Sodium Alginate–Sulfonated Graphene Oxide Membrane: Superior Proton Conductivity and Low Methanol Permeability for Direct Methanol Fuel Cells
Abstract
Direct methanol fuel cells (DMFCs) suffer from high methanol crossover and the expensive Nafion® membrane. We fabricated a non‑Nafion, sodium alginate (SA) based polymer electrolyte membrane reinforced with 0.02–0.20 wt % sulfonated graphene oxide (SGO) via a simple solution‑casting approach. Electrostatic attraction between the –SO3H groups of SGO and the SA matrix enhances mechanical integrity, optimizes water uptake, and suppresses methanol transport. The SA/SGO membrane containing 0.20 wt % SGO achieved a proton conductivity of 13.2 × 10–3 S cm–1 and a methanol permeability of 1.535 × 10–7 cm2 s–1 at 25 °C, far below Nafion’s 25.1 × 10–7 cm2 s–1. Mechanical testing confirmed improved tensile strength and elongation at break with SGO addition.
Background
DMFCs convert chemical energy into electricity via methanol oxidation, offering a clean, portable power source with low pollutant emissions. However, commercialization is limited by the high cost (~$1000 m–2), elevated methanol permeability of Nafion®, and the limited durability of current electrocatalysts.
Proton exchange membranes (PEMs) are critical in DMFCs; they must provide high proton conductivity while blocking methanol crossover. Nafion® fails to meet these dual demands, prompting the search for greener, cost‑effective alternatives.
Biopolymers such as alginates—polysaccharides derived from brown seaweed—offer a promising, sustainable platform. Alginate’s high water uptake (200–300 × its weight) is advantageous for proton transport, yet its intrinsic proton conductivity is low. Composite strategies incorporating inorganic fillers have previously enhanced alginate’s mechanical and transport properties, notably with carbon nanotubes and graphene oxide (GO).
Graphene oxide’s high aspect ratio and mechanical strength make it a compelling filler; however, its lack of proton‑carrier functional groups limits conductivity. Sulfonating GO introduces –SO3H groups that act as proton hopping sites and create stronger electrostatic interactions with the polymer matrix, improving both mechanical stability and proton transport.
While several biopolymer–GO composites have been reported, a SA–SGO nanocomposite membrane has not yet been explored. This study presents the synthesis, characterization, and DMFC performance of such a membrane, aiming to surpass Nafion® in selectivity and cost.
Methods
Materials
Graphite (TIMREX PG25), concentrated H2SO4, KMnO4, HCl, H2O2, NaNO2, glycerol, and other reagents were sourced from Sigma Aldrich. Deionized water (Milli‑Q) was used throughout.
Membrane Preparation
GO was produced by a modified Hummer’s method. Subsequent sulfonation involved refluxing GO with sulfanilic acid and NaNO2 at 70 °C, yielding SGO with ~1.76 wt % S (verified by XPS). SA was dissolved in 1 % w/v water; SGO (0.02–0.20 wt %) was dispersed into the SA solution and stirred 60 min. The mixture was cast on glass, dried at 60 °C for 72 h, then cross‑linked in 1.5 % w/v CaCl2/glycerol for 30 min. Final membranes were rinsed, dried at 25 °C, and stored in a desiccator (Scheme 1).
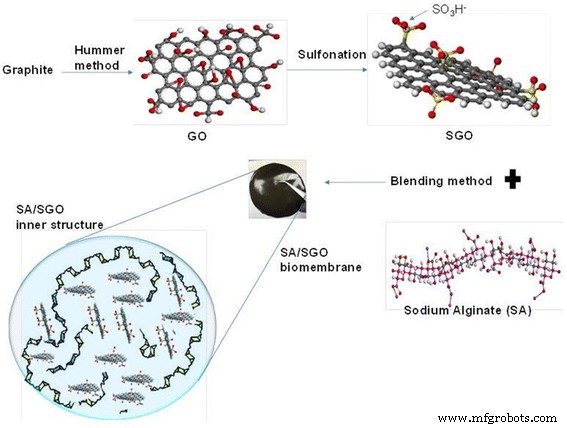
Sulfonated graphene oxide (SGO) filler and SA/SGO biomembrane preparation method
Characterization
FTIR (PerkinElmer) measured 4000–500 cm–1; FESEM (FEI QUANTA 400) examined morphology; HRTEM (HT7700) resolved nanosheets; XPS (Axis Ultra DLD) quantified surface composition. Mechanical properties were measured on a Universal Testing Machine (3 kN load). Water and methanol uptake were calculated from mass changes (Eq. 1) and dimensional changes (Eq. 2). Proton conductivity was determined using a four‑electrode cell (WonATech) under full hydration; resistance (R) was extracted from linear V–I curves (Eq. 3). Methanol permeability employed a two‑tank cell with 20 % v/v methanol, applying Eq. 4. Selectivity (φ) followed Eq. 5.
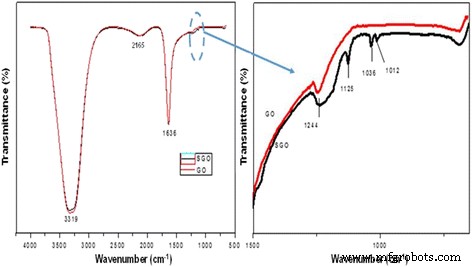
a, b FTIR spectra for graphene oxide (GO) and sulfonated graphene oxide (SGO)
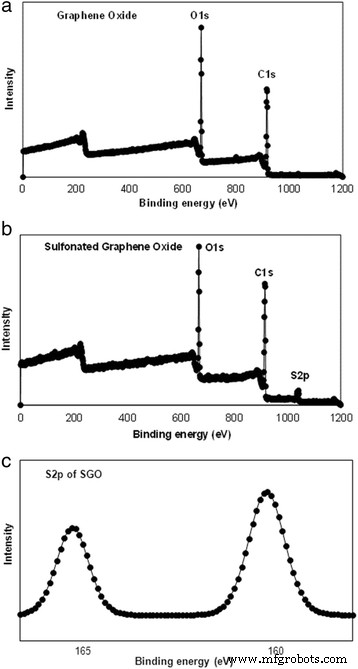
XPS of a, b wide spectra GO and SGO and c S2p spectra of SGO

a FESEM image of GO. b, c FESEM images of SGO with various magnification and d EDX of SGO
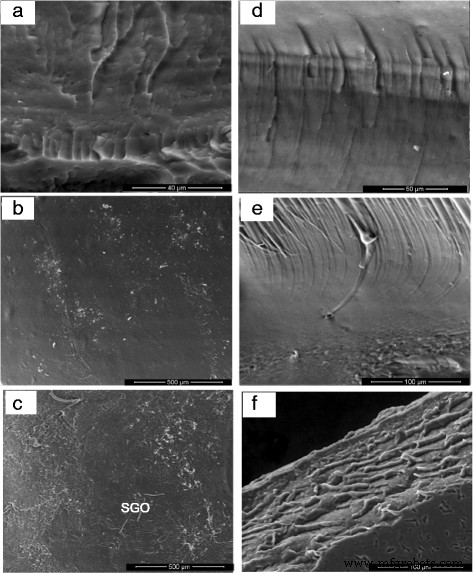
FESEM images of surface morphology and cross‑section for a, d sodium alginate, b, e SA/SGO4, and c, f SA/SGO6 biomembranes

TEM image of SGO nanosheets distributed in sodium alginate polymer matrix
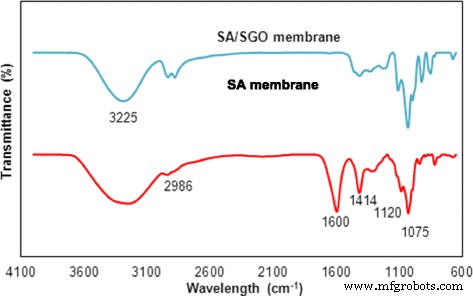
FTIR spectra of SA and SA/SGO membrane
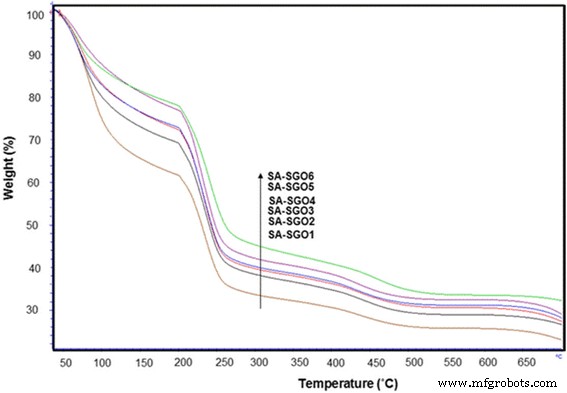
TGA curve for SA/SGO biomembranes with various SGO wt%
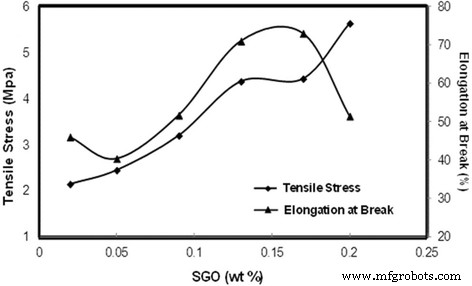
Tensile stress and elongation at break of biomembrane with various SGO wt%
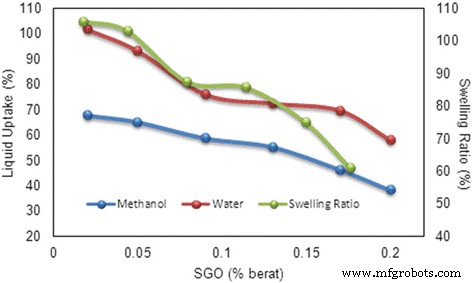
Liquid uptake and swelling ratio of SA/SGO membrane with wt.% of SGO
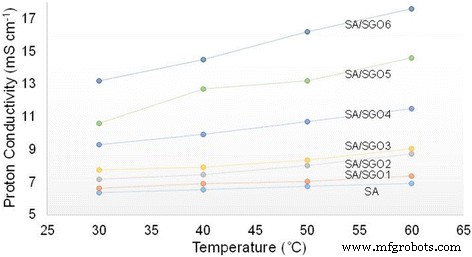
Proton conductivity of SA/SGO biomembranes with various content of SGO at different temperature
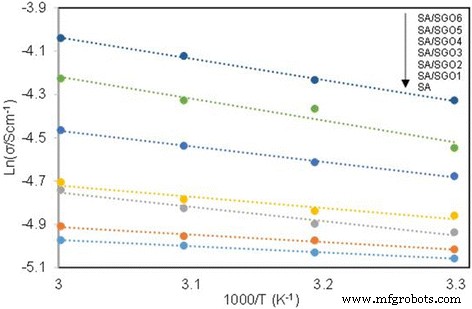
ln σ vs. 1000/T plot for the cross‑linked QAPVA membranes, the lines indicate the linear regression
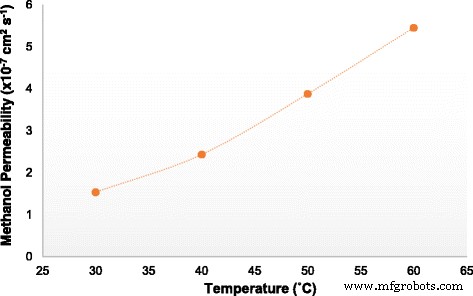
Methanol permeability of membrane SA/SGO6 vs. temperature
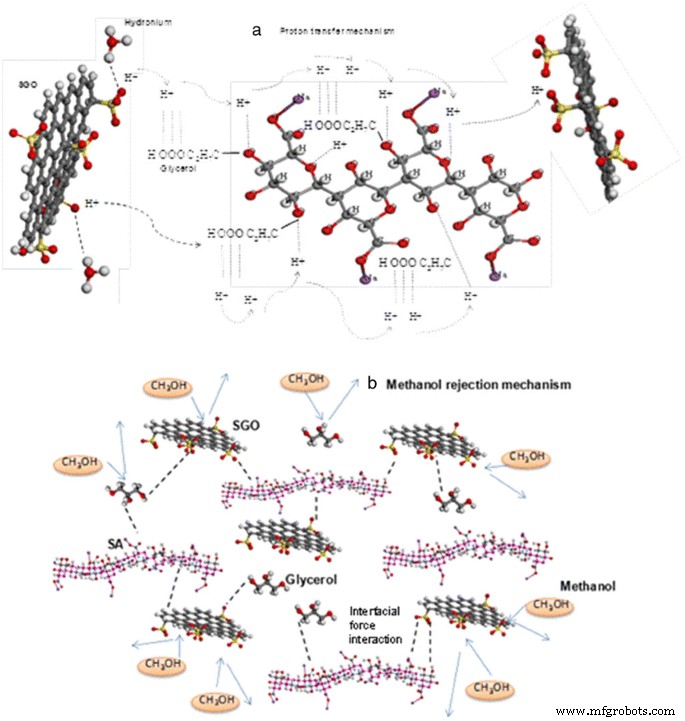
Suggested mechanism of a proton mobility and b methanol rejection
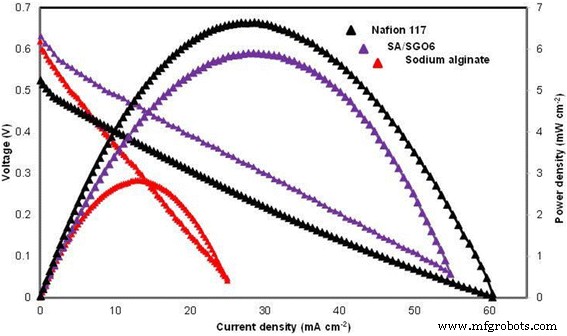
Single‑cell performance test for sodium alginate, SA/SGO6, and Nafion 117 (4 M methanol and 25 °C temperature, passive mode)
Conclusions
We successfully fabricated a SA/SGO composite membrane exhibiting low methanol permeability (1.535 × 10–7 cm2 s–1), high proton conductivity (13.2 × 10–3 S cm–1), and enhanced mechanical robustness. The –SO3H groups on SGO promote strong electrostatic coupling with SA, yielding a dense, interconnected network that blocks methanol while sustaining proton transport. The resulting selectivity (8.56 × 104 S s cm–3) surpasses that of Nafion 117, positioning SA/SGO as a viable, greener alternative for DMFCs.
Abbreviations
- BC:
Bacterial cellulose
- CNC:
Cellulose nanocrystal
- CNFs:
Cellulose nanofibers
- CNT:
Carbon nanotube
- DI:
Deionized
- DLFC:
Direct liquid fuel cell
- DMFC:
Direct methanol fuel cell
- EDX:
Energy dispersive X‑ray
- FESEM:
Field emission scanning electron microscope
- FTIR:
Fourier transform infrared
- GO:
Graphene oxide
- GOS:
Graphene oxide sheet
- HRTEM:
High‑resolution transmission electron microscopy
- IEC:
Ion exchange capacity
- LC:
Liquid capacity
- OCV:
Open circuit voltage
- P:
Membrane diffusion permeability for methanol
- PEMFC:
Polymer electrolyte membrane fuel cell
- PEMs:
Proton exchange membrane
- PMA:
Phospho molybdic acid
- PSSA:
Poly‑styrene sulfonic acid
- PVA:
Poly vinyl alcohol
- PVP:
Poly (vinyl pyrrolidone)
- R:
Resistance of the membrane
- RGO:
Reduced graphene oxide
- SA:
Sodium alginate
- SA/SGO:
Sodium alginate/sulfonated graphene oxide membrane
- SGO:
Sulfonated graphene oxide
- SHNT:
Sulfonated halloysite nanotube
- SPSF:
Sulfonated polysulfone
- SW%:
Swelling ratio percentage
- T:
Membrane thickness
- TGA:
Thermal gravimetric analysis
- W:
Width of the membrane
- WU%:
Water uptake percentage
- XPS:
X‑ray photoelectron spectroscopy
Nanomaterials
- Titanate Nanotube-Enhanced Graphene Oxide Nanocomposites: Boosting Flame Retardancy and Photodegradation in Flexible PVC
- Assessing the Biosafety and Antibacterial Efficacy of Graphene and Graphene Oxide for Orthopedic Implant Applications
- Easily Synthesized Wormhole‑Like Mesoporous SnO₂ via Evaporation‑Induced Self‑Assembly: Superior Ethanol Gas‑Sensor Performance
- Hybrid Graphene/WO₃ and Graphene/CeOx Electrodes for High‑Performance Supercapacitors
- Spherical Graphene/SiO₂ Supports Enable Uniform In Situ Polymerization of UHMWPE for High‑Performance Nanocomposites
- Titania‑Coated Silica Enhanced with Sodium Alginate: A Superior Sorbent for Cu(II), Zn(II), Cd(II), and Pb(II) Removal
- Low-Temperature Annealing of Graphene Oxide Films: Impact on Electrical Conductivity and Surface Potential via SKPFM
- Graphene‑Sheet‑Driven Nanopump: Harnessing Brownian and Directed Motion to Generate Net Water Flux through Carbon Nanotube Channels
- Nanoalginate Carriers via Inverse‑Micelle Synthesis: Doxorubicin Encapsulation and Cytotoxicity in Murine Breast Cancer Cells
- Quaternized PVA/Graphene Oxide Composite Membrane Enhances Ethanol Barrier and Ionic Conductivity for Passive Alkaline DEFCs



