Nanoparticle-Enhanced Wormlike Micellar System: Design, Rheology, and Mechanistic Insights
Abstract
We introduce a nanoparticle‑enhanced wormlike micellar system (NEWMS) built from cetyltrimethylammonium bromide (CTAB) and sodium salicylate (NaSal) and reinforced with silica nanoparticles. Prior to mixing, we characterized the silica nanofluid by dynamic light scattering (DLS) and transmission electron microscopy (TEM) to confirm size uniformity and dispersion stability. Rheological testing reveals that the zero‑shear viscosity of NEWMS rises with increasing silica loading, while oscillatory measurements confirm typical viscoelastic behavior. Compared with unmodified wormlike micelles, the entanglement length and mesh size remain essentially unchanged, but the contour length grows as silica concentration increases, highlighting the reinforcing role of the nanoparticles. We propose a mechanistic model that attributes the viscosity enhancement to micelle‑nanoparticle junctions acting as physical cross‑links, thereby expanding the micellar network. This study deepens our understanding of NEWMS and broadens their potential applications in fields ranging from enhanced oil recovery to advanced fluid formulations.
Background
Self‑assembly of surfactants into diverse aggregates—spherical micelles, rod‑like micelles, wormlike micelles, vesicles, lamellae, and liquid crystals—has become a cornerstone of modern soft‑matter science and industrial technology. Above the critical micelle concentration (cmc), surfactants form spherical micelles; further concentration increases drive the formation of elongated structures such as wormlike micelles, which exhibit viscoelasticity due to the transient entanglement of their thread‑like aggregates. Wormlike micelles are widely exploited as drag‑reducing agents, fracturing fluids for oil recovery, and key ingredients in cosmetic formulations. Unlike conventional polymer solutions, wormlike micelles can reversibly break and reform under temperature, shear, or chemical stimuli, making their stability a critical design challenge.
Previous efforts to reinforce wormlike micelles have explored polymer additives, gemini surfactants, and ionic strength modulation. For instance, the addition of hydrophobically modified polyacrylamide to a cationic surfactant system increased viscosity, while gemini surfactants produced micelles with superior viscoelastic properties. More recently, nanoparticles have emerged as powerful modulators of soft‑matter rheology. Silica nanoparticles, for example, have been shown to increase the zero‑shear viscosity and relaxation time of CTAB/NaNO3 wormlike micelles, attributed to micelle‑nanoparticle junctions acting as physical cross‑links. Despite these advances, systematic studies of silica concentration effects on CTAB/NaSal wormlike micelles remain scarce.
In this work, we synthesize a novel NEWMS by combining 50 mM CTAB and 60 mM NaSal with silica nanoparticles of 7–40 nm diameter. We examine how varying silica mass fractions (0.1, 0.3, 0.5 wt %) influence micellar morphology, rheology, and network structure, providing quantitative insights into the underlying mechanisms.
Methods
Materials
CTAB and NaSal were sourced from Shanghai Experimental Reagent Co., Ltd. Silica nanoparticles (7–40 nm) were supplied by Aladdin Industrial Co., Ltd. All solutions were prepared with triply distilled water.
Sample Preparation
Silica nanofluids were prepared by dispersing silica at 0.1, 0.3, and 0.5 wt % in water, stirring at 340 rpm for 30 min, followed by 3 h ultrasonication to achieve transparent suspensions. For NEWMS, the nanofluid served as the base solvent; CTAB (100 mM) and NaSal (120 mM) solutions were individually ultrasonicated at 35 °C for 10 min before mixing equal volumes. The mixture was stirred for 30 min to form the micellar network. Control samples without silica were prepared identically.
Characterizations
Transmission Electron Microscopy
Silica nanoparticles were imaged on a JEOL JEM‑2100 microscope.
Dynamic Light Scattering
DLS measurements were performed on a Malvern Zetasizer Nano ZS (λ = 633 nm, θ = 90°) at 25 ± 0.1 °C, with each sample measured three times.
Rheological Measurements
Steady‑shear and oscillatory rheology were conducted on a Haake Mars 60 rheometer (cone‑plate, 35 mm, 1°) at 25 ± 0.05 °C. Shear rates ranged from 0.01 to 100 s⁻¹. For oscillatory tests, the frequency was fixed at 1 Hz, and stress sweeps established the linear viscoelastic regime before performing frequency sweeps. All micellar solutions were equilibrated at 25 °C for 24 h before testing to ensure full micelle formation.
Results and Discussion
Formation of Silica Nanofluids
Figure 1 displays the TEM micrograph of the silica particles. The particles exhibit a narrow size distribution, while slight aggregation occurs due to interparticle forces. DLS data (Table 1) show a gradual increase in hydrodynamic diameter with higher silica loading, reflecting enhanced aggregation. Zeta‑potential measurements indicate the 0.3 wt % nanofluid possesses the most negative surface charge, suggesting optimal colloidal stability.
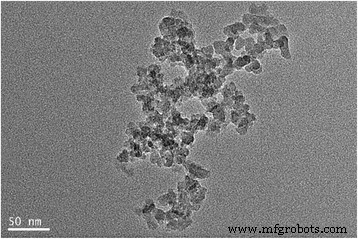
The TEM micrograph of silica nanoparticles
Rheological Properties of NEWMS
Figure 2 shows steady‑shear viscosities for the three silica concentrations. At low shear rates, all solutions exhibit a Newtonian plateau, defining the zero‑shear viscosity (η₀). Increasing silica content systematically raises η₀, indicating a stronger, more entangled network. Under higher shear, all samples display pronounced shear‑thinning, characteristic of wormlike micelles, with the rate of decline accelerating as silica loading rises, likely due to alignment and possible shear‑banding.
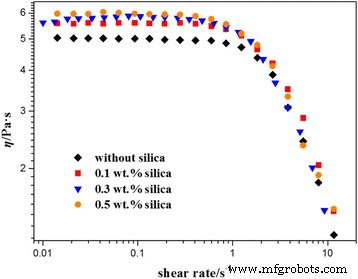
Steady‑shear viscosities of wormlike micelle solutions with varying silica mass fraction at 25 °C
Oscillatory data (Figure 3a) reveal the expected transition from viscous‑dominated (G″ > G′) at low frequencies to elastic‑dominated (G′ > G″) at high frequencies. The plateau modulus (G₀) and minimum loss modulus (G″min) are only modestly affected by silica, whereas both G′ and G″ values shift upward with higher silica loading, underscoring enhanced viscoelasticity.
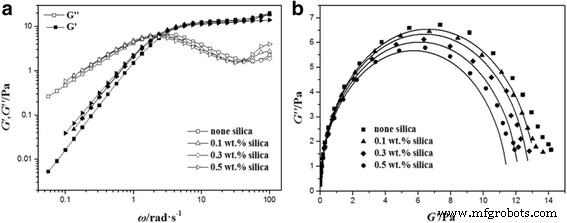
Variations of G′ (filled symbols) and G″ (open symbols) with shear frequency and Cole–Cole plots for NEWMS at 25 °C
Applying the Maxwell model (Eqs. 1–3) yields relaxation times (τ_R) that increase with silica content, while the breakage time (τ_break) remains unchanged. This indicates that silica primarily extends the reptation time (τ_rep) rather than influencing micelle scission dynamics. The calculated mesh size (ξ_M) and entanglement length (l_e) remain nearly constant, but the contour length (L) shows a clear upward trend (Table 2), corroborating the visual impression of longer micellar strands in the presence of silica.
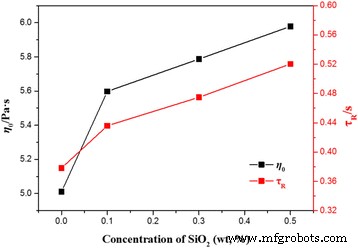
Dependences of the zero‑shear viscosity η₀ and the relaxation time τ_R on the concentration of silica nanoparticles at 25 °C
Mechanistic Insight
The viscosity enhancement can be attributed to micelle‑nanoparticle junctions that serve as physical cross‑links. The hydrophilic silica surface adsorbs micellar end‑caps, effectively reducing the energetic penalty for micelle extension and promoting linear growth. Each junction connects two micellar strands, increasing network connectivity and entanglement density. Consequently, the system behaves as a “double‑network” material, where the silica‑mediated links reinforce the entangled micellar matrix. This mechanism aligns with prior observations of nanoparticle‑induced cross‑linking in polymer and surfactant systems.
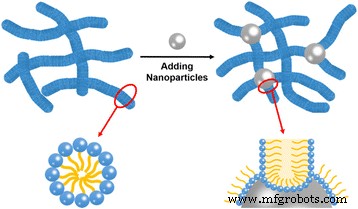
Illustration of the proposed mechanism of a complex cross‑linking network constructed by wormlike micelles and silica nanoparticles
Conclusions
We have successfully fabricated a nanoparticle‑enhanced wormlike micellar system by combining CTAB/NaSal with silica nanoparticles. Rheological measurements demonstrate that the addition of silica markedly increases zero‑shear viscosity, relaxation time, and contour length, while leaving the mesh size and entanglement length largely unchanged. The formation of micelle‑nanoparticle junctions provides a robust cross‑linking network that underpins the observed viscoelastic improvements. This work establishes a clear mechanistic framework for tailoring micellar rheology via nanoparticle additives, with implications for enhanced oil recovery, polymer‑free drilling fluids, and advanced functional liquids.
Abbreviations
- cmc
Critical micelle concentration
- cryo-TEM
Cryogenic transmission electron microscopy
- DLS
Dynamic light scattering
- NEWMS
Nanoparticle‑enhanced wormlike micellar system
- TEM
Transmission electron microscopy
Nanomaterials
- Case Study: Honda Lincoln Plant – Engineering Excellence & Reliability
- Optimizing Emulsion Droplet Size and PVA Surfactant to Enhance Stability of Quantum‑Dot Micellar Nanocrystals
- Calculating Conveyor Speed: A Practical Guide for Production Efficiency
- Transforming MEP: From Local Systems to a National Manufacturing Network
- HydroMazing Smart Garden System: Advanced IoT Plant Care
- Understanding Portable Braille Displays: Key Insights & Innovations
- Hydraulic Leak Hazards: Protecting Your System & Safety
- The American Manufacturing System: History, Techniques, and Impact
- Key Components of an Effective Robot System
- Why Hydraulic Systems Are Essential for Modern Engineering



