Boron Carbon Oxynitride: A Novel Metal‑Free Photocatalyst with Enhanced Solar‑Driven Hydrogen Production and Dye Degradation
Abstract
Boron‑based nanomaterials are rapidly emerging as non‑toxic, earth‑abundant photocatalysts for solar energy conversion and environmental remediation. Boron carbon oxynitride (BCNO) is a quaternary semiconductor whose electronic, optical, and physicochemical properties can be finely tuned by adjusting the B, C, N, and O composition. Yet, the link between BCNO’s structure and its photocatalytic performance has not been systematically explored. In this work, we performed an in‑depth spectroscopic investigation of BCNO nanostructures prepared with two distinct nitrogen precursors (guanidine hydrochloride and melamine) and at two annealing temperatures (600 °C and 800 °C). We obtained quasi‑spherical BCNO nanodisks (average diameter = 6.7 ± 1.1 nm) using guanidine hydrochloride at 800 °C, with a surface composition of B = 40.6 %, C = 7.95 %, N = 37.7 %, and O = 13.8 % (XPS). Solid‑state ^11B NMR revealed abundant tricoordinate BN_x(OH)_3−x species that act as active sites. In contrast, BCNO annealed at 600 °C with melamine formed multilayered graphene‑oxide sheets rich in carbon (75 % by weight), exhibiting superior photocatalytic activity that surpassed graphitic carbon nitride. The BCNO nanodisks showed a band gap of 5.7 eV, while the graphene‑oxide sheets displayed a 4.2 eV band gap. Time‑resolved photoluminescence lifetimes ranged from 1.58 µs to 8.14 µs, indicating long‑lived charge carriers that favor photocatalysis. These findings establish BCNO as a promising, metal‑free photocatalyst and provide the first structural insight into its activity mechanisms.
Graphical Abstract
Introduction
Metal‑free nanomaterials are increasingly recognized as cost‑effective, environmentally benign photocatalysts with exceptional stability, making them attractive for solar fuel generation, CO₂ reduction, microbial inactivation, and selective organic synthesis. Compared with metal‑containing catalysts, they resist poisoning and exhibit longer cycle lifetimes. Carbon‑based systems such as graphitic carbon nitride, carbon dots, and graphene derivatives have been widely studied, but boron‑based photocatalysts have recently shown remarkable performance, including boron carbide, boron‑doped hexagonal boron nitride (BCN), boron oxynitride, and boron phosphide. Boron carbon oxynitride (BCNO) was first reported as a semiconductor with a tunable band gap (0–5.9 eV) by incorporating B, C, N, and O into a graphene or h‑BN lattice. Despite its promising properties, BCNO’s structure‑activity relationship remains unclear. This study addresses that gap by examining how nitrogen precursor choice, annealing temperature, and time influence BCNO’s structure and photocatalytic activity.
Methods
Chemicals and Instrumentation
Boric acid, melamine, hexamethylenetetramine, and guanidine hydrochloride were purchased from Alfa Aesar and used as‑is. BCNO synthesis followed a low‑temperature annealing protocol: precursors were dissolved in water, heated to 90 °C, dried, ground, and calcined under ambient pressure at 600–800 °C (5 °C min⁻¹ ramp). Analytical techniques included TEM (JEOL JEM‑ARM200FTH), XRD (Bruker D2), UV–Vis (HITACHI U‑3900), photoluminescence (PerkinElmer LS55), XPS (ULVAC‑PHI PHI Quantera II), UPS (PHI 5000 Versaprobe II), FTIR (Bruker Vertex 80v), and solid‑state ^11B/^13C NMR (Bruker Avance III 400 MHz, 9.4 T). Time‑resolved PL employed a pulsed nitrogen laser (337.1 nm) and a photon‑counting PMT.
BCNO Preparation
Two BCNO series were synthesized: (i) guanidine hydrochloride (BH) and (ii) melamine (MH) as nitrogen sources. In all cases, boric acid (B) and hexamethylenetetramine (HMT) served as boron and carbon sources. The B:HMT ratio was fixed at 3:1, while HMT molar ratio varied from 0.1 to 0.3 to modulate carbon content (samples BMHH01/ BGH01 and BMH03/BGH03). Calcination times (0.5–12 h) and temperatures (600–800 °C) were varied systematically.
Purification
BCNO powders were purified by centrifugation (6000 rpm, 10 min) in water/ethanol (1:10 v/v), redissolved in water, diluted with ethanol, and drop‑cast onto carbon‑coated copper grids for TEM or silicon wafers for SEM/XPS.
Methylene Blue Degradation
Photocatalytic activity was evaluated by degrading 10 ppm methylene blue (MB) under a 100 W Xe lamp (250–1100 nm). 10 mg of catalyst was dispersed in 15 mL MB solution, stirred in the dark for 10 min, then irradiated. Aliquots were sampled every 20 min up to 80 min and analyzed by UV–Vis. Kinetic constants were derived from the Langmuir–Hinshelwood model.
Results and Discussion
Synthesis and Morphology
BCNO prepared with guanidine hydrochloride at 800 °C for 12 h formed quasi‑spherical nanodisks (D = 6.7 ± 1.1 nm) with a turbostratic h‑BN lattice (2θ ≈ 26.6° and 43.1°). TEM revealed a (002) lattice spacing of 0.338 nm, confirming high crystallinity. In contrast, melamine‑derived BCNO annealed at 600 °C produced multilayered graphene‑oxide sheets with ill‑defined edges and micron‑scale aggregates, as shown by TEM and SEM.
Structural Characterization
For BGH01, XPS indicated B ≈ 40 %, N ≈ 38 %, C ≈ 8 %, O ≈ 14 %. Deconvolution of B 1s and N 1s peaks identified B–O and B–N bonds, while O 1s confirmed B–O bonding. ^11B MAS NMR revealed tricoordinate BN₂(OH) species (δ = 28.3 ppm) and BN(OH)₂ or BNO(OH) sites (δ = 20 ppm), supporting an O‑doped h‑BN framework. In contrast, BMH01 exhibited a dominant carbon composition (≈ 75 %) and XPS showed BCN/BN bonding at 191.7–192.4 eV. ^13C CP‑MAS NMR displayed sp² C=C signals (≈ 40 ppm) and graphitic Cα/Cβ peaks at 160/154 ppm, confirming BCNO‑doped graphene‑oxide structure. Solid‑state ^11B NMR for BMH revealed a high fraction of tetracoordinate B(IV) sites (≈ 55 %) compared to BGH (≈ 3 %), indicating distinct boron coordination environments.
Optical Properties
UV–Vis absorption of BGH01 showed a featureless spectrum (band gap ≈ 5.7 eV). BGH03 displayed a secondary absorption peak at 330 nm due to C‑related impurity states. BMH01/03 exhibited strong UV absorption at ~240 nm (π→π* of C=C) and a 288 nm band (n→π* of C=O/C=N). PL spectra excited at 365 nm revealed BGH01 emission at 412, 445, and 489 nm, attributed to B–O luminescence centers and V_N–C/O defect transitions; BGH03 red‑shifted to 506 nm. BMH01/03 emitted at 429–447 nm with broader, lower‑energy peaks, consistent with defect‑mediated recombination. Band gaps were estimated from Tauc plots: 5.7 eV (BGH01), 3.8 eV (BGH03), and 4.2 eV (BMH01/03). Time‑resolved PL lifetimes were 1.58, 2.10, 5.18, and 8.14 µs for BGH01, BGH03, BMH01, and BMH03, respectively, indicating long‑lived charge carriers that favor photocatalysis.
Photocatalytic Activity
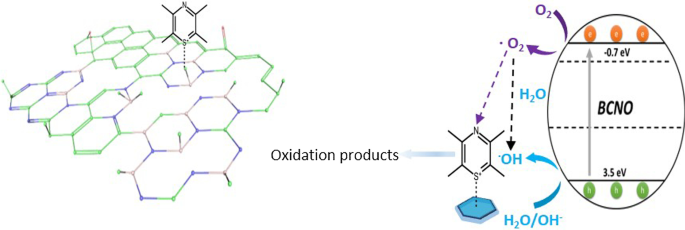
Under visible light, BMH03 achieved a methylene blue degradation rate constant of 2.31 × 10⁻³ min⁻¹, outperforming BGH01 (1.52 × 10⁻³ min⁻¹), BMH01 (1.48 × 10⁻³ min⁻¹), and BGH03 (9.38 × 10⁻⁴ min⁻¹). BMH03’s superior activity is attributed to its high graphitic sp² C=C content and optimal BN(OH)₂/BN₂(OH) sites, which enhance charge separation and provide Lewis acidic adsorption sites for MB. The mechanism involves photo‑generated holes forming hydroxyl radicals that oxidize MB, while electrons reduce O₂ to superoxide radicals that further degrade the dye.
Structure–Activity Relationship
Active catalysts (BGH01, BMH01, BMH03) exhibit a high fraction of tricoordinate BN(OH)₂/BN₂(OH) sites and substantial graphitic domains. Inactive forms (BGH01LT) are dominated by tetracoordinate B(IV) sites, which reduce Lewis acidity and suppress activity. Thus, local boron coordination and graphitic content are critical determinants of photocatalytic performance.
Conclusions
We have demonstrated that BCNO is a versatile, metal‑free photocatalyst whose activity can be tuned by selecting nitrogen precursors, annealing temperature, and carbon content. Guanidine‑derived BCNO nanodisks (800 °C) exhibit moderate activity via B–OH sites, whereas melamine‑derived BCNO graphene‑oxide sheets (600 °C) display superior activity, surpassing graphitic carbon nitride. The key to high performance lies in a high density of tricoordinate boron sites and graphitic sp² C=C domains that promote charge separation and adsorption. This work establishes BCNO as a promising platform for sustainable solar‑driven energy conversion and environmental remediation, and invites further structural optimization for applications such as hydrogen production and CO₂ reduction.
Availability of Data and Materials
All data are fully available without restriction.
Abbreviations
- BCNO
- Boron carbon oxynitride
- CN
- Carbon nitride
- hBN
- Hexagonal boron nitride
- TRPL
- Time‑resolved photoluminescence
- MAS
- Magic angle spinning
- CP
- Cross polarization
- SOLA
- Solid line shape analysis
- I
- Intensity
- ATR
- Attenuated total reflectance
Nanomaterials
- Carbon Fiber: Composition, Manufacturing, and Future Applications
- Carbon Monoxide Detectors: Technology, Design, and Safety Standards for Home Protection
- Carbon Paper: History, Materials, and Modern Manufacturing
- Carbon M2: 20× Faster Digital Light Synthesis for High‑Resolution Production Parts
- Scientists Successfully Synthesize and Visualize Cyclo[18]Carbon
- Enhanced Visible‑Light Photocatalytic Degradation of Rhodamine B Using Bi<sub>4</sub>Ti<sub>3</sub>O<sub>12</sub>/Ag<sub>3</sub>PO<sub>4</sub> Heterojunction Nanocomposites
- B-Doped gh-C₃N₄ Exhibits Robust Metal-Free Half-Metallicity for Spintronics
- Flexible Full-Cell Lithium‑Ion Battery Using Electrospun Carbon Nanofibers and a Simple Plastic Packaging Method
- Double‑Gated Nanohelix as a Tunable Binary Superlattice: Band Engineering and Optoelectronic Prospects
- How Multi‑Walled Carbon Nanotubes Promote Tomato Lateral Root Development via Nitric Oxide Signaling



