Biomedical Applications of Gold Nanoclusters: Recent Advances and Future Directions
Abstract
Gold nanoclusters (AuNCs) have emerged as a versatile class of fluorescent probes, owing to their atom‑scale size, tunable emission, and remarkable chemical stability. Their inherent water solubility, low cytotoxicity, and exceptional photostability make AuNCs ideal for long‑term cellular imaging, ultrasensitive detection, and targeted therapeutics. This review surveys recent advances in AuNCs synthesized with small‑molecule, polymeric, and biomacromolecular ligands, highlighting breakthroughs in imaging, sensing, and therapy. We also discuss current limitations—such as sub‑optimal quantum yields and ligand heterogeneity—and outline future research directions that promise to unlock the full biomedical potential of AuNCs.
Background
Nanomaterials are reshaping biomedical research by offering unprecedented control over size, shape, and surface chemistry [1–10]. Compared with bulk counterparts, AuNCs—composed of a few to a few hundred gold atoms—exhibit unique optical signatures, including bright fluorescence across the visible to near‑infrared (NIR) window, long lifetimes, and large Stokes shifts [35–37]. These attributes, combined with facile surface functionalization, position AuNCs as superior alternatives to organic dyes and semiconductor quantum dots for cellular imaging, molecular sensing, and photodynamic therapy [41–49]. Recent years have seen an explosion of work exploring AuNCs conjugated to diverse ligands, enabling tailored physicochemical properties and enhanced biocompatibility. While several comprehensive reviews have addressed analytical applications of AuNCs [50–53], this paper focuses on their biomedical utility and the strategic design of ligand architectures that drive performance in imaging, detection, and therapy.
We examine three ligand classes—small molecules, polymers, and biomacromolecules—whose distinct chemistries afford AuNCs with specific targeting capabilities, improved quantum yields, and multifunctionality. The challenges and future opportunities that emerge from these studies are summarized in the concluding section.
Small Molecule-Conjugated AuNCs
Small‑molecule ligands are the most common choice for AuNC synthesis because they provide precise control over size and surface chemistry. For instance, D‑penicillamine (DPA)–coated AuNCs (DPA‑AuNCs) are exceptionally bright, water‑soluble, and readily internalized by HeLa cells, enabling two‑photon confocal imaging after just 2 h of incubation [54,55]. The resulting 3‑D reconstructions demonstrate uniform intracellular distribution and high photostability (Figure 1).
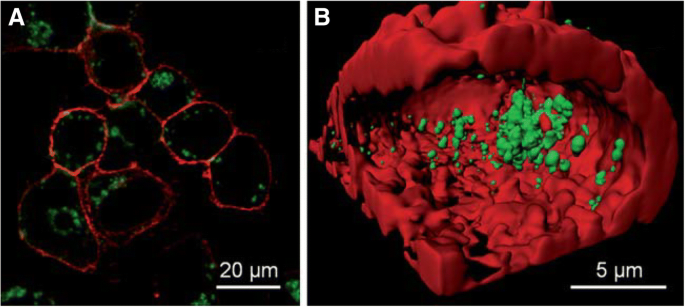
a Confocal image of HeLa cells after 2 h incubation with DPA‑AuNCs (green). b 3‑D reconstruction showing uniform internalization [55].
Dihydrolipoic acid (DHLA)–capped AuNCs (DHLA‑AuNCs) were the first nanoprobes to enable fluorescence lifetime imaging (FLIM) in living cells. While untreated HeLa cells display short lifetimes (1.5–4 ns), DHLA‑AuNCs confer long lifetimes (500–800 ns), facilitating FLIM‑based subcellular mapping (Figure 2) [56].
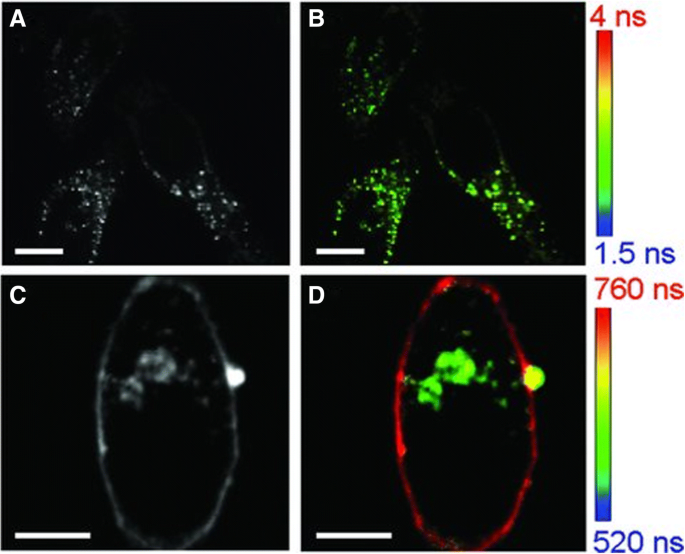
Intensity (a, c) and FLIM (b, d) images of HeLa cells before (a, b) and after (c, d) DHLA‑AuNC incubation. Scale bars = 10 µm [56].
Cell‑line‑dependent biosynthesis of AuNCs has also been demonstrated: HepG2 and K562 cells spontaneously convert chloroauric acid into AuNCs, whereas non‑cancerous L02 cells do not, suggesting a potential tumor‑selective imaging strategy [57]. Trypsin‑stabilized AuNCs (try‑AuNCs) exhibit NIR emission and can simultaneously act as a surface‑plasmon‑enhanced energy‑transfer (SPEET) sensor for heparin and as a folic‑acid‑targeted imaging probe for cancer cells (Figure 3) [58].
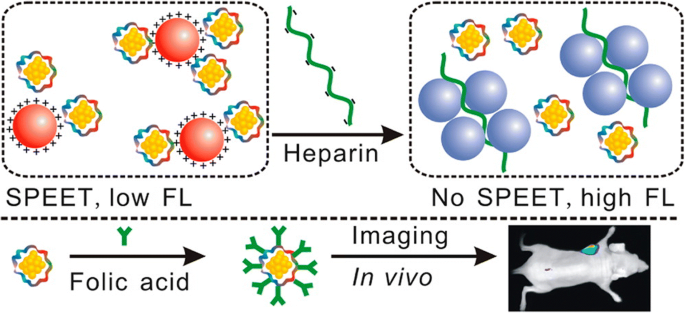
try‑AuNCs: dual‑function NIR biosensor and in vivo cancer imaging bioprobe [58].
Ligand chemistry further expands sensing capabilities. AuNCs bearing dopaquinone can complex Fe3+ ions, leading to >500 nm aggregates and a pronounced fluorescence quench, enabling Fe3+ detection in aqueous media [61]. 11‑Mercaptoundecanoic acid (MUA)–AuNCs (MUA‑AuNCs) form stable complexes with Hg2+ and bithiols, providing selective fluorescence‑based sensing of mercury and related thiols [64–65]. Vancomycin‑stabilized AuNCs (Van‑AuNCs) were applied to monitor Fe3+ in diverse water samples, demonstrating environmental relevance [66]. Chitosan‑functionalized AuNCs (AuNCs@Chi) served as a FRET‑based hydrogen sulfide sensor, capitalizing on H2S’s role in vasodilation and neurotransmission [67].
Glutathione (GSH)–capped AuNCs (GSH‑AuNCs) have been engineered for rapid, selective detection of lysine and cysteine with high photostability [73]. FRET assemblies using GSH‑AuNCs achieve ultrasensitive cysteine sensing, potentially aiding diagnosis of cysteine‑related disorders [74]. Keratin‑templated AuNCs, exploiting cysteine‑rich proteins, detect Hg2+ via dual‑emission carbon‑dot/gold‑cluster probes with ratiometric response [76]. Cyclodextrin‑capped AuNCs selectively sense Co2+ ions, with cellular uptake confirmed in vitro [77].
Most recently, ultrasmall GSH‑AuNCs have been developed as metabolizable radiosensitizers. Their small size (~2 nm) and high X‑ray attenuation enable enhanced radiotherapy through photoelectric and Compton interactions, while GSH coatings ensure tumor accumulation via the EPR effect and renal clearance post‑treatment, minimizing off‑target toxicity [78].
Polymer-Conjugated AuNCs
Polymeric ligands impart additional functionality to AuNCs, including improved colloidal stability, tunable surface charge, and biocompatibility. Multidentate thioether‑terminated poly(methacrylic acid) (PTMP‑PMAA)–capped AuNCs exhibit exceptional photostability and selectively label cord blood mononuclear cells (CBMCs) versus K562 leukemia cells, reflecting differential uptake (Figure 5) [79–80]. Such selective internalization underscores the potential of polymer‑coated AuNCs for targeted hematopoietic imaging and drug delivery.
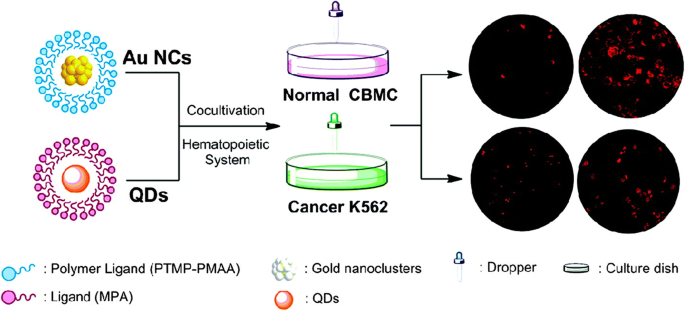
Labeling of normal (CBMCs) and cancer (K562) cells with PTMP‑PMAA‑AuNCs versus quantum dots [79].
Aldek et al. introduced PEG‑ and zwitterion‑functionalized AuNCs via lipoic‑acid bidentate ligands, demonstrating robust pH stability and salt tolerance—key attributes for biological imaging and sensing [82]. Chen et al. designed a pH‑responsive amphiphilic copolymer (PDNH) encapsulating AuNCs, yielding a nanocomposite (L‑AuNCs/FA‑PDNH) that combines imaging of folate‑overexpressing cancers with paclitaxel delivery (Figure 6) [88].
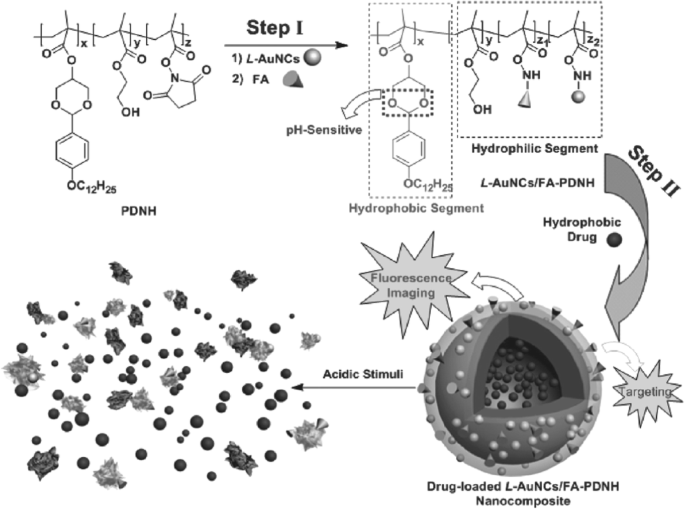
Fabrication of L‑AuNCs/FA‑PDNH nanocomposite for dual imaging and paclitaxel therapy [88].
Polyethyleneimine (PEI)‑stabilized AuNCs (PEI‑AuNCs) provide cationic surface charge, facilitating gene delivery and strong red fluorescence in HepG2 cells, thereby offering a dual imaging/therapeutic platform [89]. FA‑Ova‑AuNCs, assembled via an N‑acryloxysuccinimide linker, selectively stain folate‑receptor‑positive HeLa cells, confirming receptor‑mediated uptake [90].
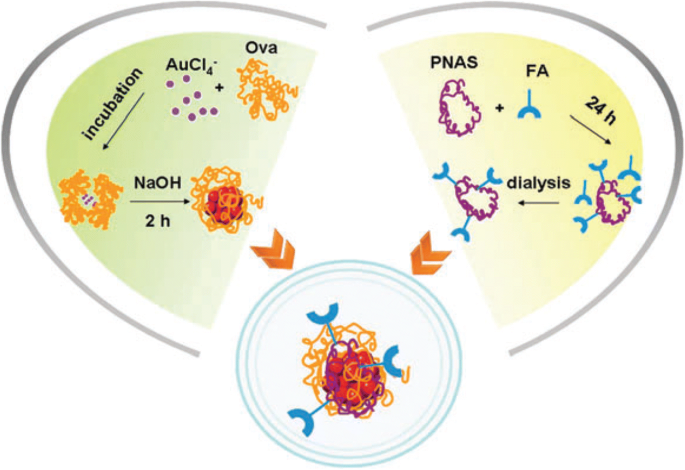
Scheme for FA‑Ova‑AuNC synthesis and HeLa cell imaging [90].
A highly phosphorescent Au(I) cluster embedded in a macroporous polymer film provides a colorimetric cyanide sensor, capable of detecting CN− in food and environmental samples with high selectivity and cost‑effectiveness—critical for public health monitoring [91–93].
Biomacromolecule-Conjugated AuNCs
Biomacromolecular ligands, rich in thiol groups, offer biocompatibility and specific recognition. Transferrin (Tf)–functionalized AuNCs combined with graphene oxide (Tf‑AuNCs/GO) constitute a turn‑on NIR probe that selectively illuminates Tf receptor‑overexpressing HeLa cells while sparing normal 3T3 fibroblasts (Figure 8) [94]. This selective uptake demonstrates the probe’s potential for targeted imaging of iron‑metabolism‑driven cancers.
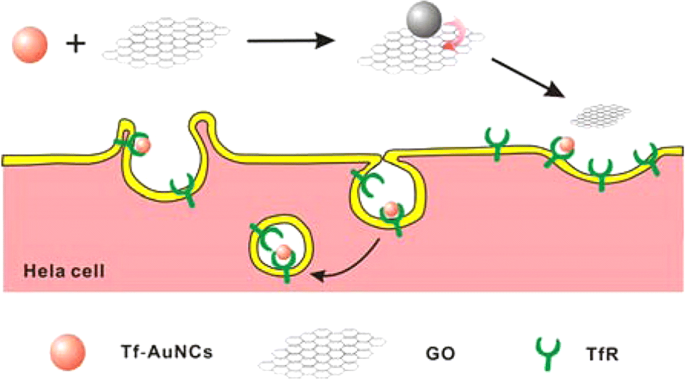
Tf‑AuNCs/GO probe for imaging TfR‑positive HeLa cells after 4 h incubation [94].
DNA‑templated AuNCs offer a rapid (2‑min) green synthesis that yields luminescent nanoclusters whose intensity scales linearly with DNA concentration, enabling real‑time DNA quantification in HeLa and A549 cells [95]. The probe’s low toxicity and tunable emission across all four DNA bases provide a versatile platform for nucleic‑acid analysis.
Nguyen et al. constructed a dual‑ligand AuNC/graphene nanocomplex that acts as a “turn‑on” probe for matrix metalloproteinase‑9 (MMP‑9). A peptide bearing the MMP‑9 cleavage site serves as both stabilizer and targeting ligand, achieving a detection limit of 0.15 nM and selective imaging of secreted MMP‑9 from MCF‑7 cells [97].
Song et al. developed a label‑free, green fluorescence assay for protein kinases by exploiting Zr‑induced aggregation of phosphorylated‑AuNC‑peptide complexes, offering high sensitivity and simplified experimental workflow for CK2 activity measurement [98].
Selvaprakash et al. leveraged inexpensive chicken egg‑white proteins (AuNCs@ew) to produce fluorescent probes for ATP and PPi detection via a microwave‑assisted, single‑step synthesis. The resulting AuNCs@ovalbumin dominate the optical response, enabling rapid, cost‑effective phosphate sensing in biological and environmental matrices [99].
Wu et al. synthesized BSA/GSH‑AuNCs with excitation at 330 nm and emission at 650 nm, demonstrating efficient NO2− quenching at pH 3.0 while maintaining high salt tolerance—ideal for complex sample analysis [100]. Concurrent studies revealed that BSA‑coated AuNCs undergo reversible conformational changes upon pH shifts, offering insights into ligand–nanocluster interactions [101].
Ghosh et al. investigated the impact of AuNCs on the activity of α‑chymotrypsin, finding that AuNC binding inactivates the enzyme, but GSH or oxidized GSH can partially restore activity—an observation that may inform targeted enzyme modulation in cancer cells with elevated glutathione levels [102].
Finally, fluorescence‑guided surgery has advanced with AuNCs conjugated to diatrizoic acid and the AS1411 aptamer, enabling dual CT/NIR imaging and intra‑operative tumor delineation in a CL1‑5 mouse model. The high photostability and tumor‑specific targeting of these probes provide a promising platform for real‑time surgical guidance [103].
Conclusions
In summary, the reviewed literature underscores the versatility of AuNCs as fluorescent probes across imaging, sensing, and therapeutic domains. Key challenges remain: (1) achieving quantum yields above 20 % to enable multiplexed imaging; (2) ensuring size uniformity to narrow emission bandwidths; and (3) precisely controlling ligand shell chemistry to tailor surface reactivity and biodistribution. Addressing these hurdles through rational ligand design, high‑throughput synthesis, and in‑depth mechanistic studies will accelerate the translation of AuNCs into clinical diagnostics and personalized medicine.
With continued interdisciplinary efforts, AuNCs are poised to become a cornerstone of next‑generation biomedical imaging and targeted therapy.
Abbreviations
- AuNCs:
Gold nanoclusters
- BSA:
Bovine serum albumin
- CBMC:
Cord blood mononuclear cells
- CD:
Circular dichroism
- Chi:
Chitosan
- ChT:
Chymotrypsin
- DHLA:
Dihydrolipoic acid
- DPA:
D‑penicillamine
- DTT:
Dithiothreitol
- FA:
Folic acid
- FLIM:
Fluorescence lifetime imaging
- FRET:
Förster resonance energy transfer
- GSH:
Glutathione
- GST:
Glutathione‑S‑transferase
- MUA:
11‑Mercaptoundecanoic acid
- Ova:
Ovalbumin
- PCR:
Polymerase chain reaction
- PDNH:
Poly(DBAM‑co‑NASco‑HEMA)
- PEI:
Polyethyleneimine
- PPi:
Pyrophosphate
- PTMP‑PMAA:
Multidentate thioether‑terminated poly(methacrylic acid)
- SPEET:
Surface plasmon‑enhanced energy transfer
- Tf:
Transferrin
- Try:
Trypsin
- VAN:
Vancomycin
Nanomaterials
- Future Directions of 3D Printing in Biomedical Applications
- Gold Nanoparticles: Advancing Diagnostic and Therapeutic Applications in Medicine – A Comprehensive Review
- Fluorescent Nanomaterials: Advances, Synthesis, and Biomedical Applications
- Graphene‑Polymer Composites for High‑Performance Supercapacitors: A Comprehensive Review
- Spherical Gold–Cockle Shell Calcium Carbonate Nanoparticles: Fabrication, Characterization, and Cytotoxicity for Biomedical Use
- Advances in Synthesis and Applications of Silver Nanostructures
- Neoglycoprotein‑Functionalized Fluorescent Gold Nanoclusters for Plant Lectin Detection and Dendritic Cell Imaging
- Magnetic Core‑Shell Nanoparticles: Advancing Targeted Drug Delivery, Imaging, and Antimicrobial Therapies
- Advances in Carbon Nanotube Assembly and Integration for Next‑Generation Applications
- Variable Speed Drives: Keeping Food, Medicine & More Cool in Industrial Refrigeration



