Synergistic Graphene-Modified TiO₂ Composites Deliver Superior Photocatalytic Activity
Abstract
Three‑dimensional graphene networks (3DGN) offer a continuous, high‑quality scaffold that can enhance TiO₂ photocatalysts. However, the limited defect density of 3DGN hampers both intimate contact with TiO₂ nanoparticles—critical for efficient electron transfer—and the adsorption of pollutant molecules. In contrast, reduced graphene oxide (RGO) nanosheets possess abundant surface functional groups that serve as active adsorption sites and facilitate charge transfer. By co‑incorporating RGO and 3DGN onto TiO₂, we achieved a synergistic composite that markedly boosts chemical adsorption and generates higher yields of reactive oxidants. Optimizing the RGO loading fraction significantly improves the overall photocatalytic performance, as demonstrated in phenol degradation studies.
Background
Graphene’s exceptional electrical conductivity and expansive BET surface area make it an attractive modifier for TiO₂ photocatalysts [1]. Yet, conventional RGO nanosheets exhibit high defect densities and a discontinuous morphology, limiting their theoretical performance potential. Recent advances in three‑dimensional graphene networks (3DGN) have shifted focus toward their naturally continuous, high‑quality structure, which improves both electron transport and nanoparticle loading capabilities [2, 3].
Our prior work revealed that an optimal defect density in 3DGN facilitates chemisorption of pollutants and tight coupling with TiO₂ nanoparticles, creating efficient electron transport pathways [2]. Controlling defect density during chemical vapor deposition is technically demanding, whereas the surface chemistry of RGO can be tuned more readily [4, 5]. Thus, integrating RGO nanosheets with 3DGN offers a promising route to enhance photocatalytic activity.
In this study, we synthesized RGO–3DGN–TiO₂ composite photocatalysts, optimized their composition, and evaluated key performance metrics—chemical adsorbability, electron transport, and phenol decomposition kinetics. We employed photoluminescence (PL), infrared (IR) spectroscopy, and electron paramagnetic resonance (EPR) to elucidate the synergistic interactions between 3DGN and RGO.
Methods
The preparation protocols and degradation experiments follow our previous reports [2, 5, 6]. Briefly, nickel foam seeded with 3DGN was immersed in 50 mL of 25 wt % aqueous ammonia containing 50 mg TiO₂–RGO mixtures (RGO mass fraction 1–8 wt %) at room temperature. The suspension was transferred to an autoclave and heated to 110 °C for 10 h in a vacuum oven, then cooled. The resulting composites were washed with deionized water and dried at 80 °C under vacuum for 2 h before use.
Results and Discussion
SEM images of pristine TiO₂, 3DGN–TiO₂, and RGO–3DGN–TiO₂ are shown in Figure 1. The 3DGN displays characteristic surface wrinkles caused by differential thermal expansion between graphene and the Ni substrate, which enhance both pollutant adsorption and TiO₂ loading. The average TiO₂ particle size on the composite (10–50 nm) confirms that the high BET area of 3DGN mitigates agglomeration [1, 2]. Raman analysis of the RGO component shows a low I_D/I_G ratio of 0.29, indicating minimal defects (Figure 1d) [7]. The adopted 3DGN also exhibits a modest D peak, reflecting its engineered defect density [6].
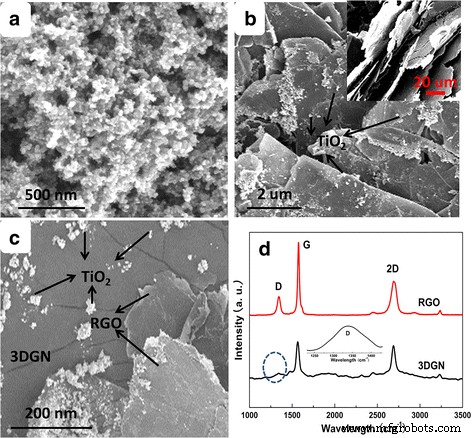
Figure 1. SEM images of (a) pure TiO₂, (b) 3DGN–TiO₂ (inset: pristine 3DGN), (c) RGO–3DGN–TiO₂, and (d) Raman spectra of RGO and 3DGN. The D peak of 3DGN is magnified.
Photocatalytic performance was evaluated via phenol degradation under UV and visible light. The RGO–3DGN–TiO₂ composite achieved a decomposition rate constant of 1.33 × 10⁻² min⁻¹ under UV, outperforming pure TiO₂, RGO–TiO₂, and 3DGN–TiO₂ by 180 %, 70 %, and 40 % respectively (Figure 2a). Under visible illumination, the composite also exhibited superior activity (Figure 2b). Key determinants include electron utilization efficiency and pollutant chemisorption capacity. While 3DGN offers a large BET area and high‑quality graphene to promote charge separation, the absence of sufficient interfacial contact limits its effectiveness. In contrast, RGO’s functional groups provide abundant chemisorption sites and bridge the graphene–TiO₂ interface, as evidenced by IR spectra (Figure 3a). The Ti–O–C vibration at ~800 cm⁻¹ confirms enhanced bonding after RGO addition [2, 5].
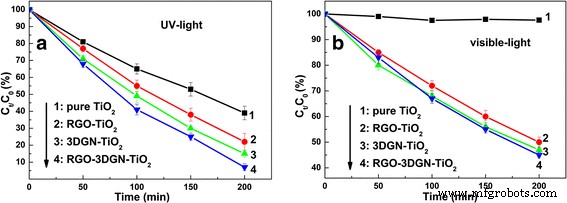
Figure 2. Phenol degradation kinetics under (a) UV and (b) visible light irradiation.
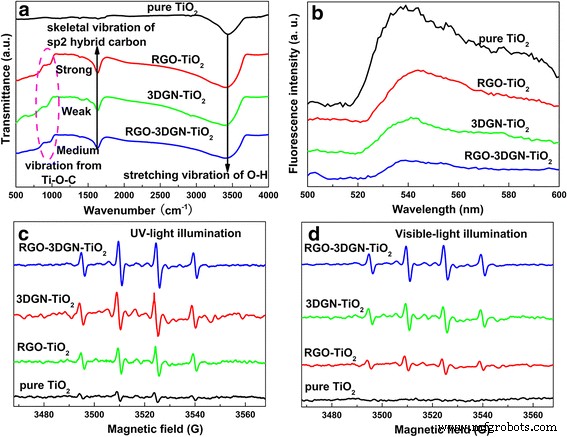
Figure 3. Characterization of composites: (a) IR spectra, (b) PL spectra, (c) EPR spectra (UV), and (d) EPR spectra (visible light).
PL measurements revealed reduced radiative recombination in the composites, with the RGO–3DGN–TiO₂ exhibiting the weakest PL signal—indicating the highest electron utilization efficiency (Figure 3b). EPR spectra confirmed higher yields of ·OH and O₂⁻ radicals for the composite under UV, underscoring enhanced electron transfer across the graphene–TiO₂ interface (Figure 3c). Visible‑light EPR signals were comparable between 3DGN–TiO₂ and the composite, reflecting the limited benefit of additional RGO under these conditions due to uncontrolled sheet thickness affecting quantum tunneling probabilities [5].
TGA analysis (Figure 4) highlighted distinct weight loss stages: 100–180 °C (adsorbed water) for 3DGN–TiO₂, and an additional 250–350 °C loss for RGO‑containing samples, attributable to removal of residual functional groups [5].
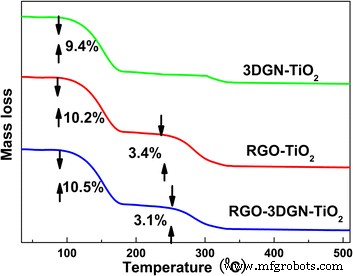
Figure 4. TGA curves of 3DGN–TiO₂, RGO–TiO₂, and RGO–3DGN–TiO₂.
Conclusions
Co‑modification of TiO₂ with both RGO nanosheets and 3DGN yields a composite that synergistically enhances photocatalytic performance. Although RGO’s discontinuity and higher defect density can shorten electron lifetimes, its surface functional groups significantly improve pollutant chemisorption and electron transfer across the graphene–TiO₂ interface, thereby mitigating the need for complex defect engineering of 3DGN. The optimized composite achieved a phenol degradation rate constant of 1.33 × 10⁻² min⁻¹ under UV light—substantially higher than that of the individual RGO–TiO₂ or 3DGN–TiO₂ systems.
Nanomaterials
- High‑Efficiency Graphene Solar Cells: 9% Power Conversion with TFSA Doping
- S,N‑Co‑Doped Graphene Quantum Dot/TiO₂ Composites for High‑Efficiency Visible‑Light Photocatalytic Hydrogen Generation
- Hollow‑Structured LiNb3O8 Photocatalysts: Synthesis, Characterization, and Superior Methylene Blue Degradation
- High‑Performance Na4Mn9O18/Carbon Nanotube Cathodes for Aqueous Sodium‑Ion Batteries
- Influence of Peptizing Acid on TiO₂ Phase Composition and Photocatalytic Efficiency: A Comparative Study of Sulfuric, Nitric, and Acetic Acids
- Enhanced Organic–Nanostructured Silicon Hybrid Solar Cells via Surface Modification for Superior Efficiency
- Enhanced Gas Separation Using Covalently Functionalized Graphene Oxide in PIM‑1 Mixed‑Matrix Thin‑Film Composite Membranes
- Enhanced Photocatalytic Degradation of Methyl Orange Using CdTe Quantum Dot/BiOI‑Coated TiO₂ Hollow Microspheres under Simulated Sunlight
- FeF3·0.33H2O Cathode Enhanced by CNTs and Graphene: A High‑Performance Solution for Lithium‑Ion Batteries
- High‑Efficiency Transparent PAN/TiO₂ Nanofiber Membranes for Advanced Particulate Matter Filtration



