Efficient One‑Pot Synthesis of Hierarchical Silicalite‑1/Ag Nanoparticle Catalyst for Rapid 4‑Nitrophenol Reduction
Abstract
We report a streamlined, one‑pot approach to encapsulate silver nanoparticles (Ag NPs) within hierarchical porous silicalite‑1 (HPS‑1), yielding a catalyst with an exceptionally high surface area (243 m2 · g−1) and dual pore sizes (1.75 nm and 3.96 nm). Comprehensive characterization by TEM, XRD, FTIR, and N2 adsorption–desorption confirms uniform dispersion of Ag NPs (≈25 nm) and preservation of the MFI framework. When applied to the aqueous reduction of 4‑nitrophenol (4‑NP) to 4‑aminophenol (4‑AP) at room temperature, the catalyst delivers a pseudo‑first‑order rate constant of 4.75 × 10−3 s−1 and achieves >98 % conversion within 20 min. Remarkably, the catalyst retains its activity after ten consecutive runs, underscoring the stability imparted by the hierarchical support.
Background
Metal nanoparticle (MNP) stability is pivotal for heterogeneous catalysis, as aggregation or leaching diminishes activity and selectivity. Traditional strategies—surfactant‑stabilized MNPs followed by porous coatings, or pre‑deposited MNPs on supports with subsequent encapsulation—often involve complex, multi‑step procedures and can mask active sites. While noble metals such as Pd, Au, and Pt offer excellent performance, their scarcity and cost drive interest in earth‑abundant alternatives (Co, Ni, Cu). However, non‑noble metal catalysts typically suffer from deactivation through agglomeration. Encapsulation within porous matrices presents a promising route to preserve dispersion and prevent sintering, yet many existing methods remain laborious and inefficient. Consequently, there is a clear need for a simple, scalable synthesis that embeds MNPs within a robust, high‑surface‑area support.
In this study, we introduce a one‑pot synthesis of Ag NPs encapsulated in hierarchical porous silicalite‑1 microspheres (Ag@HPS‑1). The support’s meso‑ and microporosity, combined with a large surface area, enhances mass transfer and stabilizes the embedded Ag NPs during catalytic cycles. This design not only achieves superior catalytic performance for 4‑NP reduction but also offers a platform for future bimetallic or multifunctional systems.
Methods
Preparation of Hierarchical Porous Silicalite‑1 Encapsulated Ag NPs
The Ag@HPS‑1 catalyst was synthesized by mixing 5.0 g of 40 wt% Ludox HS‑40 colloidal silica with 20 mL deionized water, stirring for 30 min, and adjusting the pH to 12 with 25 wt% NH3·H2O. AgNO3 was added dropwise, followed by stirring at 80 °C overnight to remove water. Next, 2.0 g of 25 wt% tetrapropylammonium hydroxide (TPAOH) was incorporated, and the mixture was transferred to a 25 mL Teflon‑lined autoclave for hydrothermal crystallization at 120 °C for 48 h. The solid was calcined at 550 °C in air for 8 h to eliminate organics, then reduced in 5 vol% H2/Ar at 400 °C for 6 h. The HPS‑1 support was prepared identically, omitting AgNO3.
Catalytic Reduction of 4‑Nitrophenol by Ag@HPS‑1 Catalyst
In a typical run, 20 mL of Ag@HPS‑1 suspension (0.8 g L−1) was mixed with 5 mL of 3 mM 4‑NP and 5 mL of 0.3 M NaBH4, stirred at room temperature. Periodic aliquots were withdrawn, placed in a quartz cuvette, and measured by UV‑Vis spectroscopy. After each cycle, the catalyst was recovered by centrifugation, washed thrice with deionized water and ethanol, and reused under identical conditions.
Characterization of Physical and Catalytic Properties
Transmission electron microscopy (TEM) was performed on a Tecnai G2F30 (200 kV). X‑ray diffraction (XRD) employed a Rigaku D/max‑2400 with Cu‑Kα radiation (10°–90° 2θ). N2 adsorption–desorption was measured on an ASAP2020 after degassing at 393 K for 8 h; specific surface area, pore volume, and pore size were calculated by BET and BJH methods. Fourier transform infrared (FTIR) spectra were recorded with a NEXUS 670 spectrophotometer (4000–400 cm−1). UV‑Vis spectra were obtained using a UV2800PC UV‑vis spectrophotometer.
Results and Discussion
The Ag@HPS‑1 catalyst was prepared following a modified one‑pot protocol, yielding a uniform distribution of ~25 nm Ag NPs within the hierarchical silicalite‑1 microspheres. The Ag loading, measured by ICP‑OES, was 2.96 wt %.
SEM images (Fig. 1) reveal that both HPS‑1 and Ag@HPS‑1 possess well‑defined spherical morphology, with the latter exhibiting a slightly larger average diameter (~1.25 µm). TEM and HRTEM images (Fig. 2) confirm the crystalline MFI framework and the presence of Ag NPs encapsulated in the porous matrix. The Ag NPs remain well‑separated, avoiding aggregation.

a SEM image of HPS‑1 support and b Ag@HPS‑1 catalyst

a–c TEM and HRTEM images of HPS‑1; d–f TEM and HRTEM images of Ag@HPS‑1 catalyst
XRD patterns (Fig. 3a) show characteristic MFI peaks for both samples and additional reflections at 2θ = 38.1°, 44.3°, 64.4°, and 77.4° corresponding to the (111), (200), (220), and (311) planes of face‑centred cubic Ag, confirming the metallic state after reduction. FTIR spectra (Fig. 3b) display typical Si–O–Si vibrations (∼1105 cm−1 and ∼799 cm−1) with negligible changes upon Ag loading, indicating that the support’s chemistry remains intact.
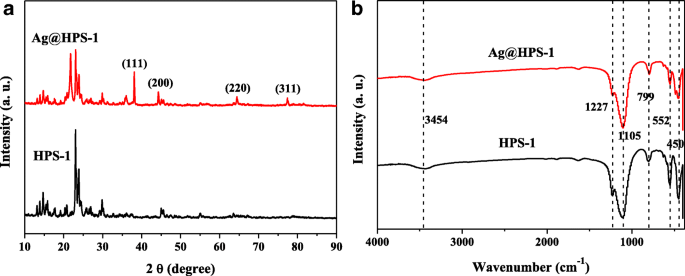
a XRD patterns; b FTIR spectra of HPS‑1 and Ag@HPS‑1
BET analysis (Fig. 4) reveals that HPS‑1 has a surface area of 413 m2 · g−1 and a total pore volume of 0.394 cm3 · g−1, with dual pore sizes of 1.76 nm and 3.67 nm. Ag@HPS‑1 retains hierarchical porosity (1.75 nm, 3.96 nm) but shows a modest decrease in surface area (243 m2 · g−1) and pore volume (0.176 cm3 · g−1), attributable to Ag NP integration within the framework. The high surface area and dual‑pore architecture facilitate rapid mass transfer and substrate concentration, underpinning the catalyst’s performance.
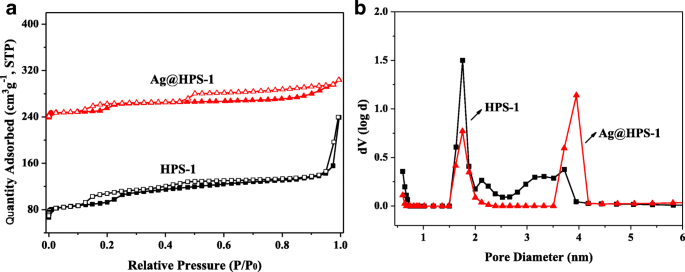
a N2 adsorption–desorption isotherms; b pore size distributions of HPS‑1 and Ag@HPS‑1
The catalytic activity was evaluated by the reduction of 4‑NP to 4‑AP. UV‑Vis monitoring (Fig. 5a) shows a rapid decline of the 400 nm absorption band and the emergence of a 300 nm band, completing within 20 min at 298 K. The pseudo‑first‑order rate constant (k) calculated from ln A vs. time is 4.75 × 10−3 s−1, surpassing many reported Ag‑based systems (e.g., bare Ag NPs: 2.1 × 10−3 s−1; Ag@HTO‑PDA: 3.14 × 10−3 s−1; CNC/CTAB‑Ag: 5.76 × 10−3 s−1; MWCNT‑Ag: 7.88 × 10−3 s−1). This performance is attributed to the synergistic effects of the confined Ag NPs, high surface area, and hierarchical porosity.
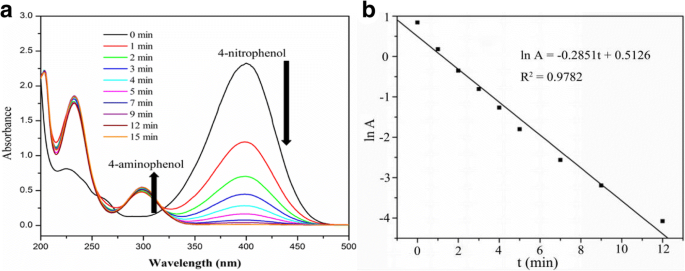
a Time‑dependent UV‑Vis spectra; b ln A vs. time plot for 4‑NP reduction
Recyclability tests (Fig. 6) demonstrate that the catalyst maintains >98 % conversion over ten consecutive runs without noticeable loss of activity. Post‑reaction TEM and XRD confirm that the Ag NPs and zeolite framework remain unchanged, underscoring the robustness of the encapsulation strategy.
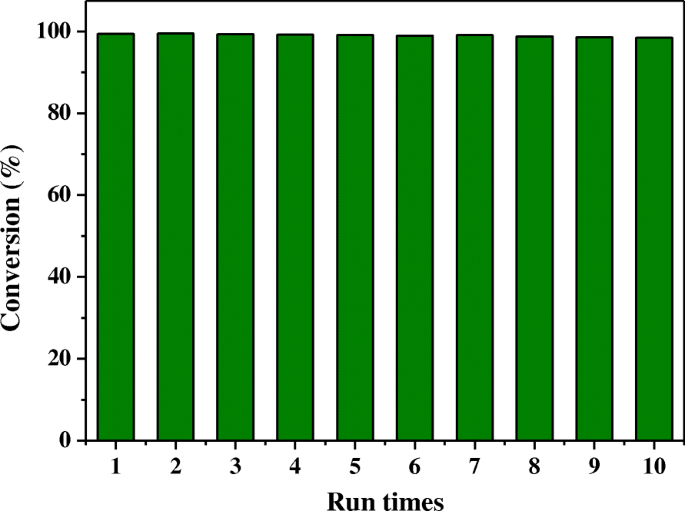
Recycling tests for the reduction of 4‑NP over Ag@HPS‑1 catalyst
Conclusions
We have developed a facile, one‑pot synthesis of Ag NPs encapsulated within hierarchical porous silicalite‑1 microspheres. The resulting Ag@HPS‑1 catalyst exhibits high thermal stability, exceptional reusability, and outstanding activity for the reduction of 4‑NP to 4‑AP, achieving >98 % yield within 20 min at ambient temperature. The confinement of Ag NPs, combined with the support’s high surface area and dual‑pore structure, is key to these properties. This platform can be extended to incorporate other single or bimetallic species, paving the way for diverse catalytic applications.
Abbreviations
- 4‑AP:
4‑Aminophenol
- 4‑NP:
4‑Nitrophenol
- Ag NPs:
Silver nanoparticles
- Ag@HPS‑1:
Hierarchical porous silicalite‑1 encapsulated Ag NPs
- BET:
Brunauer–Emmett–Teller method
- BJH:
Barrett–Joyner–Halenda model
- CNC:
Cellulose nanocrystal
- CTAB:
Hexadecyltrimethylammonium
- FTIR:
Fourier transform infrared spectroscopy
- HPS:
Hierarchical porous silicalite‑1 microspheres
- HTO‑PDA:
Hollow‑tubular‑oriented polydopamine
- MNPs:
Metal nanoparticles
- MWCNTs:
Multi‑walled carbon nanotubes
- PGMA‑SH:
Sulfhydryl‑functionalized poly(glycidyl methacrylate) microspheres
- TEM:
Transmission electron microscopy
- TPAOH:
Tetrapropylammonium hydroxide
- UV‑vis:
Ultraviolet‑visible spectroscopy
- XRD:
X‑ray powder diffraction
Nanomaterials
- Optimizing Sb-Loaded Palygorskite Nanoparticles for Superior Catalytic Hydrogenation of p-Nitrophenol
- Green One‑Pot Synthesis of Ag‑Decorated SnO₂ Microspheres: A Highly Reusable Catalyst for 4‑Nitrophenol Reduction
- Au@TiO₂ Yolk–Shell Nanostructures: Tailored Synthesis and Their Superior Visible‑Light Photocatalytic Degradation and SERS Detection of Methylene Blue
- Hollow‑Structured LiNb3O8 Photocatalysts: Synthesis, Characterization, and Superior Methylene Blue Degradation
- Enhanced Lithium‑Ion Battery Anodes: Polypyrrole‑Coated MnO₂ Core–Shell Micromaterials Deliver Superior Cyclic Stability
- Polypyrrole‑Coated FePt Nanoparticles: A Multifunctional Platform for Photothermal Therapy and Photoacoustic Imaging
- Hydrothermally Synthesized Nano-Copper Sulfide: Superior Adsorption of 17α-Ethynyl Estradiol
- Economical Synthesis and Comparative Catalytic & Antimicrobial Performance of Nb and Ta Selenide/ Telluride Nanocomposites
- One‑Step Hydrothermal Synthesis of Ru/SnₓTi₁₋ₓO₂ Catalysts for Diesel Oxidation: Enhanced Low‑Temperature Activity
- Self‑assembled SnIn4S8/TiO₂ Nanotube Photoanode for Enhanced Visible‑Light Photocathodic Protection of Q235 Steel



