Impact of Li/Nb Ratio on Hydrothermal Synthesis and Photocatalytic Performance of Li‑Nb‑O Compounds
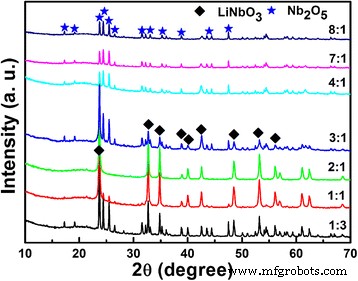
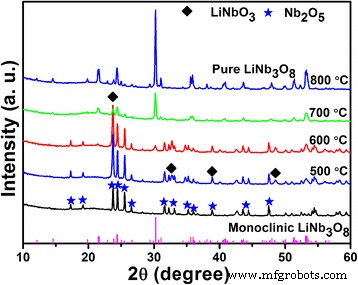
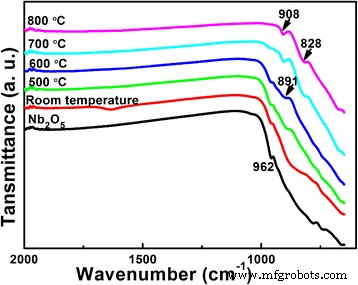
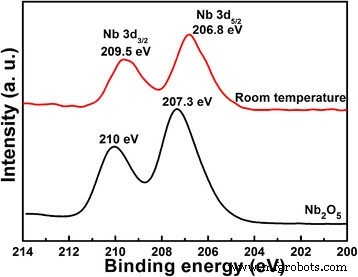
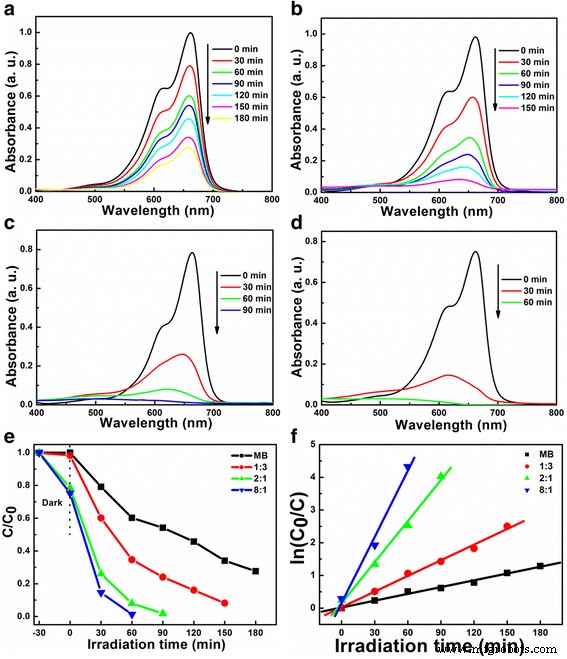
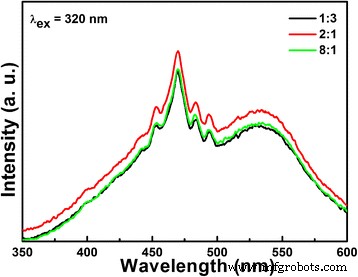
We investigated how the lithium‑to‑niobium (Li/Nb) molar ratio influences the hydrothermal synthesis of Li‑Nb‑O compounds and their subsequent photocatalytic activity. A Li/Nb ratio below 3:1 promotes the formation of LiNbO3, whereas ratios above 3:1 suppress LiNbO3 formation and instead modify the Nb2O5 precursor through lithium incorporation. Excess LiOH drives the formation of Li3NbO4 rather than LiNbO3, and any locally formed LiNbO3 dissolves in the strongly alkaline medium. Remarkably, pure LiNb3O8 powders were synthesized using two opposite Li/Nb ratios (8:1 and 1:3). The 8:1 sample displays a porous, hollow morphology that confers a 4.2‑fold higher photocatalytic activity for methylene blue (MB) degradation compared to the 1:3 counterpart, attributable to its high surface area and layered crystal structure, which enhance charge separation. Niobium‐based materials, including oxides, alkali niobates, and columbite niobates, are prized for their versatile physical properties and wide range of applications—from catalysis [1,2,3] and memristors [4] to dye‑sensitized solar cells [5] and optical devices [6,7]. LiNbO3, a prototypical alkali niobate, exhibits remarkable electro‑optical, nonlinear optical, pyroelectric, and piezoelectric behavior, making it a cornerstone of optical modulators, waveguides, and acoustic transducers. For environmental remediation and energy conversion, niobates such as (Na,K)NbO3 [8], BiNbO4 [9], LiNbO3 [10], and LiNb3O8 [11] have attracted intense scrutiny. Their distorted NbO6 octahedra facilitate charge carrier delocalization [12], while Nb4+ 4d orbitals lower the redox potential of H+/H2, promoting efficient separation and transfer of photogenerated carriers [13]. LiNb3O8 is especially noteworthy: as a lithium‑ion battery anode, it offers a theoretical capacity of 389 mAh g−1 (two‑electron transfer) [14,15]; as a supercapacitor, its nanoflakes maintain negligible capacitance loss after 15,000 cycles [16]; and as a photocatalyst, it produces 83.87 µmol h−1 of hydrogen under UV irradiation while remaining inert to visible light due to its 3.9 eV band gap [17,18]. LiNb3O8 often emerges as an impurity during LiNbO3 synthesis, especially in high‑temperature or unevenly distributed precursor systems [19,20]. Traditional LiNbO3 production methods include sol‑gel [19], hydrothermal [21], and laser‑irradiation [22]. Hydrothermal synthesis offers low‑temperature, environmentally friendly processing with homogeneous particle size distribution, but the effect of Li/Nb ratios exceeding 1:1 has not been thoroughly explored. This study addresses that gap. The Li‑Nb‑O powders were prepared hydrothermally using lithium hydroxide monohydrate (LiOH·H2O; ≥ 98 %) and niobium pentoxide (Nb2O5; 99.9 %). 3.5 mmol of Nb2O5 was dispersed in 35 mL deionized water with LiOH·H2O to achieve Li:Nb molar ratios of 1:3, 1:1, 2:1, 3:1, 4:1, 5:1, 6:1, 7:1, and 8:1. Samples with similar outcomes (4:1, 5:1, 6:1, 7:1) were grouped; only 4:1 and 7:1 are reported in detail. The suspensions were sealed in 50‑mL Teflon‑lined autoclaves and heated to 260 °C for 24 h, then cooled naturally. Powders were washed with water and ethanol, dried at 60 °C, and finally calcined at 500–800 °C (2 h, 5 °C min−1 ramp). X‑ray diffraction (XRD) employed a Bruker D8 Discover diffractometer (Cu Kα, 40 kV, 40 mA). Morphology was examined by FESEM (JSM‑6700F). Chemical bonds were probed via FTIR (2000–650 cm−1), and surface composition by XPS (Thermo‑Fisher Escalab 250Xi). Specific surface area was measured by BET (Micromeritics ASAP 2460). Photoluminescence (PL) spectra used an F‑280 fluorescence spectrophotometer (excitation 320 nm). Photocatalytic activity was evaluated by degrading 5 mg L−1 methylene blue (MB) under a 500 W Hg lamp. 50 mg of catalyst was dispersed in 50 mL MB solution, equilibrated in the dark for 1 h, then irradiated. Residual MB was quantified at 665 nm every 30 min with a UV‑vis‑NIR spectrophotometer. Figure 1 shows XRD patterns of products from different Li/Nb ratios. Pure LiNbO3 (JCPDS 20‑0631) appears at Li:Nb = 2:1. Ratios below 2:1 (1:1, 1:3) still yield LiNbO3 but with residual Nb2O5 (JCPDS 37‑1468), indicating insufficient Li for complete conversion. When Li is in excess (≥ 4:1), LiNbO3 disappears entirely; only Nb2O5 remains in the XRD patterns. This suggests that excess LiOH promotes the formation of Li3NbO4, and any nascent LiNbO3 is dissolved by the alkaline medium. XRD patterns of Li‑Nb‑O powders after hydrothermal reaction with different Li/Nb mole ratios. To elucidate the phase evolution at high Li content, Li/Nb = 8:1 samples were calcined at 500–800 °C (Figure 2). At 500 and 600 °C, a LiNbO3 peak emerges, confirming the presence of Li in the post‑hydrothermal product. A diffraction at 30.26° (monoclinic LiNb3O8, (410) plane) appears at 600 °C, following the reaction: LiNbO3 + Nb2O5 → LiNb3O8 [24]. At 700 °C, monoclinic LiNb3O8 dominates; at 800 °C, pure LiNb3O8 (JCPDS 36‑0307, space group P21/a) is obtained, providing a new, low‑temperature route to this phase. XRD patterns of Li‑Nb‑O powders (Li:Nb = 8:1) after calcination at different temperatures for 2 h. FTIR spectra (Figure 3) corroborate these findings. The Nb=O stretch at 962 cm−1 persists up to 700 °C, confirming the presence of Nb2O5. At 500 and 600 °C, a new band at 891 cm−1 appears and vanishes by 700 °C, matching the LiNbO3 formation and consumption. At 700 and 800 °C, bands at 908 and 828 cm−1 indicate LiNb3O8 formation, aligning with XRD data. FTIR spectra of Nb2O5 raw material and Li‑Nb‑O powders (Li:Nb = 8:1) calcined at different temperatures. Li/Nb ratios critically influence LiNbO3 formation: ratios < 3:1 favor LiNbO3, whereas > 3:1 suppress it, with excess Li forming Li3NbO4 or LiNb3O8 [28]. Excess LiOH dissolves any nascent LiNbO3 due to its high alkalinity [29]. XPS analysis (Figure 4) shows both raw Nb2O5 and post‑hydrothermal products contain Nb5+ (Nb 3d3/2/3d5/2 splitting of 2.7 eV). The binding energy shifts ~0.5 eV to lower values after hydrothermal treatment, indicating a changed chemical environment caused by Li⁺ proximity. XPS spectra of Nb2O5 raw material and products (Li:Nb = 8:1) after hydrothermal treatment. SEM images (Figure 5) reveal that hydrothermal processing transforms large, irregular Nb2O5 crystals into smaller (~200 nm) particles that still aggregate. This morphological refinement is attributed to the high LiOH concentration during hydrothermal synthesis. SEM images of a Nb2O5 raw material and b Li:Nb = 8:1 hydrothermal product. Calcination at 800 °C of Li/Nb = 1:3, 2:1, and 8:1 yielded distinct phases (Figure 6). Li:Nb = 2:1 produced pure LiNbO3, while 1:3 and 8:1 each yielded pure LiNb3O8. Other ratios produced mixed LiNbO3/LiNb3O8 phases. SEM images (Figure 7) highlight morphological differences: LiNb3O8-8:1 forms a porous, honeycomb‑like network of micrometer‑scale nanoparticles, whereas LiNb3O8-1:3 aggregates into dense particles. BET measurements confirm the higher surface area of LiNb3O8-8:1 (4.46 m2 g−1) versus LiNb3O8-1:3 (0.96 m2 g−1). The porous morphology arises from lithium volatilization during calcination, creating new LiNb3O8 particles and interparticle networks [11]. LiNbO3 calcined at 800 °C shows ~200 nm grains with irregular shapes and a BET area of 3.91 m2 g−1. SEM images of Li‑Nb‑O powders (a) Li:Nb = 2:1 at 500 °C, (b) Li:Nb = 1:3, (c) Li:Nb = 8:1, and (d) Li:Nb = 2:1 at 800 °C. Photocatalytic performance was evaluated by MB degradation (Figure 8). LiNb3O8-8:1 achieved ~85 % degradation after 30 min under UV, outperforming LiNb3O8-1:3, LiNbO3, and the catalyst‑free control. Rate constants (k) derived from pseudo‑first‑order kinetics were 0.71 × 10−2, 1.61 × 10−2, 4.18 × 10−2, and 6.73 × 10−2 min−1 for the control, LiNb3O8-1:3, LiNbO3, and LiNb3O8-8:1, respectively. LiNb3O8-8:1’s k is 9.5× the control, 4.2× LiNb3O8-1:3, and 1.6× LiNbO3. The superior activity stems from the high surface area and porous structure, providing abundant active sites. UV‑vis spectra of MB degradation: (a) control, (b) LiNb3O8-1:3, (c) LiNbO3, (d) LiNb3O8-8:1; (e) degradation curves; (f) kinetic fits. Compared to LiNbO3, LiNb3O8-8:1’s enhanced photocatalytic performance is also attributed to its layered crystal structure, which reduces symmetry and promotes charge separation, as evidenced by PL spectra (Figure 9). LiNb3O8 shows weaker emission at ~470 nm, indicating longer carrier lifetimes and more efficient interfacial transfer. Its slightly smaller band gap (~3.9 eV vs. 4.14 eV) allows broader light absorption. Room‑temperature PL spectra of LiNb3O8-1:3, LiNbO3, and LiNb3O8-8:1. PL data confirm that LiNb3O8 achieves superior charge carrier separation, directly translating to higher photocatalytic activity. The Li/Nb molar ratio is a decisive factor in the hydrothermal synthesis of Li‑Nb‑O compounds. Ratios below 3:1 favor LiNbO3 formation, whereas ratios above 3:1 suppress it and instead yield LiNb3O8 (when Li/Nb = 8:1) or retain Nb2O5 (when Li/Nb = 1:3). Excess LiOH promotes Li3NbO4 formation and can dissolve nascent LiNbO3 due to high alkalinity. Pure LiNb3O8 powders were synthesized at both 8:1 and 1:3 Li/Nb ratios; the 8:1 sample’s porous, hollow morphology yields a 4.2‑fold higher photocatalytic activity for MB degradation compared to the 1:3 counterpart. Compared to LiNbO3, LiNb3O8-8:1’s layered structure and reduced symmetry enhance electron–hole separation, explaining its superior photocatalytic performance.Abstract
Background
Methods
Results and Discussion


Conclusions
Nanomaterials
- Tuning Morphology, Optics, and Conductivity of Al₂O₃/ZnO Nanolaminates Through Bilayer Thickness Control
- Hollow‑Structured LiNb3O8 Photocatalysts: Synthesis, Characterization, and Superior Methylene Blue Degradation
- Coupling of Surface Plasmon Polaritons and Magnetic Dipole Resonances in Ag Nanodisk Metamaterials
- Influence of Peptizing Acid on TiO₂ Phase Composition and Photocatalytic Efficiency: A Comparative Study of Sulfuric, Nitric, and Acetic Acids
- Impact of Etching Parameters on Ge/Si FinFET Channel Fabrication and Device Performance
- Optimizing ZnO‑Based Nanohybrids: How Materials, Heterojunctions, and Crystal Orientation Enhance Methyl Orange Degradation
- How Ambient Gases Influence Electrical Performance in Solution-Processed C8-BTBT Thin-Film Transistors
- Engineering High-Quality Graphene/TMD Heterostructures: Fabrication and Spectral Analysis
- Optimizing β-Phase PVDF Films and Nanofibers for Superior Electroactive Performance
- Precision Metal Cutting with Plasma & Oxy/Fuel Systems



