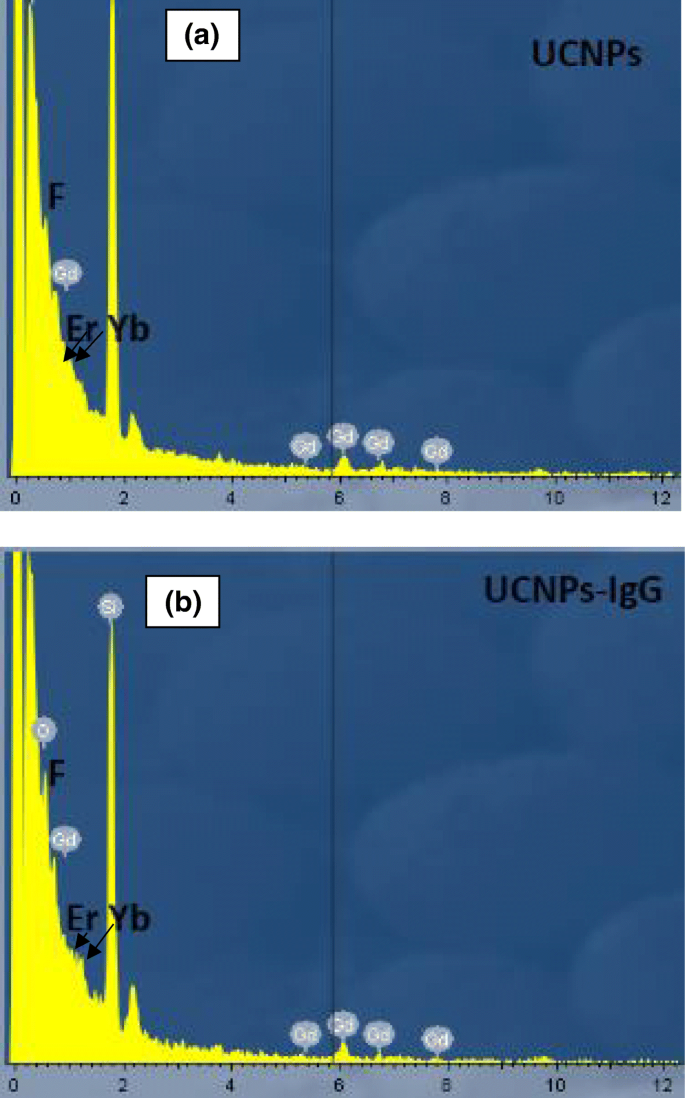
EDX mapping of glass substrates coated with UCNPs and UCNPs‑IgG confirms the presence of Gd, Er, Yb, and F (Fig. 6a,b). PL measurements show retained green (540 nm) and red (650 nm) emissions under 980 nm excitation, with only minor intensity variations attributable to surface defects or measurement error.

EDX spectra of a samples after MAPLE treatment and b samples without MAPLE treatment (bare glass)
FTIR of the coated surfaces further confirms the preservation of functional groups: the –OH band at 3648 cm–1 (IgG) appears only in UCNPs‑IgG (Fig. 7a), while the amine band at 1575 cm–1 is evident in UCNPs (Fig. 7b). The absence of these bands in bare glass (Fig. 7c) underscores the successful deposition of the nanoparticle layers.
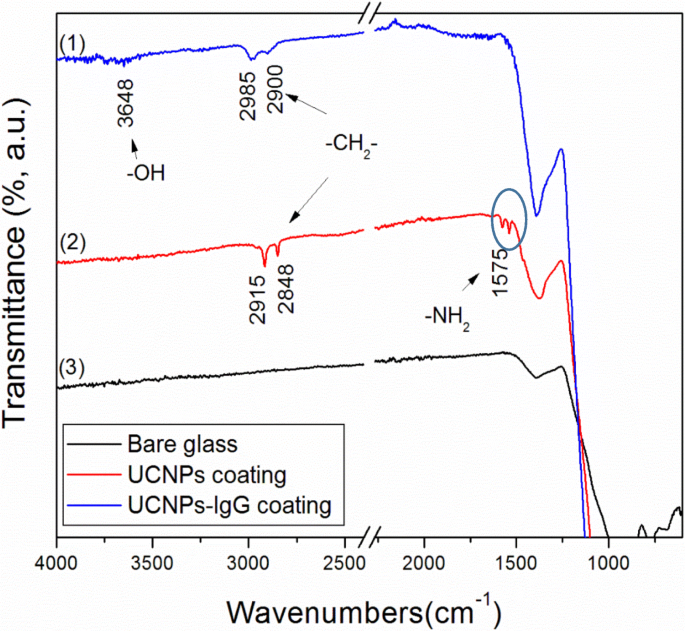
FTIR spectra of bare glass and glass coated with UCNPs‑IgG and UCNPs, respectively
Cell Behaviors on the Different Coatings
Confocal images after 24 h of culture reveal comparable cell numbers across all surfaces, but UCNPs‑IgG surfaces exhibit larger cell spread, longer cytoplasmic extensions, and increased inter‑cellular connectivity (Fig. 8). Quantitative analysis shows 11.2 % and 22.2 % increases in cell area for UCNPs and UCNPs‑IgG, respectively, relative to gelatin controls; connection lengths increase by 12.5 % and 17.5 %; cell lengths grow by 8.2 % and 17.3 %; and cell counts rise by ~8 % on nanoparticle‑coated surfaces (Fig. 9). These metrics confirm that both UCNPs and UCNPs‑IgG enhance endothelial adhesion and spreading without inducing cytotoxicity.
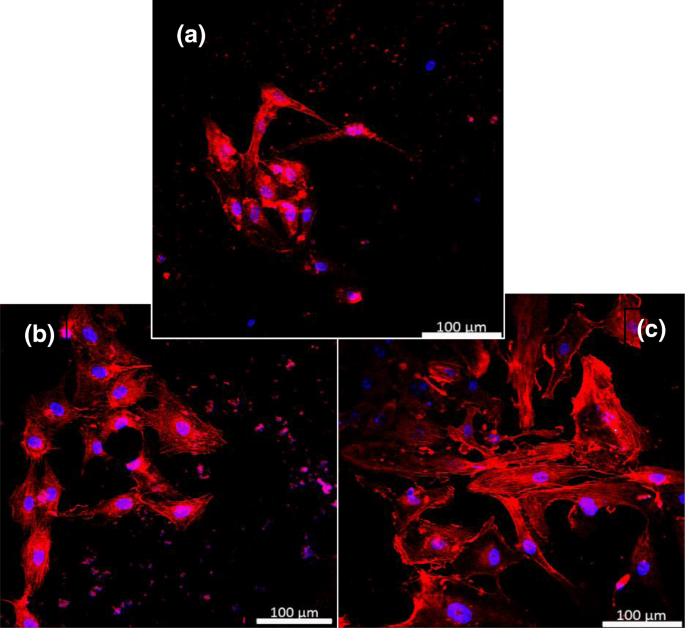
Confocal micrographs of HUVECs on a control, b UCNPs, and c IgG‑modified UCNPs
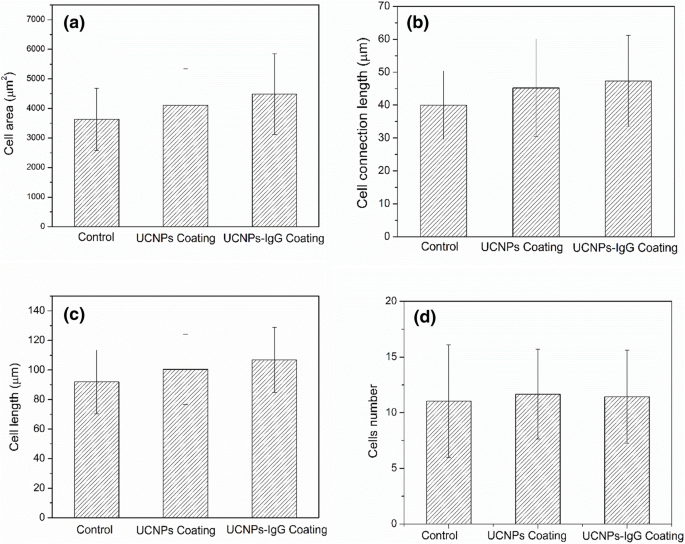
HUVECs cultured on different coatings: a cell area, b connection length, c cell length, d cell count
Effect of the Coating on Cell Viability
MTT assays demonstrate >99 % relative viability on bare glass, UCNPs, and UCNPs‑IgG, exceeding the >85 % benchmark for biocompatible materials. The slight increase in viability (105 % for UCNPs, 104 % for UCNPs‑IgG) suggests a mild stimulatory effect of the nanoparticle coatings, likely due to enhanced surface roughness and protein adsorption.
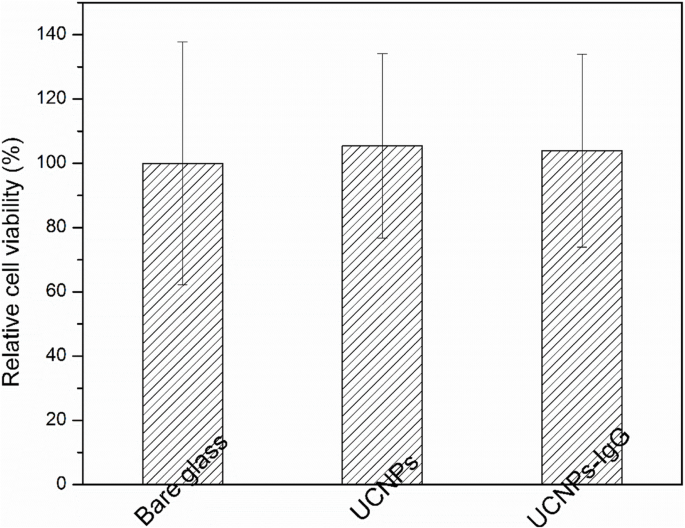
Cell viability of HUVECs on bare glass, UCNPs, UCNPs‑IgG, and gelatin‑coated control
Conclusions
We have demonstrated that NaGdF4:Yb3+,Er3+ UCNPs and their IgG‑functionalized counterparts can be synthesized in a single‑pot hydrothermal process and deposited onto glass culture dishes by MAPLE without compromising their structural or photoluminescent integrity. TEM, FTIR, XRD, and EDX confirm successful synthesis, conjugation, and deposition. Biocompatibility assays reveal no cytotoxicity, while confocal microscopy shows enhanced adhesion, spreading, and inter‑cellular networking of HUVECs on nanoparticle‑coated surfaces. These findings position MAPLE‑deposited, antibody‑functionalized UCNPs as promising, low‑contamination coatings for advanced tissue engineering and regenerative medicine platforms.
Abbreviations
- DAPI:
4′,6‑Diamidine‑2′‑phenylindole dihydrochloride
- EG:
Ethylene glycol
- HUVEC:
Human umbilical vein endothelial cells
- IgG:
Immunoglobulin G
- MAPLE:
Matrix‑assisted pulsed laser evaporation
- PEI:
Polyethylenimine
- Phalloidin‑TRITC:
Phalloidin–Tetramethylrhodamine B isothiocyanate
- UCNPs:
Upconversion nanoparticles
Nanomaterials
- Enhancing Tungsten‑Copper Alloy Performance: Proven Strategies and Processing Techniques
- Glass Fiber Performance: Physical, Mechanical & Chemical Properties Explained
- Visible‑Light‑Assisted Au Nanoparticle‑Modified Glassy Carbon Electrodes for Sensitive Uric Acid Detection
- Optimized Post‑Treatment Strategy for Monodisperse FePt–Fe3O4 Binary Nanoparticles
- Gold Nanoparticle–Modified 6‑Mercaptopurine and Rabies‑Derived Peptide Enhance SH‑SY5Y Neural Cell Proliferation and Neurite Outgrowth
- Lithocholic Acid–Functionalized Gold Nanoparticles Induce Selective Apoptosis in Liver Cancer Cells
- Enhanced Organic–Nanostructured Silicon Hybrid Solar Cells via Surface Modification for Superior Efficiency
- Optimized Mitoxantrone Delivery Using Cholesterol‑Modified Pullulan Nanoparticles: Size‑Dependent Antitumor Efficacy Against Bladder Cancer
- Copper Nanoparticles on Porous Carbon Substrates Deliver High-Performance Glucose Sensors
- Ultrasmall Superparamagnetic Iron Oxide Nanoparticles for Stem Cell Tracking in Parkinson’s Therapy



